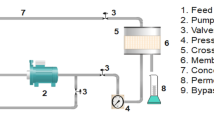
Abstract
This study introduces poly(amidoamine) PAMAM dendrimers as new macromolecular complexation agents for Co(II) removal in polymer-assisted ultrafiltration (PAUF). A five-level three-factor design of experiments (DOE), central composite design type, and response surface methodology (RSM) were used together to find out the interaction effects and optimize three variables, i.e., initial Co(II) concentration ([Co2+]0), PAMAM dendrimer-to-Co(II) ratio (r), and pH of solution. Multiple-response simultaneous optimization was conducted by using desirability function. The goal of 73.6% overall desirability was attained for the removal efficiency (R) and metal retention capacity (q). The predicted results obtained for the simultaneous optimization are R = 76.78% and q = 392.09 mg/g. The optimum conditions derived via RSM were found to be as follows: [Co2+]0 = 4.14 mg/L, r = 2, and pH = 9.0. Verification experiments (R = 75.73% and q = 387.32 mg/g) confirmed the validity of the predicted model. The DOE–RSM-utilized and desirability function-optimized PAMAM dendrimer-enhanced ultrafiltration (PAMAM-DEUF) process was used for the first time in this study. The results are comparable to those provided by the reference PAUF technique.
Graphical Abstract








Similar content being viewed by others
References
R. Solanki, R. Dhankhar, Biochemical changes and adaptive strategies of plants under heavy metal stress. Biologia 66(2), 195–204 (2011). https://doi.org/10.2478/s11756-011-0005-6
H.A. Wayland, S.N. Boury, B.P. Chhetri, A. Brandt, M.A. Proskurnin, V.A. Filichkina, V.P. Zharov, A.S. Biris, A. Ghosh, Advanced cellulosic materials for treatment and detection of industrial contaminants in wastewater. Chem Sel 1(15), 4472–4488 (2016). https://doi.org/10.1002/slct.201600653
A. Tripathi, M.R. Ranjan, Heavy metal removal from wastewater using low cost adsorbents. J. Bioremdiat. Biodegrad. 6(6), 1000315/1000311–1000315/1000315 (2015). https://doi.org/10.4172/2155-6199.1000315
F. Qin, B. Wen, X.Q. Shan, Y.N. Xie, T. Liu, S.Z. Zhang, S.U. Khan, Mechanisms of competitive adsorption of Pb, Cu, and Cd on peat. Environ. Pollut. 144(2), 669–680 (2006). https://doi.org/10.1016/j.envpol.2005.12.036
S.E. Bailey, T.J. Olin, R.M. Bricka, D.D. Adrian, A review of potentially low-cost sorbents for heavy metals. Water Res. 33(11), 2469–2479 (1999). https://doi.org/10.1016/S0043-1354(98)00475-8
A. Netzer, D.E. Hughes, Adsorption of copper, lead and cobalt by activated carbon. Water Res. 18(8), 927–933 (1984). https://doi.org/10.1016/0043-1354(84)90241-0
C. Gómez-Lahoz, F. García-Herruzo, J.M. Rodríguez-Maroto, J.J. Rodríguez, Cobalt(II) removal from water by chemical reduction with sodium borohydride. Water Res. 27(6), 985–992 (1993). https://doi.org/10.1016/0043-1354(93)90062-M
WHO, World Health Organization, A compendium of standards for wastewater reuse in the Eastern Mediterranean Region, (World Health Organization Regional Office for the Eastern Mediterranean, Cairo, Egypt; Regional Centre for Environmental Health Activities CEHA, Amman, Jordan, 2006)
E.I. Hamilton, The geobiochemistry of cobalt. Sci. Total Environ. 150(1–3), 7–39 (1990). https://doi.org/10.1016/0048-9697(94)90126-0
