Abstract
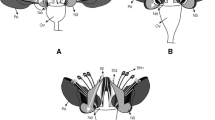
Floral innovations are key for pollinator specialization and play a significant role in plant diversification. Orchidaceae present many examples of floral innovations that allow its high degrees of pollinator specialization and promoted speciation. The rich neotropical genus Telipogon evolved an uncinate (=hook-like) viscidium on the pollinarium and setae-bearing flowers, which are uncommon in Orchidaceae; however, the importance of them on pollination success and whether they are floral innovations or exaptations in Telipogon have not been investigated. Here we investigate the morphology of the viscidium and floral setae within the Telipogon alliance (including the genera Hofmeisterella, Trichoceros and Telipogon), test their significance in pollination, and their occurrence and evolution across the Oncidiinae. We used Telipogon peruvianus as a model species to test whether uncinate viscidium and floral setae increased pollination success compared with a cochleariform (=spoon-like) viscidium and lack of floral setae condition. We show that the uncinate viscidium is a synapomorphy for Telipogon; setae-bearing flowers are not universally found among all species of Telipogon and evolved once in the Telipogon alliance. Furthermore, Telipogon peruvianus flowers with an uncinate viscidium have achieved higher pollinia export than those with cochleariform viscidium (ancestral condition), whereas flowers with setae have both higher success in pollinia removal, although not significant, and pollinia deposition than those lacking of setae (ancestral condition). We demonstrate that uncinate viscidia and floral setae in Telipogon are a key innovation and exaptation, respectively, that enhance pollination success and they might act as drivers of diversification and pollinator specialization in this genus.






Similar content being viewed by others
Data availability
The sequences analysed during the current study are available in the Dryad digital repository (https://datadryad.org/stash/dataset/doi:10.5061/dryad.2rk39). All the other data generated and analysed during this study are included in this published article (and its supplementary information files).
References
Ackerman, J. D. (1983). Specificity and mutual dependency of the orchid-euglossine bee interaction. Biological Journal of the Linnean Society, 20(3), 301–314. https://doi.org/10.1111/j.1095-8312.1983.tb01878.x.
Ackerman, J. D. (1986). Mechanisms and evolution of food-deceptive pollination systems in orchids. Lindleyana, 1, 108–113.
Ågren, L., Kullenberg, B., & Sensenbaugh, T. (1984). Congruences in pilosity between three species of Ophrys (Orchidaceae) and their hymenopteran pollinators. Nova acta Regiae Societatis Scientiarum Upsaliensis, 3, 15–25.
Arathi, H. S., & Kelly, J. K. (2004). Corolla morphology facilitates both autogamy and bumblebee pollination in Mimulus guttatus. International Journal of Plant Sciences, 165(6), 1039–1045. https://doi.org/10.1086/423876.
Ayasse, M. (2006). Floral scent and pollinator attraction in sexually deceptive orchids. In N. Dudareva & E. Pichersky (Eds.), Biology of floral scent (pp. 219–241). Boca Raton: CRC Press.
Ayasse, M., Schiestl, F. P., Paulus, H. F., Ibarra, F., & Francke, W. (2003). Pollinator attraction in a sexually deceptive orchid by means of unconventional chemicals. Proceedings of the Royal Society of London. Series B: Biological Sciences, 270(1514), 517–522. https://doi.org/10.1098/rspb.2002.2271.
Benitez-Vieyra, S., Medina, A. M., & Cocucci, A. A. (2009). Variable selection patterns on the labellum shape of Geoblasta pennicillata, a sexually deceptive orchid. Journal of Evolutionary Biology, 22(11), 2354–2362. https://doi.org/10.1111/j.1420-9101.2009.01840.x.
Bennett, D. E., & Christenson, E. A. (1998). New species of Peruvian Orchidaceae, V. Lindleyana, 13, 31–56.
Blanco, M. A., & Barboza, G. (2005). Pseudocopulatory pollination in Lepanthes (Orchidaceae: Pleurothallidinae) by fungus gnats. Annals of Botany, 95(5), 763–772. https://doi.org/10.1093/aob/mci090.
