Abstract
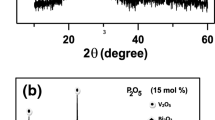
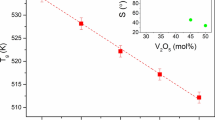
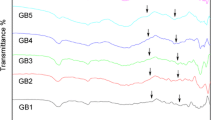
30B2O3-70V2O5 binary-system glass was prepared, and variations in structural and electrical property were examined using crystallization. While different related research studies exist, few have evaluated the variations in the structure and properties with changes in the crystallization rate. 30B2O3-70V2O5 glass was annealed in the graphite mold above the glass transition temperature for 2 h and heat-treated at each crystallization temperature for 3 h. 30B2O3-70V2O5 glass showed predominantly electronic conductive characteristic. FTIR was preferentially used for analyzing the structural changes of B-O bond after crystallization, while XRD was utilized to verify the inferred changes in the structure array (BO3 + V2O5 ↔ BO4 + 2VO2). Structural changes induced by heat treatment were confirmed by analyzing the molecular volume determined from the sample density, and conductance was measured to correlate structural and property changes. Conductivity is discussed based on the migration of vanadate ions with different valence states because of the increase in VO2 crystallinity at 130°C, which, however, was not observed at 170°C. After VO2 structures were reinforced, a 1.8-fold increase in conductance was observed (as compared to the annealed sample) after crystallization at 130°C for 3 h.
Similar content being viewed by others
References
I. Kashif, H. Farouk, S. A. Aly, A. A. Abdel-Rahman, and A. M. Sanad, Mater. Sci. Eng. B10, 1 (1991).
Y. Kawamoto, J. Tanida, H. Hamada, and H. Kiriyama, J. Non-Cryst. Solids 38, 301 (1980).
G. S. Linsley, A. E. Owen, and F. M. Hayatee, J. Non-Cryst. Solids 4, 208 (1970).
T. Allersma, R. Hakim, T. N. Kennedy, and J. D. Mackenzie, J. Chem. Phys. 1, 46 (1967).
Y. Limb and R. F. Davis, J. Am. Ceram. Soc. 26, 403 (1979).
H. Kyungseok, H. Chawon, K. Donghwan, G. Donnggun, K. Taehee, C. Wonngyu, K. Kyungbumb, K. Jonghwan, and R. Bongki, Electron. Mater. Lett. 8, 655 (2012).
M. L. Huggins and K. H. Sun, J. Am. Ceram. Soc. 26, 4 (1943).
L. G. Gehring and M. A. Knight, J. Am. Ceram. Soc. 27, 260 (1944).
Y. Ito, K. Miyauchi, and T. Oi, J. Non-Cryst. Solids 57, 389 (1983).
B. K. Sharma and D. C. Dube, J. Non-Cryst. Solids 65, 39 (1984).
B. N. Meera and J. Ramakrishna, J. Non-Cryst. Solids 159, 1 (1993).
E. I. Kamitsos, M. A. Karakassides, and G. D. Chryssikos, J. Phys. Chem. 91, 1067 (1987).
S. G. Motke, S. P. Yawale, and S. S. Yawale, Bull. Mater. Sci. 25, 75 (2002).
A. Abd-El-Moneim, Mater. Chem. Phys. 73, 318 (2002).
R. Lal and N. D. Sharma, Ind. J. Pure Appl. Phys. 43, 828 (2005).
A. A. Alemi, H. Sedghi, A. R. Mirmohseni, and V. Golsanamlu, Bull. Mater. Sci. 29, 55 (2006).
Y. D. Yiannopoulos, G. D. Chryssikos, and E. I. Kamitsos, Phys. Chem. Glasses 42, 164 (2001).
E. E. Horopanitis, G. Perentzis, A. Beck, L. Guczi, G. Peto, and Papadimitriou, J. Non-Cryst. Solids 354, 374 (2008).
E. Kamitos, A. Patsis, M. Karakassides, and G. Chryssikos, J. Non-Cryst. Solids 126, 52 (1990).
E. Kamitos, Karakassides, and G. Chryssikos, J. Phys. Chem. Glasses 91, 1073 (1987).
E. Kamitos, A. Patsis, and G. Chryssikos, J. Non-Cryst. Solids 152, 246 (1993).
V. Dimitrov, Y. Dimitriev, and A. Montenero, J. Non-Cryst. Solids 180, 51 (1994).
Y. Dimitriev, V. Dimitrov, M. Arnaudov, and D. Topalov, J. Non-Cryst. Solids 17, 147 (1983).
I. Plusnina, Infrared Spectra of Minerals, p. 67, MGU, MOSCOW (1977).
V. Kundu, R. L. Dhiman, A. S. Maan, and D. R. Goyal, Adv. Condens. Mater. Phys. 25, 324 (2008).
O. Gzowski, L. Murawskit, W. Lizakf, H. Binczyckat, and J. Sawicki, J. Phys. D: Appl. Phys. 14, L77 (1981).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gwoo, D., Kim, T., Han, K. et al. Electrical properties of crystallized 30B2O3-70V2O5 glass. Electron. Mater. Lett. 9, 309–313 (2013). https://doi.org/10.1007/s13391-013-2199-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13391-013-2199-y