A. Kabata-Pendias, H. Pendias, Trace Elements in Soils and Plants (CRC Press, Boca Raton, 1984), pp. 238–245
Q. Wang, L. Chen, Y. Sun, Removal of radiocobalt from aqueous solution by oxidized MWCNT. J. Radioanal. Nucl. Chem. 291(3), 787–795 (2012). https://doi.org/10.1007/s10967-011-1352-z
Y. Huang, L. Chen, H. Wang, Removal of Co(II) from aqueous solution by using hydroxyapatite. J. Radioanal. Nucl. Chem. 291(3), 777–785 (2012). https://doi.org/10.1007/s10967-011-1351-0
K. Shang, Y.Z. Yang, J.X. Guo, W.J. Lu, F. Liu, W. Wang, Extraction of cobalt by the AOT microemulsion system. J. Radioanal. Nucl. Chem. 291(3), 629–633 (2012). https://doi.org/10.1007/s10967-011-1443-x
H. Omar, H. Arida, A. Daifullah, Adsorption of 60Co radionuclides from aqueous solution by raw and modified bentonite. Appl. Clay Sci. 44(1–2), 21–26 (2009). https://doi.org/10.1016/j.clay.2008.12.013
J. Oliva, J. De Pablo, J.L. Cortina, J. Cama, C. Ayora, Removal of cadmium, copper, nickel, cobalt and mercury from water by Apatite II™: column experiments. J. Hazard. Mater. 194, 312–323 (2011). https://doi.org/10.1016/j.jhazmat.2011.07.104
A. Ahmadpour, M. Tahmasbi, T.R. Bastami, J.A. Besharati, Rapid removal of cobalt ion from aqueous solutions by almond green hull. J. Hazard. Mater. 166(2–3), 925–930 (2009). https://doi.org/10.1016/j.jhazmat.2008.11.103
J. Mizera, G. Mizerová, V. Machovič, L. Borecká, Sorption of cesium, cobalt and europium on low-rank coal and chitosan. Water Res. 41(3), 620–626 (2007). https://doi.org/10.1016/j.watres.2006.11.008
V.K. Gupta, C.K. Jain, I. Ali, M. Sharma, V.K. Saini, Removal of cadmium and nickel from wastewater using bagasse fly ash—a sugar industry waste. Water Res. 37(16), 4038–4044 (2003). https://doi.org/10.1016/S0043-1354(03)00292-6
V.K. Gupta, I. Ali, T.A. Saleh, A. Nayak, S. Agarwal, Chemical treatment technologies for waste-water recycling—an overview. RSC Adv. 2(16), 6380–6388 (2012). https://doi.org/10.1039/c2ra20340e
M. Nourbakhsh, Y. Sag, D. Ozer, Z. Aksu, T. Kutsal, A. Caglar, A comparative-study of various biosorbents for removal of chromium(VI) ions from industrial-waste waters. Process Biochem. 29(1), 1–5 (1994). https://doi.org/10.1016/0032-9592(94)80052-9
P. Xu, G.M. Zeng, D.L. Huang, C.L. Feng, S. Hu, M.H. Zhao, C. Lai, Z. Wei, C. Huang, G.X. Xie, Z.F. Liu, Use of iron oxide nanomaterials in wastewater treatment: a review. Sci. Total Environ. 424, 1–10 (2012). https://doi.org/10.1016/j.scitotenv.2012.02.023
B.Y. Spivakov, K. Geckeler, E. Bayer, Liquid-phase polymer-based retention—the separation of metals by ultrafiltration on polychelatogens. Nature 315(6017), 313–315 (1985)
M.K. Aroua, F.M. Zuki, N.M. Sulaiman, Removal of chromium ions from aqueous solutions by polymer-enhanced ultrafiltration. J. Hazard. Mater. 147(3), 752–758 (2007). https://doi.org/10.1016/j.jhazmat.2007.01.120
B.L. Rivas, E.D. Pereira, I. Moreno-Villoslada, Water-soluble polymer–metal ion interactions. Prog. Polym. Sci. 28(2), 173–208 (2003). https://doi.org/10.1016/S0079-6700(02)00028-X
M.S. Diallo, S. Christie, P. Swaminathan, L. Balogh, X. Shi, W. Um, C. Papelis, W.A. Goddard III, J.H. Johnson Jr., Dendritic chelating agents. 1. Cu(II) binding to ethylene diamine core poly(amidoamine) dendrimers in aqueous solutions. Langmuir 20(7), 2640–2651 (2004). https://doi.org/10.1021/la036108k
