Abstract
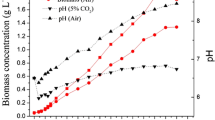
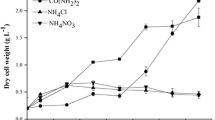
Activity of acetyl-CoA carboxylase (ACCase) and lipid accumulation were assayed in the microalga Chlorella vulgaris co-immobilised in alginate beads with Azospirillum brasilense, under autotrophic and heterotrophic growth conditions, with and without ammonium starvation. ACCase is a key enzyme in de novo fatty acid biosynthesis. Under the two growth conditions, co-immobilisation always enhanced the activity of ACCase and yielded a higher level of lipids when compared with immobilisation of the alga alone. The highest lipid content obtained under autotrophic conditions was with ammonium starvation. Cultivation under heterotrophic conditions without limitation of nitrogen, with or without the presence of bacteria, yielded a higher growth rate and accumulated more lipids than under autotrophic conditions. No correlation was found between total lipids and ACCase activity. Unusually, ammonium starvation significantly reduced lipid accumulation under heterotrophic conditions. Consequently, co-immobilisation, sufficient ammonium and heterotrophic growth conditions were the most significant parameters for lipid accumulation and ACCase activity in C. vulgaris where the two latter parameters are not directly linked.



Similar content being viewed by others
References
Alban C, Baldet P, Douce R (1994) Localization and characterization of two structurally different forms of acetyl-CoA carboxylase in young pea leaves, of which one is sensitive to aryloxyphenoxypropionate herbicides. Biochem J 300:557–565
Bashan Y (1986) Alginate beads as synthetic inoculant carriers for the slow release of bacteria that affect plant growth. Appl Environ Microb 51:1089–1098
Bashan Y, de-Bashan LE (2010) How the plant growth-promoting bacterium Azospirillum promotes plant growth-a critical assessment. Adv Agron 108:77–136
Bashan Y, Holguin G, Lifshitz R (1993) Isolation and characterization of plant growth-promoting rhizobacteria. In: Glick B, Thompson JE (eds) Methods in plant molecular biology and biotechnology. CRC, Boca Raton, pp 331–345
Bashan Y, Hernandez JP, Leyva LA, Bacilio M (2002) Alginate microbeads as inoculant carrier for plant growth-promoting bacteria. Biol Fert Soils 35:359–368
Bashan Y, Holguin G, de-Bashan LE (2004) Azospirillum-plant relationships: physiological, molecular, agricultural, and environmental advances (1997–2003). Can J Microbiol 50:521–577
Bligh GE, Dyer JW (1959) A rapid method f total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bumbak F, Cook S, Zachleder V, Hauser S, Kovar K (2011) Best practices in heterotrophic high-cell-density microalgal processes: achievements, potential and possible limitations. Appl Microbiol Biotechnol 91:31–46
Choix FJ, de-Bashan LE, Bashan Y (2012a) Enhanced accumulation of starch and total carbohydrates in alginate-immobilized Chlorella spp. induced by Azospirillum brasilense. I. Autotrophic conditions. Enzym Microb Technol 51:294–299
Choix FJ, de-Bashan LE, Bashan Y (2012b) Enhanced accumulation of starch and total carbohydrates in alginate-immobilized Chlorella spp. induced by Azospirillum brasilense. II. Heterotrophic conditions. Enzyme Microb Technol 51:300–309
Covarrubias SA, de-Bashan LE, Moreno M, Bashan Y (2012) Alginate beads provide a beneficial physical barrier against native microorganisms in wastewater treated with immobilized bacteria and microalgae. Appl Microbiol Biotechnol 93:2669–2680
Cruz I, Bashan Y, Hernàndez-Carmona G, de-Bashan LE (2013) Biological deterioration of alginate beads containing immobilized microalgae and bacteria during tertiary wastewater treatment. Appl Microbiol Biotechnol 97:9847–9858
