Abstract
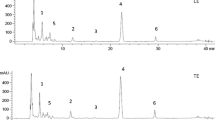
There is an increasing interest to bio-components obtained from fruit & vegetable wastes. Anthocyanin is one of the components regained from fruits especially from red ones. Sour cherry is considered as industrial fruit since most of the grown fruit is processed into juice and hence considerable amount of pomace is removed from process. The influences of process parameters on the extraction of phenolic compounds from sour cherry pomace were investigated. Fifty-one percent ethanol concentration, 75 °C temperature and 12 mL/g solvent to solid ratio were selected as optimum process parameters. Time effect on the process efficiency was monitored at three different temperatures (25, 50 and 75 °C) and total phenolic and total anthocyanin contents were found to reach equilibrium concentrations between 80 and 100 min time intervals. Ethanol concentrations affected differently extraction yield of anthocyanins and non-anthocyanin phenolics. Various individual phenolic compounds present in sour cherry pomace were identified and quantified by HPLC equipped with UV/Vis PDA. Of the phenolic compounds, cyanidin-3-glucosylrutinoside, neochlorogenic acid and catechin were the most abundant ones found in the pomace. 14.23 ± 0.38 mg/g total phenolic, 0.41 ± 0.02 mg/g total anthocyanin, 0.19 ± 0.02 mg/g cyanidin-3-glucosylrutinoside, 0.22 ± 0.01 mg/g neochlorogenic acid and 0.22 ± 0.02 mg/g catechin contents (dry weight) were determined in the pomace at optimum extraction conditions.



Similar content being viewed by others
References
Abou-Arab AA, Abu-Salem FM, Abou-Arab EA (2011) Physico-chemical properties of natural pigments (anthocyanin) extracted from roselle calyces (Hibiscus subdariffa). J Am Sci 7(7):445–456
Adil İH, Yener ME, Bayındırlı A (2008) Extraction of total phenolics of sour cherry pomace by high pressure solvent and subcritical fluid and determination of the antioxidant activities of the extracts. Separ Sci Technol 43:1091–1110
Aguedo M, Kohnen S, Rabetafika N, Bossche SV, Sterckx J, Blecker C, Beauve C, Paquot M (2012) Composition of by-products from cooked fruit processing and potential use in food products. J Food Comp Anal 27:61–69
Amendola D, Faveri DD, Spigno G (2010) Grape marc phenolics: extraction kinetics, quality and stability of extracts. J Food Eng 97:384–392
Ayala-Zavala J, Vega-Vega V, Rosas-Dominguez C, Palafox-Carlos H, Villa-Rodriguez J, Siddiqui MW, Davila-Avina J, Gonzalez-Aguilar G (2011) Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res Int 44:1866–1874
Ballard TS, Mallikarjunan P, Zhou K, O’Keefe SF (2009) Optimizing the extraction of phenolic antioxidants from peanut skins using response surface methodology. J Agr Food Chem 57:3064–3072
Baysal T, Ersus S, Starmans DAJ (2000) Supercritical CO2 extraction of β-Carotene and lycopene from tomato paste waste. J Agr Food Chem 48:5507–5511
Bonilla F, Mayen M, Merida J, Medina M (1999) Extraction of phenolic compounds from red grape marc for use as food lipid antioxidants. Food Chem 66:209–215
Boonchu T, Utama-ang N (2013) Optimization of extraction and microencapsulation of bioactive compounds from red grape (Vitis vinifera L.) pomace. J Food Sci Technol. doi:10.1007/s13197-013-1079-7
Bridle P, Timberlake C (1997) Anthocyanins as natural food colours-selected aspects. Food Chem 58:103–109
Cacace J, Mazza G (2003) Optimization of extraction of anthocyanins from black currants with aqueous ethanol. J Food Sci 68:240–248
