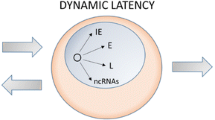
Abstract
The fundamental question posed here is why in dorsal root ganglia herpes simplex viruses (HSV) can establish a silent infection in which only latency associate transcripts (LAT) and miRNAs are expressed and the neuronal cell survives whereas in non-neuronal cells HSV replicates and destroys the infected cells. Current evidence indicates that in productive infection there are two checkpoints. The first is at activation of α genes and requires a viral protein (VP16) that recruits HCF-1, Oct1, LSD1, and the CLOCK histone acetyl transferase to demethylate histones and initiate transcription. The second checkpoint involves activation of β and γ genes. An α protein, ICP0, activates transcription by displacing HDAC1 or 2 from the HDAC/CoREST/LSD1/REST repressor complex at its DNA binding sites. Current data suggest that in dorsal root ganglia VP16 and HCF-1 are not translocated to neuronal nucleus and that the HDAC/CoREST/LSD1/REST complex is not suppressed—a first step in silencing of the viral genome and establishment of heterochromatin. The viral genome remains in a state of equilibrium with respect to viral gene expression. The function of both LAT and the micro RNAs is to silence low level expression of viral genes that could reactivate the latent genomes.


Similar content being viewed by others
References
Ballas N, Mandel G (2005) The many faces of REST oversee epigenetic programming of neuronal genes. Curr Opin Neurobiol 15:500–506
Boissiere S, Hughes T, O’Hare P (1999) HCF-dependent nuclear import of VP16. EMBO J 18:480–489
Boutell C, Sadis S, Everett RD (2002) Herpes simplex virus type 1 immediate-early protein ICP0 and its isolated RING finger domain act as ubiquitin E3 ligases in vitro. J Virol 76:841–850
Chee AV, Lopez P, Pandolf PP, Roizman B (2003) Promyelocytic leukemia protein mediates interferon-based anti-herpes simplex virus 1 effects. J Virol 77:7101–7105
Chen S, Lee L, Garber D, Shaffer P, Knipe D, Coen D (2002) Neither LAT nor open reading frame P mutations increase expression of spliced or intron-containing ICP0 transcripts in mouse ganglia latently infected with herpes simplex virus. J Virol 76:4764–4772
Cui C, Griffiths A, Li G, Silva LM, Kramer MF, Gaasterland T, Wang XJ, Coen DM (2006) Prediction and identification of herpes simplex virus 1-encoded microRNAs. J Virol 80:5499–5508
Du T, Zhou G, Khan S, Gu H, Roizman B (2010) Disruption of HDAC/CoREST/REST repressor by dnREST reduces genome silencing and increases virulence of herpes simplex virus. Proc Natl Acad Sci USA 2010(107):15904–15909
Gopalakrishnan V (2009) REST and the RESTless: in stem cells and beyond. Futur Neurol 4:317–329
Gu H, Roizman B (2003) The degradation of PML and Sp100 proteins by herpes simplex virus 1 is mediated by the ubiquitin-conjugating enzyme UbcH5a. Proc Natl Acad Sci USA 100:8963–8968
Gu H, Roizman B (2007) Herpes simplex virus-infected cell protein 0 blocks the silencing of viral DNA by dissociating histone deacetylases from the CoREST/REST complex. Proc Natl Acad Sci USA 104:17134–17139
Gu H, Roizman B (2009) The two functions of HSV-1 ICP0, inhibition of silencing by the CoREST/REST/HDAC complex and degradation of PML, are executed in tandem. J Virol 83:181–187
Gu H, Liang Y, Mandel G, Roizman B (2005) Components of the REST/CoREST/histone deacetylase repressor are disrupted, modified and translocated in HSV-1-infected cells. Proc Natl Acad Sci USA 102:7571–7576
Hagglund R, Van Sant C, Lopez P, Roizman B (2002) Herpes simplex virus 1-infected cell protein 0 contains two E3 ubiquitin ligase sites specific for different E2 ubiquitin-conjugating enzymes. Proc Natl Acad Sci USA 99:631–666
Javier RT, Stevens JG, Dessette VB, Wagner EK (1988) A herpes simplex virus transcript abundant in latently infected neurons is dispensable for establishment of the latent state. Virology 166:254–257
Kalamvoki M, Roizman B (2010) The circadian CLOCK histone acetyl transferase localizes at ND10 nuclear bodies and enables herpes simplex virus genes expression. Proc Natl Acad Sci USA 107:17721–17726
Kalamvoki M, Roizman B (2011) The histone acetyl transferase CLOCK is an essential component of the herpes simplex virus 1 transcriptome that includes TFIID, ICP4, ICP27 and ICP22. J Virol 85:9472–9477
Kawaguchi Y, Tanaka M, Yokoymama A, Matsuda G, Kato K, Kagawa H, Hirai K, Roizman B (2001) Herpes simplex virus 1 alpha regulatory protein ICP0 functionally interacts with cellular transcription factor BMAL1. Proc Natl Acad Sci USA 98:1877–1882
Knipe DM, Cliffe AR (2008) Chromatin control of herpes simplex virus lytic and latent infection. Nat Rev Microbiol 6:211–221