de-Bashan LE, Bashan Y (2008) Joint immobilization of plant growth-promoting bacteria and green microalgae in alginate beads as an experimental model for studying plant-bacterium interactions. Appl Environ Microb 74:6797–6802
de-Bashan LE, Bashan Y (2010) Immobilized microalgae for removing pollutants: review of practical aspects. Bioresource Technol 101:1611–1627
de-Bashan LE, Bashan Y, Moreno M, Lebsky VK, Bustillos JJ (2002a) Increased pigment and lipid content, lipid variety, and cell and population size of the microalgae Chlorella spp. when coimmobilized in alginate beads with the microalgae-growth-promoting bacteria Azospirillum brasilense. Can J Microbiol 48:514–521
de-Bashan LE, Moreno M, Hernandez JP, Bashan Y (2002b) Removal of ammonium and phosphorus ions from synthetic wastewater by the microalgae Chlorella vulgaris coimmobilized in alginate beads with the microalage growth-promoting bacterium Azospirillum brasilense. Water Res 36:2941–2948
de-Bashan LE, Hernandez JP, Morey T, Bashan Y (2004) Microalgae growth-promoting bacteria as helpers for microalgae: a novel approach for removing ammonium and phosphorus from municipal wastewater. Water Res 38:466–474
de-Bashan LE, Antoun H, Bashan Y (2005) Cultivation factors and population size control uptake of nitrogen by the microalgae Chlorella vulgaris when interacting with the microalgae growth-promoting bacterium Azospirillum brasilense. FEMS Microbiol Ecol 54:197–203
de-Bashan LE, Magallon P, Antoun H, Bashan Y (2008a) Role of glutamate dehydrogenase and glutamine synthetase in Chlorella vulgaris during assimilation of ammonium when jointly immobilized with the microalgae-growth-promoting bacterium Azospirillum brasilense. J Phycol 44:1188–1196
de-Bashan LE, Trejo A, Huss VAR, Hernandez JP, Bashan Y (2008b) Chlorella sorokiniana UTEX 2805, a heat and intense, sunlight-tolerant microalgae with potential for removing ammonium from wastewater. Bioresource Technol 99:4980–4989
de-Bashan LE, Schmid M, Rothballer M, Hartmann A, Bashan Y (2011) Cell-cell interaction in the eukaryote-prokaryote model using the microalgae Chlorella vulgaris and the bacterium Azospirillum brasilense immobilized in polymer beads. J Phycol 47:1350–1359
Dunahay TG, Jarvis EE, Dais SS, Roessler PG (1996) Manipulation of microalgal lipid production using genetic engineering. Appl Biochem Biotechnol 57(58):223–231
Egli MA, Gengenbach BG, Gronwald JW, Somers DA, Wyse DL (1993) Characterization of maize acetyl-Coenzyme A carboxylase. Plant Physiol 101:499–506
Francki M, Whitaker P, Smith P, Atkins C (2002) Differential expression of a novel gene during seed triacylglycerol accumulation in lupin species (Lupinus angustifolius L. and L. mutabilis L.). Funct Integr Genom 2:292–300
Gonzalez LE, Bashan Y (2000) Growth promotion of the microalga Chlorella vulgaris when coimmobilized and cocultured in alginate beads with the plant-growth-promoting bacterium Azospirillum brasilense. Appl Environ Microb 66:1527–31
Gonzalez LE, Cañizares RO, Baena S (1997) Efficiency of ammonia and phosphorus removal from a Colombian agroindustrial wastewater by the microalgae Chlorella vulgaris and Scenedesmus dimorphus. Bioresource Technol 60:259–262
Gouveia L, Oliveira AC (2009) Microalgae as raw material for biofuels production. J Ind Microbiol Biot 36:269–274
Hayashi O, Satoh K (2006) Determination of acetyl-CoA and Malonyl-CoA in germinating rice seeds using the LC-MS/MS Technique. Biosci Biotech Biochem 70:2676–2681
