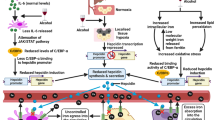
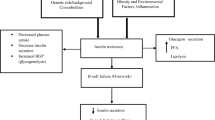
Abstract
Background
Beta-thalassemia major (β-TM) comprises a group of inherited blood disorders characterized by the reduced synthesis of β-globin chains. Iron overload following blood transfusion can affect major tissues involved in glucose metabolism, leading to different glucose metabolic disorders in these patients. The aim of the present study was to compare glucose metabolism and iron overload indices in β-TM patients with prediabetic and normoglycemic states.
Methods
This analytical study was performed on 49 patients with β-TM (age > 18 years), receiving regular blood transfusions. The fasting plasma glucose (FPG) and glucose tolerance tests indicated 32 normoglycemic and 17 prediabetic cases. The serum levels of C-peptide, fructosamine, fasting serum insulin, and serum ferritin were measured. In addition, T2*-weighted magnetic resonance imaging (MRI) of the heart and liver was carried out. Glycemic metabolism indices, including quantitative insulin sensitivity check index (QUICKI) and homeostatic model assessment for insulin resistance (HOMA-IR), were also calculated.
Results
The HOMA-IR score was significantly higher, while the QUICKI score was significantly lower in prediabetic patients, compared to normoglycemic patients (median [IR], 2.59 [2.19] vs. 1.46 [1.03], p = 0.007; mean ± SD, 0.34 ± 0.03 vs. 0.37 ± 0.04, p = 0.01). On the other hand, β-cell function was not significantly different between the groups. The liver iron concentration (LIC) at a cutoff point of 5.82 mg/g dry weight showed 93% sensitivity and 70% specificity for differentiation of prediabetic and normoglycemic states (AUC, 0.81 [95% CI, 0.65–0.95]; p = 0.002).
Conclusion
Based on the findings, HOMA-IR and QUICKI can be applied as useful glycemic metabolism indices for predicting prediabetic and normoglycemic states among β-TM patients. Also, LIC was an independent risk factor for the prediabetic state.


Similar content being viewed by others
References
Papakonstantinou O, Ladis V, Kostaridou S, Maris T, Berdousi H, Kattamis C, et al. The pancreas in β-thalassemia major: MR imaging features and correlation with iron stores and glucose disturbances. Eur Radiol. 2007;17(6):1535–43.
Cario H, Holl RW, Debatin K-M, Kohne E. Insulin sensitivity and β-cell secretion in thalassaemia major with secondary haemochromatosis: assessment by oral glucose tolerance test. Eur J Pediatr. 2003;162(3):139–46.
Karami H, Kosaryan M, Amree AH, Darvishi-Khezri H, Mousavi M. Combination iron chelation therapy with deferiprone and deferasirox in iron-overloaded patients with transfusion-dependent β-thalassemia major. Clin Pract. 2017;7(1).
Simcox JA, McClain DA. Iron and diabetes risk. Cell Metab. 2013;17(3):329–41.
Fernández-Real JM, Manco M. Effects of iron overload on chronic metabolic diseases. Lancet Diabetes Endocrinol. 2014;2(6):513–26.
Chern JP, Lin K-H, Lu M-Y, Lin D-T, Lin K-S, Chen J-D, et al. Abnormal glucose tolerance in transfusion-dependent β-thalassemic patients. Diabetes Care. 2001;24(5):850–4.
Ghergherehchi R, Habibzadeh A. Insulin resistance and β cell function in patients with β-thalassemia major. Hemoglobin. 2015;39(1):69–73.
Christoforidis A, Perifanis V, Tsatra I, Vlachaki E, Athanassiou-Metaxa M. Evolution of OGTT in patients with β-thalassaemia major in relation to chelation therapy. Diabetes Res Clin Pract. 2007;76(1):6–11.
Merkel PA, Simonson DC, Amiel SA, Plewe G, Sherwin RS, Pearson HA, et al. Insulin resistance and hyperinsulinemia in patients with thalassemia major treated by hypertransfusion. N Engl J Med. 1988;318(13):809–14.
Suvarna J, Ingle H, Deshmukh C. Insulin resistance and beta cell function in chronically transfused patients of thalassemia major. Indian Pediatr. 2006;43:393–400.
Dandona P, Hussain MA, Varghese Z, et al. Insulin resistance and iron overload. Ann Clin Biochem. 1983;20:77–9.
Sjaarda LG, Bacha F, Lee S, Tfayli H, Andreatta E, Arslanian S. Oral disposition index in obese youth from normal to prediabetes to diabetes: relationship to clamp disposition index. J Pediatr. 2012;161(1):51–7.
