Abstract
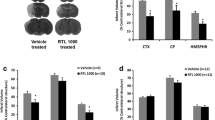
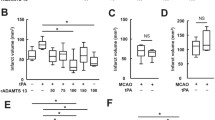
RTL1000 is a partial human MHC molecule coupled to a human myelin peptide. We previously demonstrated that RTL1000 was protective against experimental ischemic stroke in HLA-DR2 transgenic (DR2-Tg) mice. Since thrombolysis with recombinant tissue plasminogen activator (t-PA) is a standard therapy for stroke, we determined if RTL1000 efficacy is altered when combined with t-PA in experimental stroke. Male DR2-Tg mice underwent 60 min of intraluminal middle cerebral artery occlusion (MCAO). t-PA or vehicle was infused intravenously followed by either a single or four daily subcutaneous injections of RTL1000 or vehicle. Infarct size was measured by 2, 3, 5-triphenyltetrazolium chloride staining at 24 or 96 h of reperfusion. Our data showed that t-PA alone reduced infarct size when measured at 24 h but not at 96 h after MCAO. RTL1000 alone reduced infarct size both at 24 and 96 h after MCAO. Combining RTL1000 with t-PA did not alter its ability to reduce infarct size at either 24 or 96 h after MCAO and provides additional protection in t-PA treated mice at 24 h after ischemic stroke. Taken together, RTL1000 treatment alone improves outcome and provides additional protection in t-PA-treated mice in experimental ischemic stroke.


Similar content being viewed by others
References
Fact sheet N 310. The top 10 causes of death. Media center of World Health Organization website. http://who.int/mediacentre/factsheets/fs310/en/index.html. Accessed July 2013.
Wang Y, Zhang Z, Chow N, et al. An activated protein C analog with reduced anticoagulant activity extends the therapeutic window of tissue plasminogen activator for ischemic stroke in rodents. Stroke. 2012;43:2444–9.
Offner H, Subramanian S, Parker SM, et al. Experimental stroke induces massive, rapid activation of the peripheral immune system. J Cereb Blood Flow Metab. 2006;26:654–65.
Offner H, Vandenbark AA, Hurn PD. Effect of experimental stroke on peripheral immunity: CNS ischemia induces profound immunosuppression. Neuroscience. 2009;158:1098–111.
Offner H, Subramanian S, Parker SM, et al. Splenic atrophy in experimental stroke is accompanied by increased regulatory T cells and circulating macrophages. J Immunol. 2006;176:6523–31.
Dirnagl U, Klehmet J, Braun JS, et al. Stroke-induced immunodepression: experimental evidence and clinical relevance. Stroke. 2007;38:770–3.
Muir KW, Tyrrell P, Sattar N, Warburton E. Inflammation and ischemic stroke. Curr Opin Neurol. 2007;20:334–42.
Gee JM, Kalil A, Shea C, et al. Lymphocytes: potential mediators of postischemic injury and neuroprotection. Stroke. 2007;38(2 Suppl):783–8.
Nilupul Perera M, Ma HK, Arakawa S, et al. Inflammation following stroke. J Clin Neurosci. 2006;13:1–8.
Burrows GG, Chang JW, Bachinger HP, et al. Design, engineering and production of functional single-chain T cell receptor ligands. Protein Eng. 1999;12:771–8.
Wang C, Mooney JL, Meza-Romero R, et al. Recombinant TCR ligand induces early TCR signaling and a unique pattern of downstream activation. J Immunol. 2003;171:1934–40.
Vandenbark AA, Rich C, Mooney J, et al. Recombinant TCR ligand induces tolerance to myelin oligodendrocyte glycoprotein 35–55 peptide and reverses clinical and histological signs of chronic experimental autoimmune encephalomyelitis in HLA-DR2 transgenic mice. J Immunol. 2003;171:127–33.
Dziennis S, Mader S, Akiyoshi K, et al. Therapy with recombinant T-cell receptor ligand reduces infarct size and infiltrating inflammatory cells in brain after middle cerebral artery occlusion in mice. Metab Brain Dis. 2011;26:123–33.
Subramanian S, Zhang B, Kosaka Y, et al. Recombinant T cell receptor ligand treats experimental stroke. Stroke. 2009;40:2539–45.
Gonzalez-Gay MA, Zanelli E, Khare SD, et al. Human leukocyte antigen-DRB1*1502 (DR2Dw12) transgene reduces incidence and severity of arthritis in mice. Hum Immunol. 1996;50:54–60.
Stroke therapy academic industry roundtable. Recommendations for standards regarding preclinical neuroprotective and restorative drug development. Stroke. 1999;30:2752–2758.