Bohman, B., Phillips, R. D., Menz, M. H. M., Berntsson, B. W., Flematti, G. R., Barrow, R. A., Dixon, K. W., & Peakall, R. (2014). Discovery of pyrazines as pollinator sex pheromones and orchid semiochemicals: Implications for the evolution of sexual deception. New Phytologist, 203(3), 939–952. https://doi.org/10.1111/nph.12800.
Bohman, B., Flematti, G. R., Barrow, R. A., Pichersky, E., & Peakall, R. (2016). Pollination by sexual deception—It takes chemistry to work. Current Opinion in Plant Biology, 32, 37–46. https://doi.org/10.1016/j.pbi.2016.06.004.
Bohman, B., Phillips, R. D., Flematti, G. R., Barrow, R. A., & Peakall, R. (2017). The spider orchid Caladenia crebra produces sulfurous pheromone mimics to attract its male wasp pollinator. Angewandte Chemie International Edition, 56(29), 8455–8458. https://doi.org/10.1002/anie.201702864.
Bohman, B., Tan, M. M. Y., Phillips, R. D., Scaffidi, A., Sobolev, A. N., Moggach, S. A., Flematti, G. R., & Peakall, R. (2020). A specific blend of drakolide and hydroxymethylpyrazines: An unusual pollinator sexual attractant used by the endangered orchid Drakaea micrantha. Angewandte Chemie International Edition, 59(3), 1124–1128. https://doi.org/10.1002/anie.201911636.
Borba, E. L., & Semir, J. (2001). Pollinator specificity and convergence in fly-pollinated Pleurothallis (Orchidaceae) species: A multiple population approach. Annals of Botany, 88(1), 75–88. https://doi.org/10.1006/anbo.2001.1434.
Breitkopf, H., Onstein, R. E., Cafasso, D., Schlüter, P. M., & Cozzolino, S. (2015). Multiple shifts to different pollinators fuelled rapid diversification in sexually deceptive Ophrys orchids. New Phytologist, 207(2), 377–389. https://doi.org/10.1111/nph.13219.
Christensen, D. E. (1994). Fly pollination in the Orchidaceae. In J. Arditti (Ed.), Orchid biology: Reviews and perspectives VI (pp. 415–454). New York: John Wiley and Sons, Inc..
Ciotek, L., Giorgis, P., Benitez-Vieyra, S., & Cocucci, A. A. (2006). First confirmed case of pseudocopulation in terrestrial orchids of South America: Pollination of Geoblasta pennicillata (Orchidaceae) by Campsomeris bistrimacula (Hymenoptera, Scoliidae). Flora - Morphology, Distribution, Functional Ecology of Plants, 201(5), 365–369. https://doi.org/10.1016/j.flora.2005.07.012.
Claßen-Bockhoff, R., Speck, T., Tweraser, E., Wester, P., Thimm, S., & Reith, M. (2004). The staminal lever mechanism in Salvia L. (Lamiaceae): A key innovation for adaptive radiation? Organisms Diversity & Evolution, 4(3), 189–205. https://doi.org/10.1016/j.ode.2004.01.004.
Cocucci, A. A., Marino, S., Baranzelli, M., Wiemer, A. P., & Sérsic, A. (2014). The buck in the milkweed: Evidence of male–male interference among pollinaria on pollinators. New Phytologist, 203(1), 280–286. https://doi.org/10.1111/nph.12766.
Coombs, G., Dold, A. P., & Peter, C. I. (2011). Generalized fly-pollination in Ceropegia ampliata (Apocynaceae–Asclepiadoideae): The role of trapping hairs in pollen export and receipt. Plant Systematics and Evolution, 296(1), 137–148. https://doi.org/10.1007/s00606-011-0483-6.
Cozzolino, S., & Widmer, A. (2005). Orchid diversity: An evolutionary consequence of deception? Trends in Ecology & Evolution, 20(9), 487–494. https://doi.org/10.1016/j.tree.2005.06.004.