M. Tulu, K.E. Geckeler, Synthesis and properties of hydrophilic polymers. Part 7. Preparation, characterization and metal complexation of carboxy-functional polyesters based on poly(ethylene glycol). Polym. Int. 48(9), 909–914 (1999). https://doi.org/10.1002/(sici)1097-0126(199909)48:9<909::aid-pi244>3.0.co;2-e
M.S. Diallo, S. Christie, P. Swaminathan, J.H. Johnson, W.A. Goddard, Dendrimer enhanced ultrafiltration. 1. Recovery of Cu(II) from aqueous solutions using PAMAM dendrimers with ethylene diamine core and terminal NH2 groups. Environ. Sci. Technol. 39(5), 1366–1377 (2005). https://doi.org/10.1021/es048961r
L. Dambies, A. Jaworska, G. Zakrzewska-Trznadel, B. Sartowska, Comparison of acidic polymers for the removal of cobalt from water solutions by polymer assisted ultrafiltration. J. Hazard. Mater. 178(1–3), 988–993 (2010). https://doi.org/10.1016/j.jhazmat.2010.02.035
B.L. Rivas, E.D. Pereira, I. Moreno-Villoslada, Water-soluble polymer–metal ion interactions. Prog. Polym. Sci. 28(2), 173–208 (2003). https://doi.org/10.1016/S0079-6700(02)00028-X
A. Rether, M. Schuster, Selective separation and recovery of heavy metal ions using water-soluble N-benzoylthiourea modified PAMAM polymers. React. Funct. Polym. 57(1), 13–21 (2003). https://doi.org/10.1016/j.reactfunctpolym.2003.06.002
F. Zeng, S.C. Zimmerman, Dendrimers in supramolecular chemistry: from molecular recognition to self-assembly. Chem. Rev. 97(5), 1681–1712 (1997)
L. Balogh, D.A. Tomalia, Poly(amidoamine) dendrimer-templated nanocomposites. 1. Synthesis of zerovalent copper nanoclusters. J. Am. Chem. Soc. 120(29), 7355–7356 (1998). https://doi.org/10.1021/ja980861w
M.S. Diallo, L. Balogh, A. Shafagati, J.H. Johnson, W.A. Goddard, D.A. Tomalia, Poly(amidoamine) dendrimers: a new class of high capacity chelating agents for Cu(II) ions. Environ. Sci. Technol. 33(5), 820–824 (1999). https://doi.org/10.1021/es980521a
C.W. Li, C.H. Cheng, K.H. Choo, W.S. Yen, Polyelectrolyte enhanced ultrafiltration (PEUF) for the removal of Cd(II): effects of organic ligands and solution pH. Chemosphere 72(4), 630–635 (2008). https://doi.org/10.1016/j.chemosphere.2008.02.036
R. Molinari, T. Poerio, P. Argurio, Chemical and operational aspects in running the polymer assisted ultrafiltration for separation of copper(II)–citrate complexes from aqueous media. J. Membr. Sci. 295(1–2), 139–147 (2007). https://doi.org/10.1016/j.memsci.2007.03.002
C. Cojocaru, G. Zakrzewska-Trznadel, A. Jaworska, Removal of cobalt ions from aqueous solutions by polymer assisted ultrafiltration using experimental design approach. Part 1: optimization of complexation conditions. J. Hazard. Mater. 169(1), 599–609 (2009). https://doi.org/10.1016/j.jhazmat.2009.03.145
N. Uzal, A. Jaworska, A. Miśkiewicz, G. Zakrzewska-Trznadel, C. Cojocaru, Optimization of Co2+ ions removal from water solutions via polymer enhanced ultrafiltration with application of PVA and sulfonated PVA as complexing agents. J. Colloid Interface Sci. 362(2), 615–624 (2011). https://doi.org/10.1016/j.jcis.2011.06.072
G. Derringer, R. Suich, Simultaneous optimization of several response variables. J. Qual. Technol. 12(4), 214–219 (1980)
L. Vera Candioti, M.M. De Zan, M.S. Cámara, H.C. Goicoechea, Experimental design and multiple response optimization. Using the desirability function in analytical methods development. Talanta 124, 123–138 (2014). https://doi.org/10.1016/j.talanta.2014.01.034