Çam M, İçyer N (2013) Phenolics of pomegranate peels: extraction optimization by central composite design and alpha glucosidase inhibition potentials. J Food Sci Technol. doi:10.1007/s13197-013-1148-y
Dai J, Mumper R (2010) Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15:7313–7352
Fan G, Han Y, Gu Z, Chen D (2008) Optimizing conditions for anthocyanins extraction from purple sweet potato using response surface methodology (RSM). LWT-Food Sci Technol 41:155–160
FAOSTAT (2010) Food and Agricultural commodities production, countries by commodity (http://faostat.fao.org/site/339/default.aspx) (June 2, 2011)
Ferreira-Dias S, Valente D, Abreu J (2003) Comparison between ethanol and hexane for oil extraction from Quercus suber L. fruits. Grasas Aceites 54:378–383
Garofulic IE, Dragovic-Uzelac V, Jambrak AR, Jukic M (2013) The effect of microwave assisted extraction on the isolation of anthocyanins and phenolic acids from sour cherry Marasca (Prunus cerasus var. Marasca). J Food Eng 117:437–442
Gould K, Davies K, Winefield C (2009) Anthocyanins; biosynthesis, functions and applications. Springer, New York, pp 283–305
MEYED (Fruit Juice Industry Corporation) (2011) Turkey fruit juice like products industry report (http://www.meyed.org.tr/userfiles/file/sektor_istatistikleri/meyve_suyu_sektoru_raporu___2011.pdf) (Aug 16, 2012)
Kang S-Y, Seeram NP, Nair MG, Bourquin LD (2003) Tart cherry anthocyanins inhibit tumor development in ApcMin mice and reduce proliferation of human colon cancer cells. Cancer Lett 194:13–19
Kim D-O, Heo HJ, Kim YJ, Yang HS, Lee CY (2005) Sweet and sour cherry phenolics and their protective effects on neuronal cells. J Agr Food Chem 53:9921–9927
Kitanovic S, Milenovic D, Veljkovic VB (2008) Empirical kinetic models for the resinoid extraction from aerial parts of St. John’s wort (Hypericum perforatum L.). Biochem Eng J 41:1–11
Kolodziejczyk K, Sojka M, Abadias M, Vinas I, Guyot S, Baron A (2013) Polyphenol composition, antioxidant capacity, and antimicrobial activity of the extracts obtained from industrial sour cherry pomace. Ind Crop Prod 51:279–288
Kumcuoglu S, Yilmaz T, Tavman S (2013) Ultrasound assisted extraction of lycopene from tomato processing wastes. J Food Sci Technol. doi:10.1007/s13197-013-0926-x
Luthria DL (2008) Influence of experimental conditions on the extraction of phenolic compounds from parsley (Petroselinum crispum) flakes using a pressurized liquid extractor. Food Chem 107:745–752
Mitic MN, Obradovic MV, Grahovac ZB, Pavlovic AN (2010) Antioxidant capacities and phenolic levels of different varieties of Serbian white wines. Molecules 15:2016–2027
Nakamiya K, Furuichi T, Ishii K (2003) Evaluation of the optimal washing conditions for dioxin-contaminated soils from the circumference of an incinerator. J Mater Cycles Waste 5:63–68
O’Shea N, Arendt EK, Gallagher E (2012) Dietary fiber and phytochemical characteristics of fruit and vegetable by-products and their recent applications as novel ingredients in food products. Innov Food Sci Emerg 16:1–10
Parmar I, Sharma S, Rupasinghe H (2014) Optimization of β-cyclodextrin-based flavonol extraction from apple pomace using response surface methodology. J Food Sci Technol. doi:10.1007/s13197-014-1282-1
Pinelo M, Rubilar M, Jerez M, Sineiro J, Nunez MJ (2005) Effect of solvent, temperature, and solvent-to-solid ratio on the total phenolic content and antiradical activity of extracts from different components of grape pomace. J Agr Food Chem 53:2111–2117
Pompeu D, Silva E, Rogez H (2009) Optimisation of the solvent extraction of phenolic antioxidants from fruits of Euterpe oleracea using Response Surface Methodology. Bioresour Technol 100:6076–6082