Cuervo, M., Rakosy, D., Martel, C., Schulz, S., & Ayasse, M. (2017). Sexual deception in the Eucera-pollinated Ophrys leochroma: A chemical intermediate between wasp- and Andrena-pollinated species. Journal of Chemical Ecology, 43(5), 469–479. https://doi.org/10.1007/s10886-017-0848-6.
Damon, A., & Nieto, L. G. (2013). A guide to the morphology of the pollinia and pollinaria of orchids from the biological corridor Tacaná-Boquerón in Southeast Mexico. Selbyana, 31(1), 5–39.
Darwin, C. R. (1862). On the various contrivances by which British and foreign orchids are fertilised by insects, and on the good effects of intercrossing. London: John Murray.
de Jager, M. L., & Peakall, R. (2016). Does morphology matter? An explicit assessment of floral morphology in sexual deception. Functional Ecology, 30(4), 537–546. https://doi.org/10.1111/1365-2435.12517.
Dodson, C. H. (1962). The importance of pollination in the evolution of the orchids of tropical America. American Orchid Society Bulletin, 31, 525-534; 641-649; 731-735.
Dodson, C. H., & Escobar, R. (1987). The Telipogons of Costa Rica (1). Orquideología, 17, 3–69.
Dressler, R. L. (1968). Pollination by euglossine bees. Evolution, 22(1), 202–210. https://doi.org/10.1111/j.1558-5646.1968.tb03463.x.
Dressler, R. L. (1981). The orchids. Natural history and classification. Cambridge: Harvard University Press.
Dressler, R. L. (1993). Phylogeny and classification of the orchid family. Portland: Timber Press.
Erwin, D. H. (2015). Novelty and innovation in the history of life. Current Biology, 25(19), R930–R940. https://doi.org/10.1016/j.cub.2015.08.019.
Faegri, K., & van der Pijl, L. (1979). The principles of pollination ecology (3rd ed.). Oxford: Pergamon.
Fay, M. F. (2018). Orchid conservation: How can we meet the challenges in the twenty-first century? Botanical Studies, 59(1), 16. https://doi.org/10.1186/s40529-018-0232-z.
Fenster, C. B., Armbruster, W. S., Wilson, P., Dudash, M. R., & Thomson, J. D. (2004). Pollination syndromes and floral specialization. Annual Review of Ecology, Evolution, and Systematics, 35(1), 375–403. https://doi.org/10.1146/annurev.ecolsys.34.011802.132347.
Fernández, M., Karremans, A. P., & Pupulin, F. (2011). Orchid pollinaria: Up close and personal. Orchids, 80, 362–363.
Franken, E. P., Pansarin, L. M., & Pansarin, E. R. (2016). Osmophore diversity in the Catasetum cristatum Lindl. alliance (Orchidaceae: Catasetinae). Lankesteriana, 16(3), 317–327. https://doi.org/10.15517/lank.v16i3.26649.
Gaskett, A. C. (2011). Orchid pollination by sexual deception: Pollinator perspectives. Biological Reviews, 86(1), 33–75. https://doi.org/10.1111/j.1469-185X.2010.00134.x.
Gerlach, G. (1999). Sievekingia and Soterosanthus: Related but different. Orquideología, 21(2), 214–227.
Gerlach, G. (2003). La subtribu Stanhopeinae: sus notables mecanismos de polinización, la química de sus aromas florales e implicaciones en sistemática y taxonomía. Lankesteriana, 7, 104–106. https://doi.org/10.15517/lank.v3i2.23029.
Gerlach, G. (2011). The genus Coryanthes: A paradigm in ecology. Lankesteriana, 11, 253–264. https://doi.org/10.15517/lank.v11i3.18280.
Givnish, T. J., Spalink, D., Ames, M., Lyon, S. P., Hunter, S. J., Zuluaga, A., Iles, W. J. D., Clements, M. A., Arroyo, M. T. K., Leebens-Mack, J., Endara, L., Kriebel, R., Neubig, K. M., Whitten, W. M., Williams, N. H., & Cameron, K. M. (2015). Orchid phylogenomics and multiple drivers of their extraordinary diversification. Proceedings of the Royal Society B: Biological Sciences, 282(1814), 20151553. https://doi.org/10.1098/rspb.2015.1553.