E. Ghasemi, A. Heydari, M. Sillanpaa, Superparamagnetic Fe3O4@EDTA nanoparticles as an efficient adsorbent for simultaneous removal of Ag(I), Hg(II), Mn(II), Zn(II), Pb(II) and Cd(II) from water and soil environmental samples. Microchem. J. 131, 51–56 (2017). https://doi.org/10.1016/j.microc.2016.11.011
M. Amini, H. Younesi, N. Bahramifar, Statistical modeling and optimization of the cadmium biosorption process in an aqueous solution using Aspergillus niger. Colloids Surf. A 337(1–3), 67–73 (2009). https://doi.org/10.1016/j.colsurfa.2008.11.053
M. Amini, H. Younesi, Biosorption of Cd(II), Ni(II), and Pb(II) from aqueous solution by dried biomass of Aspergillus niger: application of response surface methodology to the optimization of process parameters. Clean Soil Air Water 37(10), 776–786 (2009). https://doi.org/10.1002/clen.200900090
M. Amini, H. Younesi, N. Bahramifar, A.A.Z. Lorestani, F. Ghorbani, A. Daneshi, M. Sharifzadeh, Application of response surface methodology for optimization of lead biosorption in an aqueous solution by Aspergillus niger. J. Hazard. Mater. 154(1–3), 694–702 (2008). https://doi.org/10.1016/j.jhazmat.2007.10.114
A.S. Ertürk, M.U. Gürbüz, M. Tülü, The effect of PAMAM dendrimer concentration, generation size and surface functional group on the aqueous solubility of candesartan cilexetil. Pharm. Dev. Technol. 22(1), 111–121 (2017). https://doi.org/10.1080/10837450.2016.1219372
H. Watanabe, Spectrophotometric determination of cobalt with 1-(2-pyridylazo)-2-naphthol and surfactants. Talanta 21(4), 295–302 (1974). https://doi.org/10.1016/0039-9140(74)80007-X
D. Rana, T. Matsuura, M. Kassim, A. Ismail, Radioactive decontamination of water by membrane processes—a review. Desalination 321, 77–92 (2013)
M.A. Bezerra, R.E. Santelli, E.P. Oliveira, L.S. Villar, L.A. Escaleira, Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76(5), 965–977 (2008). https://doi.org/10.1016/j.talanta.2008.05.019
R.H. Myers, D.C. Montgomery, C.M. Anderson-Cook, Response Surface Methodology: Process and Product Optimization Using Designed Experiments (Wiley, Hoboken, 2016)
W.G. Cochran, G.M. Cox, Experimental designs (Wiley, Hoboken, 1957)
M.S. Diallo, W. Arasho, J.H. Johnson Jr., W.A. Goddard Iii, Dendritic chelating agents. 2. U(VI) binding to poly(amidoamine) and poly(propyleneimine) dendrimers in aqueous solutions. Environ. Sci. Technol. 42(5), 1572–1579 (2008)
M. Palencia, B.L. Rivas, E. Pereira, A. Hernández, P. Prádanos, Study of polymer–metal ion–membrane interactions in liquid-phase polymer-based retention (LPR) by continuous diafiltration. J. Membr. Sci. 336(1–2), 128–139 (2009). https://doi.org/10.1016/j.memsci.2009.03.016
B.L. Rivas, E.D. Pereira, M. Palencia, J. Sánchez, Water-soluble functional polymers in conjunction with membranes to remove pollutant ions from aqueous solutions. Prog. Polym. Sci. 36(2), 294–322 (2011). https://doi.org/10.1016/j.progpolymsci.2010.11.001
J.W. Osborne, Improving your data transformations: applying the Box–Cox transformation. Pract. Assess. Res. Eval. 15(12), 2 (2010)
M.J. Anderson, P.J. Whitcomb, RSM Simplified: Optimizing Processes Using Response Surface Methods for Design of Experiments (Taylor & Francis, New York, 2005)