Kristie TM, Vogel JL, Sears AE (1999) Nuclear localization of the C1 factor (host cell factor) in sensory neurons correlates with reactivation of herpes simplex virus from latency. Proc Natl Acad Sci USA 96:1229–1233
Kutluay SB, Triezenberg SJ (2009) Regulation of histone deposition on the herpes simplex virus type 1 genome during lytic infection. J Virol 83:5835–5845
Kwiatkowski DL, Thompson HW, Bloom DC (2009) The polycomb group protein Bmi1 binds to the herpes simplex virus 1 latent genome and maintains repressive histone marks during latency. J Virol 83:8173–8181
Lacasse JJ, Schang LM (2010) During lytic infections, herpes simplex virus type 1 is in complexes with properties of unstable nucleosomes. J Virol 84:1920–1933
Lee MG, Wynder C, Cooch N, Shiekhattar R (2005) An essential role for CoREST in nucleosomal histone 3 lysine 4 demethylation. Nature 437:432–435
Liang Y, Vogel JL, Narayanan A, Peng H, Kristie TM (2009) Inhibition of the histone demethylase LSD1 blocks alpha-herpesvirus lytic replication and reactivation from latency. Nat Med 15:1312–1317
Maillet S, Naas T, Crepin S, Roque-Afonso AM, Lafay F, Efstathiou S, Labetoulle M (2006) Herpes simplex virus type 1 latently infected neurons differentially express latency-associated and ICP0 transcripts. J Virol 80:9310–9321
Metzger E, Wissmann M, Yin N, Muller J, Schneider R, Peters AHFM, Gunther T, Buettner S, Schule R (2005) LSD1 demethylates repressive histone marks to promote androgen-receptor-dependent transcription. Nature 437:436–439
Neumann DM, Bhattacharjee PS, Giordani NV, Bloom DC, Hill JM (2007) In vivo changes in the patterns of chromatin structure associated with the latent herpes simplex virus type 1 genome in mouse trigeminal ganglia can be detected at early times after butyrate treatment. J Virol 81:13248–13253
Roizman B (2011) The checkpoints of viral gene expression in productive and latent infection: the role of the HDAC/CoREST/LSD1/REST repressor complex. J Virol 85:7474–7482
Roizman B, Sears AE (1987) An inquiry into the mechanism of herpes simplex virus latency. Annu Rev Microbiol 41:543–571
Roizman B, Knipe DM, Whitley RJ et al (2007) Herpes simplex viruses. In: Knipe DM (ed) Fields virology, 5th edn. Lippincott Williams & Wilkins, New York, pp 2501–2601
Shi YJ, Matson C, Iwase S, Baba T, Shi Y (2005) Regulation of LSD1 histone demethylase activity by its associated factors. Mol Cell 19:857–864
Tang S, Bertke AS, Patel A, Wang K, Cohen JI, Krause PR (2008) An acutely and latently expressed herpes simplex virus 2 viral microRNA inhibits expression of ICP34.5, a viral neurovirulence factor. Proc Natl Acad Sci USA 105:10931–10936
Tapia-Ramírez J, Eggen BJ, Peral-Rubio MJ, Toledo-Aral JJ, Mandel G (1997) A single zinc finger motif in the silencing factor REST represses the neural-specific type II sodium channel promoter. Proc Natl Acad Sci USA 94:1177–1182
Thompson RL, Preston CM, Sawtell NM (2009) De novo synthesis of VP16 coordinates the exit from HSV latency in vivo. PLoS Pathog 5:e1000352
Umbach JL, Kramer MF, Jurak I, Karnowski HW, Coen DM, Cullen BR (2008) MicroRNAs expressed by herpes simplex virus 1 during latent infection regulate viral mRNAs. Nature 454:780–783
Umbach JL, Nagel M, Cohrs R, Gilden D, Cullen BR (2009) Analysis of human alphaherpesvirus microRNA expression in latently infected human trigeminal ganglia. J Virol 83:10677–10683
Wang K, Pesnicka L, Guancial E, Krause P, Straus S (2001) The 2.2-kilobase latency-associated transcript of herpes simplex virus type 2 does not modulate viral replication, reactivation, or establishment of latency in transgenic mice. J Virol 75:8166–8172
Wang QY, Zhou C, Johnson KE, Colgrove RC, Coen DM, Knipe DM (2005) Herpesviral latency-associated transcript gene promotes assembly of heterochromatin on viral lytic-gene promoters in latent infection. Proc Natl Acad Sci USA 102:16055–16059
Wu T, Su Y, Block T, Taylor J (1996) Evidence that two latency-associated transcripts of herpes simplex virus type 1 are nonlinear. J Virol 70:5962–5967
Yang M, Gocke C, Luo X, Borek D, Tomchick D, Machius M, Otwinowski Z, Yu H (2006) Structural basis for CoREST-dependent demethylation of nucleosomes by the human LSD1 histone demethylase. Mol Cell 23:377–387
Zhou G, Du T, Roizman B (2010) The CoREST/REST repressor is both necessary and inimical for expression of herpes simplex virus gene expression. MBio 2:e00313-10
Acknowledgments
These studies were aided by a grant from the National Cancer Institute grant R37 CA78766.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roizman, B., Zhou, G. & Du, T. Checkpoints in productive and latent infections with herpes simplex virus 1: conceptualization of the issues. J. Neurovirol. 17, 512–517 (2011). https://doi.org/10.1007/s13365-011-0058-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-011-0058-x