Glover, B. J., & Martin, C. (1998). The role of petal cell shape and pigmentation in pollination success in Antirrhinum majus. Heredity, 80(6), 778–784. https://doi.org/10.1046/j.1365-2540.1998.00345.x.
Govaerts, R., Bernet, P., Kratochvil, K., Gerlach, G., Carr, G., Alrich, P., et al. (2019). World checklist of Orchidaceae. http://apps.kew.org/wcsp. Accessed 10 Feb 2019.
Grant, V. (1994). Modes and origins of mechanical and ethological isolation in angiosperms. Proceedings of the National Academy of Sciences, 91(1), 3–10. https://doi.org/10.1073/pnas.91.1.3.
Gravendeel, B., Smithson, A., Slik, F. J. W., & Schuiteman, A. (2004). Epiphytism and pollinator specialization: Drivers for orchid diversity? Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences, 359(1450), 1523–1535. https://doi.org/10.1098/rstb.2004.1529.
Hashimoto, T. (1990). New and noteworthy orchids from Peru. Bulletin of the National Science Museum. Series B, Botany, 16(1), 21–27.
Hobbhahn, N., Johnson, S. D., Bytebier, B., Yeung, E. C., & Harder, L. D. (2013). The evolution of floral nectaries in Disa (Orchidaceae: Disinae): Recapitulation or diversifying innovation? Annals of Botany, 112(7), 1303–1319. https://doi.org/10.1093/aob/mct197.
Hodges, S. A. (1997). Floral nectar spurs and diversification. International Journal of Plant Sciences, 158(S6), S81–S88. https://doi.org/10.1086/297508.
Hodges, S. A., & Arnold, M. L. (1995). Spurring plant diversification: Are floral nectar spurs a key innovation? Proceedings of the Royal Society of London. Series B: Biological Sciences, 262(1365), 343–348. https://doi.org/10.1098/rspb.1995.0215.
Hunter, J. P. (1998). Key innovations and the ecology of macroevolution. Trends in Ecology & Evolution, 13(1), 31–36. https://doi.org/10.1016/S0169-5347(97)01273-1.
Janes, J. K., Steane, D. A., Vaillancourt, R. E., & Duretto, M. F. (2010). A molecular phylogeny of the subtribe Pterostylidinae (Orchidaceae): Resolving the taxonomic confusion. Australian Systematic Botany, 23(4), 248–259. https://doi.org/10.1071/SB10006.
Jersáková, J., Johnson, S. D., & Kindlmann, P. (2006). Mechanisms and evolution of deceptive pollination in orchids. Biological Reviews, 81(2), 219–235. https://doi.org/10.1017/s1464793105006986.
Jersáková, J., Jürgens, A., Šmilauer, P., & Johnson, S. D. (2012). The evolution of floral mimicry: Identifying traits that visually attract pollinators. Functional Ecology, 26(6), 1381–1389. https://doi.org/10.1111/j.1365-2435.2012.02059.x.
Johnson, S. D. (2005). Specialized pollination by spider-hunting wasps in the African orchid Disa sankeyi. Plant Systematics and Evolution, 251(2), 153–160. https://doi.org/10.1007/s00606-004-0234-z.
Johnson, S. D., & Edwards, T. J. (2000). The structure and function of orchid pollinaria. Plant Systematics and Evolution, 222(1), 243–269. https://doi.org/10.1007/BF00984105.
Johnson, S. D., & Schiestl, F. P. (2016). Floral mimicry. Oxford: Oxford University Press.
Johnson, S. D., & Steiner, K. E. (1995). Long-proboscid fly pollination of two orchids in the Cape Drakensberg mountains, South Africa. Plant Systematics and Evolution, 195(3/4), 169–175.