L.V. Candioti, J.C. Robles, V.E. Mantovani, H.C. Goicoechea, Multiple response optimization applied to the development of a capillary electrophoretic method for pharmaceutical analysis. Talanta 69(1), 140–147 (2006). https://doi.org/10.1016/j.talanta.2005.09.021
J. Zolgharnein, A. Shahmoradi, J.B. Ghasemi, Comparative study of Box–Behnken, central composite, and Doehlert matrix for multivariate optimization of Pb(II) adsorption onto Robinia tree leaves. J. Chemom. 27(1–2), 12–20 (2013). https://doi.org/10.1002/cem.2487
J. Zolgharnein, N. Asanjarani, T. Shariatmanesh, Taguchi L16 orthogonal array optimization for Cd(II) removal using Carpinus betulus tree leaves: adsorption characterization. Int. Biodeterior. Biodegrad. 85, 66–77 (2013). https://doi.org/10.1016/j.ibiod.2013.06.010
J. Zolgharnein, A. Shahmoradi, Adsorption of Cr(VI) onto Elaeagnus tree leaves: statistical optimization, equilibrium modeling, and kinetic studies. J. Chem. Eng. Data 55(9), 3428–3437 (2010). https://doi.org/10.1021/je100157y
K. Geckeler, V. N. Rajasekharan Pillai, M. Mutter, Applications of soluble polymeric supports, in Polymer Products (Springer, Berlin, 1981), pp 65–94. https://doi.org/10.1007/3-540-10218-3_2
E. Bayer, B.Y. Spivakov, K. Geckeler, Poly(ethyleneimine) as complexing agent for separation of metal ions using membrane filtration. Polym. Bull. 13(4), 307–311 (1985). https://doi.org/10.1007/bf00262113
F. Fang, L. Kong, J. Huang, S. Wu, K. Zhang, X. Wang, B. Sun, Z. Jin, J. Wang, X.-J. Huang, J. Liu, Removal of cobalt ions from aqueous solution by an amination graphene oxide nanocomposite. J. Hazard. Mater. 270, 1–10 (2014). https://doi.org/10.1016/j.jhazmat.2014.01.031
GdC Pizarro, O.G. Marambio, M. Jeria-Orell, D.P. Oyarzún, B.L. Rivas, W.D. Habicher, Preparation, characterization, and metal ion retention capacity of Co(II) and Ni(II) from poly(p-HO- and p-Cl-phenylmaleimide-co-2-hydroxypropylmethacrylate) using the ultra filtration technique. J. Appl. Polym. Sci. 106(4), 2448–2455 (2007). https://doi.org/10.1002/app.26948
O.G. Marambio, GdC Pizarro, M. Jeria-Orell, M. Huerta, C. Olea-Azar, W.D. Habicher, Poly(N-phenylmaleimide-co-acrylic acid)–copper(II) and poly(N-phenylmaleimide-co-acrylic acid)–cobalt(II) complexes: synthesis, characterization, and thermal behavior. J. Polym. Sci. Part A: Polym. Chem. 43(20), 4933–4941 (2005). https://doi.org/10.1002/pola.20955
P. Ilaiyaraja, A. Deb, D. Ponraju, Removal of uranium and thorium from aqueous solution by ultrafiltration (UF) and PAMAM dendrimer assisted ultrafiltration (DAUF). J. Radioanal. Nucl. Chem. 303(1), 441–450 (2015). https://doi.org/10.1007/s10967-014-3462-x
Acknowledgements
This research has been supported by Adıyaman University Scientific Research Projects Coordination Department (Project Number: MÜFMAP/2015-0004). There is no conflict of interest. The author is grateful to his wife, Assist. Prof. Dr. Müzeyyen Ertürk, for many helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ertürk, A.S. PAMAM dendrimer-enhanced removal of cobalt ions based on multiple-response optimization using response surface methodology. J IRAN CHEM SOC 15, 1685–1698 (2018). https://doi.org/10.1007/s13738-018-1366-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1366-3