Herbert D, Price L, Alban C, Dehaye L, Job D, Cole D, Pallet K, Hardwood J (1996) Kinetic studies on two isoforms of acetyl-CoA carboxylase from maize leaves. Biochem J 318:997–1006
Hernandez JP, de-Bashan LE, Bashan Y (2006) Starvation enhances phosphorus removal from wastewater by the microalga Chlorella spp. co-immobilized with Azospirillum brasilense. Enzyme Microb Tech 38:190–198
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuels production: perspectives and advances. Plant J 54:621–639
James ES, Cronan JE (2004) Expression of two Escherichia coli acetyl-CoA carboxylase subunits is autoregulated. J Biol Chem 279:2520–2527
Khozin-Goldberg I, Cohen Z (2011) Unraveling algal lipid metabolism: recent advances in gene identification. Biochimie 93:91–100
Klaus D, Ohlrogge J, Ekkerhard Neuhaus H, Dörmann P (2004) Increased fatty acid production in potato by engineering of Acetyl-CoA carboxylase. Planta 219:389–396
Lebeau T, Robert JM (2006) Biotechnology of immobilized micro algae: a culture technique for the future? In: Rao S (ed) Algal cultures, analogues of blooms and applications. Science Publishers, Enfield, pp 801–837
Levert KL, Waldrop GL, Stephens JM (2002) A biotin analogue inhibits acetyl CoA carboxylase activity and adipogenesis. J Biol Chem 277:16347–16350
Liu W, Harrison DK, Chalupska D, Gornicki P, O’Donnell C, Adkins S, Haselkorn R, Williams R (2007) Single-site mutations in the carboxyltransferase domain of plastid acetyl-CoA carboxylase confer resistance to grass-specific herbicides. Proc Natl Acad Sci USA 104:3627–3632
Liu J, Huang J, Sun Z, Zhong Y, Jiang Y, Chen F (2011) Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: assessment of algal oils for biodiesel production. Bioresource Technol 102:106–110
Livne A, Sukenik A (1992) Lipid synthesis and abundance of acetyl-CoA carboxylase in Isochrysis galbana (Prymnesiophyceae) following nitrogen starvation. Plant Cell Physiol 33:1175–1181
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sust Energ Rev 14:217–232
O’Grady J, Morgan A (2011) Heterotrophic growth and lipid production of Chlorella protothecoides on glycerol. Bioprocess Eng 34:121–125
Oh-Hama T, Miyachi S (1992) Chlorella. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algae biotechnology. Cambridge University Press, Cambridge, pp 3–26
Ohlrogge J, Browse J (1995) Lipid biosynthesis. Plant Cell 7:957–970
Pande SV, Parvin RK, Venkitasubramanian TA (1963) Microdetermination of lipids and serum total fatty acids. Anal Biochem 6:415–423
Perez-Garcia O, de-Bashan LE, Hernandez JP, Bashan Y (2010) Efficiency of growth and nutrient uptake from wastewater by heterotrophic, autotrophic, and mixotrophic cultivation of Chlorella vulgaris immobilized with Azospirillum brasilense. J Phycol 46:800–812
Perez-Garcia O, Bashan Y, Puente ME (2011) Organic carbon supplementation of sterilized municipal wastewater is essential for heterotrophic growth and removing ammonium by the microalga Chlorella vulgaris. J Phycol 47:190–199
Porra RJ, Thomson WA, Kriedemann PA (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentrations of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394
Prasad K, Kadokawa JI (2009) Alginate-based blends and nano/microbeads. Microbiol Monogr 13:175–210
Přibyl P, Cepák V, Zachleder V (2012) Production of lipids in 10 strains of Chlorella and Parachlorella, and enhanced lipid productivity in Chlorella vulgaris. Appl Microbiol Biotechnol 94:549–561
Radakovits R, Jinkerson RE, Darzins A, Pasewitz C (2010) Genetic engineering of algae for enhanced biofuel production. Eukaryot Cell 9:486–501
Rawat I, Ranjith Kumar R, Mutanda T, Bux F (2013) Biodiesel from microalgae: a critical evaluation from laboratory to large scale production. Appl Energ 103:444–467