Karremans, A. P., Pupulin, F., Grimaldi, D., Beentjes, K. K., Butôt, R., Fazzi, G. E., Kaspers, K., Kruizinga, J., Roessingh, P., Smets, E. F., & Gravendeel, B. (2015). Pollination of Specklinia by nectar-feeding Drosophila: The first reported case of a deceptive syndrome employing aggregation pheromones in Orchidaceae. Annals of Botany, 116(3), 437–455. https://doi.org/10.1093/aob/mcv086.
Kay, K. M., Reeves, P. A., Olmstead, R. G., & Schemske, D. W. (2005). Rapid speciation and the evolution of hummingbird pollination in neotropical Costus subgenus Costus (Costaceae): Evidence from nrDNA ITS and ETS sequences. American Journal of Botany, 92(11), 1899–1910. https://doi.org/10.3732/ajb.92.11.1899.
Kevan, P. G., & Lane, M. A. (1985). Flower petal microtexture is a tactile cue for bees. Proceedings of the National Academy of Sciences, 82(14), 4750–4752. https://doi.org/10.1073/pnas.82.14.4750.
Kullenberg, B. (1961). Studies in Ophrys pollination. Zoologiska Bidrag Uppsala, 34, 1–340.
Maddison, W. P., & Maddison, D. R. (2004). Mesquite: a modular system for evolutionary analysis. http://mesquiteproject.org.
Martel, C., & Nauray Huari, W. (2013). Notes and emended description of Telipogon peruvianus T. Hashim. (Orchidaceae). Candollea, 68(2), 245–250. https://doi.org/10.15553/c2012v682a8.
Martel, C., & Trujillo, D. (2015). Taxonomical notes on Telipogon falcatus with comments on Hofmeisterella (Orchidaceae: Oncidiinae). Harvard Papers in Botany, 20(2), 93–99. https://doi.org/10.3100/hpib.v20iss2.2015.n1.
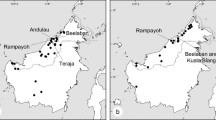
Martel, C., Cairampoma, L., Stauffer, F. W., & Ayasse, M. (2016). Telipogon peruvianus (Orchidaceae) flowers elicit pre-mating behaviour in Eudejeania (Tachinidae) males for pollination. PLoS One, 11(11), e0165896. https://doi.org/10.1371/journal.pone.0165896.
Martel, C., Collantes, B., & Egoavil, L. (2017). Telipogon huancavelicanus sp. nov. (Orchidaceae) from Peru, and an updated description of T. deuterocuscoensis. Nordic Journal of Botany, 35(5), 539–545. https://doi.org/10.1111/njb.01520.
Martel, C., Francke, W., & Ayasse, M. (2019). The chemical and visual bases of the pollination of the Neotropical sexually deceptive orchid Telipogon peruvianus. New Phytologist, 223(4), 1989–2001. https://doi.org/10.1111/nph.15902.
Micheneau, C., Johnson, S. D., & Fay, M. F. (2009). Orchid pollination: From Darwin to the present day. Botanical Journal of the Linnean Society, 161(1), 1–19. https://doi.org/10.1111/j.1095-8339.2009.00995.x.
Milet-Pinheiro, P., & Gerlach, G. (2017). Biology of the Neotropical orchid genus Catasetum: A historical review on floral scent chemistry and pollinators. Perspectives in Plant Ecology, Evolution and Systematics, 27, 23–34. https://doi.org/10.1016/j.ppees.2017.05.004.
Neubig, K. M., Whitten, W. M., Williams, N. H., Blanco, M. A., Endara, L., Burleigh, J. G., et al. (2012a). Data from: Generic recircumscriptions of Oncidiinae (Orchidaceae: Cymbidieae) based on maximum likelihood analysis of combined DNA datasets. Dryad Digital Repository. https://doi.org/10.5061/dryad.2rk39.
Neubig, K. M., Whitten, W. M., Williams, N. H., Blanco, M. A., Endara, L., Burleigh, J. G., et al. (2012b). Generic recircumscriptions of Oncidiinae (Orchidaceae: Cymbidieae) based on maximum likelihood analysis of combined DNA datasets. Botanical Journal of the Linnean Society, 168(2), 117–146. https://doi.org/10.1111/j.1095-8339.2011.01194.x.