Messina M, Lombardo F, Meo A, Miceli M, Wasniewska M, Valenzise M, et al. Three-year prospective evaluation of glucose tolerance, β-cell function and peripheral insulin sensitivity in non-diabetic patients with thalassemia major. J Endocrinol Investig. 2002;25(6):497–501.
Rabasa-Lhoret RM, Bastard J-P, Jan VR, Ducluzeau P-H, Andreelli F, Guebre F, et al. Modified quantitative insulin sensitivity check index is better correlated to hyperinsulinemic glucose clamp than other fasting-based index of insulin sensitivity in different insulin-resistant states. J Clin Endocrinol Metab. 2003;88(10):4917–23.
Association AD. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37:S81–90.
Kosaryan M, Rahimi M, Darvishi-Khezri H, Gholizadeh N, Akbarzadeh R, Aliasgharian A. Correlation of pancreatic iron overload measured by T2*-weighted magnetic resonance imaging in diabetic patients with β-thalassemia major. Hemoglobin. 2017;41(3):151–6.
Bahar A, Shekarriz R, Janbabai G, Shirzad R, Aarabi M, Kashi Z. Insulin resistance, impaired glucose tolerance and alpha-thalassemia carrier state. J Diabetes Metab Disord. 2015;14(1):2.
Gutch M, Kumar S, Razi SM, Gupta KK, Gupta A. Assessment of insulin sensitivity/resistance. Indian J Endocrinol Metab. 2015;19(1):160–4.
Brittenham GM, Griffith PM, Nienhuis AW, McLaren CE, Young NS, Tucker EE, et al. Efficacy of deferoxamine in preventing complications of iron overload in patients with thalassemia major. N Engl J Med. 1994;331(9):567–73.
Angelopoulos N, Zervas A, Livadas S, Adamopoulos I, Giannopoulos D, Goula A, et al. Reduced insulin secretion in normoglycaemic patients with β-thalassaemia major. Diabet Med. 2006;23(12):1327–31.
Olivieri NF, Nathan DG, MacMillan JH, Wayne AS, Liu PP, McGee A, et al. Survival in medically treated patients with homozygous β-thalassemia. N Engl J Med. 1994;331(9):574–8.
Gamberini M, De VS, Gilli G. Hypogonadism, diabetes mellitus, hypothyroidism, hypoparathyroidism: incidence and prevalence related to iron overload and chelation therapy in patients with thalassaemia major followed from 1980 to 2007 in the Ferrara Centre. Pediatr Endocrinol Rev. 2008;6:158–69.
Aregbesola A, Voutilainen S, Virtanen JK, et al. Serum hepcidin concentrations and type 2 diabetes. World J Diabetes. 2015;10(6):978.
Le Guenno G, Chanséaume E, Ruivard M, Morio B, Mazur A. Study of iron metabolism disturbances in an animal model of insulin resistance. Diabetes Res Clin Pract. 2007;77(3):363–70.
Sam A, Busbridge M, Amin A, Webber L, White D, Franks S, et al. Hepcidin levels in diabetes mellitus and polycystic ovary syndrome. Diabet Med. 2013;30(12):1495–9.
Fernández-Real JM, Equitani F, JMa M, Manco M, Ortega F, Ricart W. Study of circulating prohepcidin in association with insulin sensitivity and changing iron stores. J Clin Endocrinol Metab. 2009;94(3):982–8.
Aso Y, Takebayashi K, Wakabayashi S, Momobayashi A, Sugawara N, Terasawa T, et al. Relation between serum high molecular weight adiponectin and serum ferritin or prohepcidin in patients with type 2 diabetes. Diabetes Res Clin Pract. 2010;90(3):250–5.
Acknowledgments
We would like to thank β-TM patients, as well as the TRC staff, for their sincere cooperation. This study was extracted from a thesis in pediatrics. We would also like to thank the Research Deputy of Mazandaran University of Medical Sciences (Sari, Iran) for the financial support and the University Ethics Committee for accepting our proposal.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Statement of human rights
The study conformed to the Declaration of Helsinki principles.
Informed consent
All subjects gave informed consent and were informed of their ability to discontinue participation at any time.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix. Interpretation and categorization of cardiac and liver MRI
Appendix. Interpretation and categorization of cardiac and liver MRI
Rights and permissions
About this article
Cite this article
Kosaryan, M., Rahimi, M., Zamanfar, D. et al. Liver iron concentration is an independent risk factor for the prediabetic state in β-thalassemia patients. Int J Diabetes Dev Ctries 40, 227–234 (2020). https://doi.org/10.1007/s13410-019-00789-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-019-00789-0