Albers GW, Goldstein LB, Hess DC, et al. Stroke Treatment Academic Industry Roundable (STAIR) recommendations for maximizing the use of intravenous thrombolytics and expanding treatment options with intra-arterial and neuroprotective therapies. Stroke. 2011;42:2645–50.
Saver JL, Jovin TG, Smith WS, et al. Stroke treatment academic industry roundtable: research priorities in the assessment of neurothrombectomy devices. Stroke. 2013;44:3596–601.
Huan JY, Meza-Romero R, Mooney JL, et al. Rationally designed mutations convert complexes of human recombinant T cell receptor ligands into monomers that retain biological activity. J Chem Technol Biotechnol. 2005;80:2–12.
Chang JW, Mechling DE, Bächinger HP, et al. Design, engineering and production of human recombinant T cell receptor ligands derived from human leukocyte antigen DR2. J Biol Chem. 2001;276:24170–6.
Zhu W, Wang L, Zhang L, et al. Isoflurane preconditioning neuroprotection in experimental focal stroke is androgen-dependent in male mice. Neuroscience. 2010;169:758–69.
Zhang W, Davis CM, Edin ML, et al. Role of endothelial soluble epoxide hydrolase in cerebrovascular function and ischemic injury. PLoS One. 2013;8(4):e61244.
Kilic E, Hermann DM, Hossmann KA. Recombinant tissue plasminogen activator reduces infarct size after reversible thread occlusion of middle cerebral artery in mice. Neuroreport. 1999;10:107–11.
Beerny-Lang MA, Hurst S, Tucker EI, et al. Thrombin mutant W215A/E217A treatment improves neurological outcome and reduces cerebral infarct size in a mouse model of ischemic stroke. Stroke. 2011;42:1736–41.
Pham M, Kleinschnitz C, Helluy X, et al. Enhanced cortical reperfusion protects coagulation factor XII-deficient mice from ischemic stroke as revealed by high-field MRI. Neuroimaging. 2010;49:2907–14.
Momi S, Tantucci M, Van Roy M, et al. Reperfusion of cerebral artery thrombosis by the GPIb-VWF blockade with the Nanobody ALX-0081 reduces brain infarct size in guinea pigs. Blood. 2013;121:5088–97.
Nieswandt B, Kleinschnitz C, Stoll G. Ischaemic stroke: a thrombo-inflammatory disease? J Physiol Lond. 2011;589:4115–23.
Gauberti M, de Martinez LS, Orset C, et al. Lack of secondary microthrombosis after thrombin-induced stroke in mice and non-human primates. J Thromb Haemost. 2014;12:409–14.
Chandler WL, Alessi MC, Aillaud MF, et al. Clearance of tissue plasminogen activator (t-PA) and t-PA/plasminogen activator inhibitor type 1 (PAI-1) complex: relationship to elevated t-PA antigen in patients with high PAI-1 activity levels. Circulation. 1997;96:761–8.
Narita M, Bu G, Herz J, et al. Two receptor systems are involved in the plasma clearance of tissue-type plasminogen activator (t-PA) in vivo. J Clin Invest. 1995;96:1164–8.
Liebeskind DS. Recanalization and reperfusion in acute ischemic stroke. F1000 Med Rep. 2010; 2. pii: 71. doi:10.3410/M2-71.
Itakura A, Aslan JE, Sinha S, et al. Characterization of human platelet binding of recombinant T cell receptor ligand. J Neuroinflammation. 2010;7:75. doi:10.1186/1742-2094-7-75.
Acknowledgments
This work was supported by NIH Grants #NS076013 (STTR) and by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development.
Conflict of Interest
Dr. Offner and OHSU have a significant financial interest in Artielle ImmunoTherapeutics, Inc., a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by the OHSU and VAMC Conflict of Interest in Research Committees.
Wenbin Zhu declares that he has no conflict of interest. Nicole L. Libal declares that she has no conflict of interest. Amanda Casper declares that she has no conflict of interest. Sheetal Bodhankar declares that she has no conflict of interest. Nabil J. Alkayed declares that he has no conflict of interest. All institutional and national guidelines for the care and use of laboratory animals were followed. This article does not contain any studies with human subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, W., Libal, N.L., Casper, A. et al. Recombinant T Cell Receptor Ligand Treatment Improves Neurological Outcome in the Presence of Tissue Plasminogen Activator in Experimental Ischemic Stroke. Transl. Stroke Res. 5, 612–617 (2014). https://doi.org/10.1007/s12975-014-0348-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-014-0348-8