Nilsson, L. A. (1992). Orchid pollination biology. Trends in Ecology & Evolution, 7(8), 255–259. https://doi.org/10.1016/0169-5347(92)90170-G.
Oelschlägel, B., Gorb, S., Wanke, S., & Neinhuis, C. (2009). Structure and biomechanics of trapping flower trichomes and their role in the pollination biology of Aristolochia plants (Aristolochiaceae). New Phytologist, 184(4), 988–1002. https://doi.org/10.1111/j.1469-8137.2009.03013.x.
Pansarin, E. R. (2016). Recent advances on evolution of pollination systems and reproductive biology of Vanilloideae (Orchidaceae). Lankesteriana, 16(2), 255–267. https://doi.org/10.15517/lank.v16i2.26010.
Pansarin, E. R., Bittrich, V., & Amaral, M. C. E. (2006). At daybreak - reproductive biology and isolating mechanisms of Cirrhaea dependens (Orchidaceae). Plant Biology, 8(4), 494–502. https://doi.org/10.1055/s-2006-923800.
Parachnowitsch, A. L., & Kessler, A. (2010). Pollinators exert natural selection on flower size and floral display in Penstemon digitalis. New Phytologist, 188(2), 393–402. https://doi.org/10.1111/j.1469-8137.2010.03410.x.
Pauw, A. (2006). Floral syndromes accurately predict pollination by a specialized oil-collecting bee (Rediviva peringueyi, Melittidae) in a guild of South African orchids (Coryciinae). American Journal of Botany, 93(6), 917–926. https://doi.org/10.3732/ajb.93.6.917.
Pérez-Escobar, O. A., Chomicki, G., Condamine, F. L., Karremans, A. P., Bogarín, D., Matzke, N. J., Silvestro, D., & Antonelli, A. (2017). Recent origin and rapid speciation of Neotropical orchids in the world’s richest plant biodiversity hotspot. New Phytologist, 215(2), 891–905. https://doi.org/10.1111/nph.14629.
Peter, C. I., Coombs, G., Huchzermeyer, C. F., Venter, N., Winkler, A. C., Hutton, D., Papier, L. A., Dold, A. P., & Johnson, S. D. (2009). Confirmation of hawkmoth pollination in Habenaria epipactidea: Leg placement of pollinaria and crepuscular scent emission. South African Journal of Botany, 75(4), 744–750. https://doi.org/10.1016/j.sajb.2009.08.007.
Phillips, R. D., Scaccabarozzi, D., Retter, B. A., Hayes, C., Brown, G. R., Dixon, K. W., & Peakall, R. (2013). Caught in the act: Pollination of sexually deceptive trap-flowers by fungus gnats in Pterostylis (Orchidaceae). Annals of Botany, 113(4), 629–641. https://doi.org/10.1093/aob/mct295.
Policha, T., Davis, A., Barnadas, M., Dentinger, B. T. M., Raguso, R. A., & Roy, B. A. (2016). Disentangling visual and olfactory signals in mushroom-mimicking Dracula orchids using realistic three-dimensional printed flowers. New Phytologist, 210(3), 1058–1071. https://doi.org/10.1111/nph.13855.
Pupulin, F., & Karremans, A. (2008). The orchid pollinaria collection at Lankester Botanical Garden, University of Costa Rica. Selbyana, 29(1), 69–86.
Rakosy, D., Cuervo, M., Paulus, H. F., & Ayasse, M. (2017). Looks matter: Changes in flower form affect pollination effectiveness in a sexually deceptive orchid. Journal of Evolutionary Biology, 30(11), 1978–1993. https://doi.org/10.1111/jeb.13153.
Ren, Z.-X., Li, D.-Z., Bernhardt, P., & Wang, H. (2011). Flowers of Cypripedium fargesii (Orchidaceae) fool flat-footed flies (Platypezidae) by faking fungus-infected foliage. Proceedings of the National Academy of Sciences, 108(18), 7478–7480. https://doi.org/10.1073/pnas.1103384108.