Roldan E, Sanchez-Moreno C, de Ancos B, Cano MP (2008) Characterisation of onion (Allium cepa L.) by-products as food ingredients with antioxidant and antibrowning properties. Food Chem 108:907–916
Silva E, Rogez H, Larondelle Y (2007) Optimization of extraction of phenolics from Inga edulis leaves using response surface methodology. Sep Purif Technol 55:381–387
Simsek M, Sumnu G, Sahin S (2012) Microwave assisted extraction of phenolic compounds from sour cherry pomace. Separ Sci Technol 47:1248–1254
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Sun Y, Xu W, Zhang W, Hu Q, Zeng X (2011) Optimizing the extraction of phenolic antioxidants from kudingcha made frrom Ilex kudingcha C.J. Tseng by using response surface methodology. Sep Purif Technol 78:311–320
Tantoush Z, Stanic D, Stojadinovic M, Ognjenovic J, Mihajlovic L, Atanaskovic-Markovic M, Velickovic TC (2011) Digestibility and allergenicity of β-lactoglobulin following laccase-mediated cross-linking in the presence of sour cherry phenolics. Food Chem 125:84–91
Tsao R, Deng Z (2004) Separation procedures for naturally occurring antioxidant phytochemicals. J Chromatogr B 812:85–99
Türkyılmaz M, Özkan M (2012) Kinetics of anthocyanin degradation and polymeric colour formation in black carrot juice concentrates during storage. Int J Food Sci Tech 47:2273–2281
Wang H, Nair MG, Strasburg GM, Chang Y-C, Booren AM, Gray JI, DeWitt DL (1999) Antioxidant and antiinflammatory activities of anthocyanins and their aglycon, cyanidin, from tart cherries. J Nat Prod 62:294–296
Wang C, Shi L, Fan L, Ding Y, Zhao S, Liu Y, Ma C (2013) Optimization of extraction and enrichment of phenolics from pomegranate (Punica granatum L.) leaves. Ind Crop Prod 42:587–594
Wijngaard H, Hossain MB, Rai DK, Brunton N (2012) Techniques to extract bioactive compounds from food by-products of plant origin. Food Res Int 46:505–513
Wrolstad RE (1976) Color and pigment analyses in fruit products. Station Bulletin 624, Agricultural Experimental Station. Oregon State University, Corvallis
Yağcı S, Göğüş F (2008) Response surface methodology for evaluation of physical and functional properties of extruded snack foods developed from food-by-products. J Food Eng 86:122–132
Yang B, Liu X, Gao Y (2009) Extraction optimization of bioactive compounds (crocin, geniposide and total phenolic compounds) from Gardenia (Gardenia jasminoides Ellis) fruits with response surface methodology. Innov Food Sci Emerg 10:610–615
Yang H, Ye X, Sun Y, Wu D, Chen Y, Liu D (2012) Optimization of extraction of prodelphinidins from bayberry (Myrica rubra Sieb. et Zucc.) leaves. J Food Sci Technol. doi:10.1007/s13197-012-0719-7
Yim HS, Chye FY, Koo SM, Matanjun P, How SE, Ho CW (2012) Optimization of extraction time and temperature for antioxidant activity of edible wild mushroom, Pleurotus porrigens. Food Bioprod Process 90:235–242
Acknowledgments
This study was supported with a fund from Harran University Scientific Research Committee (Project no: 1180). The authors would like to give special thanks to Prof. Dr. Mehmet ÖZKAN (Ankara University, Food Engineering Dept), Dr. Betül ERKAN KOÇ and spec. Çiğdem GÖVER (Harran University, HUMEL) for their kindest technical supports.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yılmaz, F.M., Karaaslan, M. & Vardin, H. Optimization of extraction parameters on the isolation of phenolic compounds from sour cherry (Prunus cerasus L.) pomace. J Food Sci Technol 52, 2851–2859 (2015). https://doi.org/10.1007/s13197-014-1345-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-014-1345-3