Roessler PG, Ohlrogge JB (1993) Cloning and characterization of the gene that encodes acetyl-coenzyme A carboxylase in the alga Cyclotella cryptica. J Biol Chem 268:19254–19259
Sartory D, Grobbelaar J (1984) Extraction of chlorophyll a from freshwater phytoplankton for spectrophotometric analysis. Hydrobiologia 114:177–187
Sasaki Y, Nagano Y (2004) Plant acetyl-CoA carboxylase: structure, biosynthesis, regulation and gene manipulation for plant breeding. Biosci Biotechnol Biochem 68:1175–1184
Sato N, Moriyama T (2007) Genomic and biochemical analysis of lipid biosynthesis in the unicellular rhodophyte Cyanidioschyzon merolae: lack of plastidic desaturation pathway results in the coupled pathway of galactolipid synthesis. Eukariot Cell 6:1006–1017
Sheehan J, Dunahay T, Benemann J, Roessler PG (1998) US Department of Energy’s Office of Fuels Development. A look back at the US Department of Energy’s aquatic species program – biodiesel from algae, close Out Report TP-580-24190. National Renewable Energy Laboratory, Golden
Sukenik A, Livne A (1991) Variations in lipid and fatty acid content in relation to acetyl CoA carboxylase in the marine Prymnesiophyte Isochrysis galbana. Plant Cell Physiol 32:371–378
Tang H, Chen M, Garcia MED, Abunasser N, Simon Ng KY, Salley SO (2011) Culture of microalgae Chlorella minutissima for biodiesel feedstock production. Biotechnol Bioeng 108:2280–2287
Tong L, Hardwood HJ (2006) Acetyl-coenzyme A carboxylases: versatile targets for drug discovery. J Cell Biochem 99:1476–1488
Wan M, Wang R, Xia J, Rosenberg J, Nie Z, Kobayashi N, Oyler G, Betenbaugh M (2012) Physiological evaluation of a new Chlorella sorokiniana isolate for its biomass production and lipid accumulation in photoautotrophic and heterotrophic cultures. Biotechnol Bioeng 109:1958–1964
Xiong W, Gao CF, Yan D, Wu C, Wu QY (2010) Double CO2 fixation in photosynthesis–fermentation model enhances algal lipid synthesis for biodiesel production. Bioresource Technol 101:2287–2293
Yu Q, Collavo A, Zheng M, Owen M, Sattin M, Powles S (2007) Diversity of acetyl-Coenzyme A carboxylase mutations in resistant Lolium populations: evaluation using clethodim. Plant Physiol 145:547–558
Acknowledgments
At CIBNOR, we thank Manuel Moreno for technical support, Ira Fogel for editorial improvements, Fernando Garcia-Carreño for free use of the HPLC, and Mariana Diaz- Tenorio of Instituto Tecnologico de Sonora, Cd. Obregon, Mexico, for help in enzymatic analysis. This study was supported by the Secretaria de Medio Ambiente y Recursos Naturales, (SEMARNAT contract 23510) and Consejo Nacional de Ciencia y Tecnologia of Mexico (CONACYT-Basic Science-2009, contracts 130656 and 164548). Time for writing was provided by The Bashan Foundation, USA. L.A.L. was mainly supported by a graduate fellowship (CONACYT #48487) and additional periodic grants from The Bashan Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study is dedicated to the memory of the Italian microbiologist Prof. Franco Favilli (1933–2012) of the University of Florence, Italy, one of the pioneers of Azospirillum studies
Rights and permissions
About this article
Cite this article
Leyva, L.A., Bashan, Y. & de-Bashan, L.E. Activity of acetyl-CoA carboxylase is not directly linked to accumulation of lipids when Chlorella vulgaris is co-immobilised with Azospirillum brasilense in alginate under autotrophic and heterotrophic conditions. Ann Microbiol 65, 339–349 (2015). https://doi.org/10.1007/s13213-014-0866-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-014-0866-3