Schiestl, F. P., & Cozzolino, S. (2008). Evolution of sexual mimicry in the orchid subtribe Orchidinae: The role of preadaptations in the attraction of male bees as pollinators. BMC Evolutionary Biology, 8(1), 27. https://doi.org/10.1186/1471-2148-8-27.
Schiestl, F. P., & Johnson, S. D. (2013). Pollinator-mediated evolution of floral signals. Trends in Ecology & Evolution, 28(5), 307–315. https://doi.org/10.1016/j.tree.2013.01.019.
Schiestl, F. P., & Schlüter, P. M. (2009). Floral isolation, specialized pollination, and pollinator behavior in orchids. Annual Review of Entomology, 54(1), 425–446. https://doi.org/10.1146/annurev.ento.54.110807.090603.
Schiestl, F. P., Ayasse, M., Paulus, H. F., Löfstedt, C., Hansson, B. S., Ibarra, F., & Francke, W. (1999). Orchid pollination by sexual swindle. Nature, 399(6735), 421–421. https://doi.org/10.1038/20829.
Schiestl, F. P., Peakall, R., Mant, J. G., Ibarra, F., Schulz, C., Franke, S., & Francke, W. (2003). The chemistry of sexual deception in an orchid-wasp pollination system. Science, 302(5644), 437–438. https://doi.org/10.1126/science.1087835.
Scopece, G., Cozzolino, S., Johnson, S. D., & Schiestl, F. P. (2010). Pollination efficiency and the evolution of specialized deceptive pollination systems. The American Naturalist, 175(1), 98–105. https://doi.org/10.1086/648555.
Shrestha, M., Dyer, A. G., Dorin, A., Ren, Z.-X., & Burd, M. (2020). Rewardlessness in orchids: How frequent and how rewardless? Plant Biology, 22, 555–561. https://doi.org/10.1111/plb.13113.
Simpson, G. G. (1953). The major features of evolution. New York: Columbia University Press.
Singer, R. B. (2002). The pollination mechanism in Trigonidium obtusum Lindl (Orchidaceae: Maxillariinae): Sexual mimicry and trap-flowers. Annals of Botany, 89(2), 157–163. https://doi.org/10.1093/aob/mcf021.
Singer, R. B., & Cocucci, A. A. (1999). Pollination mechanism in southern Brazilian orchids which are exclusively or mainly pollinated by halictid bees. Plant Systematics and Evolution, 217(1), 101–117. https://doi.org/10.1007/BF00984924.
Singer, R. B., Flach, A., Koehler, S., Marsaioli, A. J., & Amaral, M. d. C. E. (2004). Sexual mimicry in Mormolyca ringens (Lindl.) Schltr. (Orchidaceae: Maxillariinae). Annals of Botany, 93(6), 755–762. https://doi.org/10.1093/aob/mch091.
Singer, R. B., Gravendeel, B., Cross, H., & Ramirez, S. R. (2008). The use of orchid pollinia or pollinaria for taxonomic identification. Selbyana, 29(1), 6–19.
Smith, S. D., Ané, C., & Baum, D. A. (2008). The role of pollinator shifts in the floral diversification of Iochroma (Solanaceae). Evolution, 62(4), 793–806. https://doi.org/10.1111/j.1558-5646.2008.00327.x.
Tremblay, R. L. (1992). Trends in the pollination ecology of the Orchidaceae: Evolution and systematics. Canadian Journal of Botany, 70(3), 642–650. https://doi.org/10.1139/b92-083.
Tuomi, J., Lämsä, J., Wannas, L., Abeli, T., & Jäkäläniemi, A. (2015). Pollinator behaviour on a food-deceptive orchid Calypso bulbosa and coflowering species. The Scientific World Journal, 2015, 482161–482169. https://doi.org/10.1155/2015/482161.
Valenta, K., Nevo, O., Martel, C., & Chapman, C. A. (2017). Plant attractants: Integrating insights from pollination and seed dispersal ecology. Evolutionary Ecology, 31(2), 249–267. https://doi.org/10.1007/s10682-016-9870-3.
van der Niet, T., Hansen, D. M., & Johnson, S. D. (2011). Carrion mimicry in a South African orchid: Flowers attract a narrow subset of the fly assemblage on animal carcasses. Annals of Botany, 107(6), 981–992. https://doi.org/10.1093/aob/mcr048.
van der Pijl, L., & Dodson, C. H. (1966). Orchid flowers: Their pollination and evolution. Coral Gables: University of Miami Press.
Vieira, B. C., Pansarin, L. M., Martucci, M. E. P., Gobbo-Neto, L., & Pansarin, E. R. (2017). Pollinarium size as a hybridisation barrier between sympatric inter-compatible orchids. Australian Journal of Botany, 65(7), 497–506. https://doi.org/10.1071/BT17081.
Werker, E. (2000). Trichome diversity and development. In D. L. Hallahan & J. C. Gray (Eds.), Plant trichomes (Vol. 31, pp. 1–35). San Diego: Academic Press.
Whitney, H. M., Chittka, L., Bruce, T. J. A., & Glover, B. J. (2009). Conical epidermal cells allow bees to grip flowers and increase foraging efficiency. Current Biology, 19(11), 948–953. https://doi.org/10.1016/j.cub.2009.04.051.
Whitney, H. M., Bennett, K. M. V., Dorling, M., Sandbach, L., Prince, D., Chittka, L., & Glover, B. J. (2011). Why do so many petals have conical epidermal cells? Annals of Botany, 108(4), 609–616. https://doi.org/10.1093/aob/mcr065.
Xu, H., Bohman, B., Wong, D. C. J., Rodriguez-Delgado, C., Scaffidi, A., Flematti, G. R., Phillips, R. D., Pichersky, E., & Peakall, R. (2017). Complex sexual deception in an orchid is achieved by co-opting two independent biosynthetic pathways for pollinator attraction. Current Biology, 27(13), 1867–1877. https://doi.org/10.1016/j.cub.2017.05.065.
Zhang, G.-Q., Li, M.-H., Su, Y.-Y., Chen, L.-J., Lan, S.-R., & Liu, Z.-J. (2015). A new myco-heterotrophic genus, Yunorchis, and the molecular phylogenetic relationships of the tribe Calypsoeae (Epidendroideae, Orchidaceae) inferred from plastid and nuclear DNA sequences. PLoS One, 10(4), e0123382. https://doi.org/10.1371/journal.pone.0123382.
Acknowledgements
Abigail Dextre kindly assisted us during fieldwork. Juana and José Gutierrez hosted us in Marcapata town during fieldwork. Benjamin Collantes (Inka-Terra Association) took the pictures of Fig. 4. We thank two anonymous that reviewers provided helpful comments on an earlier version of the manuscript. C.M. thanks to Pedro Romero and Ernesto Razuri for comments on parts of this manuscript. The phylogenetic portion of this study would not have been possible without the dedication, knowledge and mentorship of the late W. Mark Whitten, to whom we dedicated this article.
Funding
The study was supported by the German Academic Exchange Service (DAAD) through a PhD. scholarship to C.M. and the National Geographic through a research grant to M.A and C.M. DNA data were produced with the support of NSF grants DEB-9815821, DEB-9509071, DEB-0234064, and IOB-0543659, by grants from the American Orchid Society Fund for Education and Research, the Florida Museum of Natural History, and the Royal Botanic Gardens, Kew.
Author information
Authors and Affiliations
Contributions
C.M. and M.A. planned the research; C.M. designed the research, collected bibliographic data, conducted lab work and fieldwork, and performed experiments; K.M.N. and N.H.W. collected genetic data; C.M. analysed data and wrote the original draft; C.M., K.M.N., N.H.W. and M.A. reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1272 kb)
Rights and permissions
About this article
Cite this article
Martel, C., Neubig, K.M., Williams, N.H. et al. The uncinate viscidium and floral setae, an evolutionary innovation and exaptation to increase pollination success in the Telipogon alliance (Orchidaceae: Oncidiinae). Org Divers Evol 20, 537–550 (2020). https://doi.org/10.1007/s13127-020-00457-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-020-00457-w