Abstract
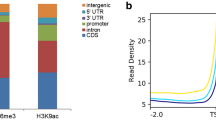
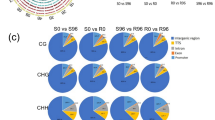
Powdery mildew has been recognized as a destructive foliar disease for Tibetan hulless barley and causes huge economic losses every year. However, little is known about the genetic and molecular basis of resistance to powdery mildew in hulless barley, especially the epigenetic mechanism. To understand the change patterns of histone modification after pathogen inoculation, we performed a genome-wide analysis of H3K4 and H3K27 trimethylations of samples from five time points using two different barley varieties (G7 is resistant to powdery mildew and Z13 is susceptible). By integration analysis of ChIP-seq and transcriptome data, we found that more than 80% of differential expressed genes may be regulated by differentially chromatin modification, suggesting that epigenetic changes have an important regulatory role in resistance response to powdery mildew. Our study provides a first glimpse into epigenetic regulation in disease response in barley, and these data represent a resource to the community for future exploitation of molecular mechanisms underlying disease resistance response.






Similar content being viewed by others
References
Bender J (2002) Plant epigenetics. Curr Biol 12(12):R412–R414
Biselli C, Bulgarelli D, Collins NC, Schulze-Lefert P, Stanca AM, Cattivelli L, Valè G (2013) The cc-nb-lrr-type rdg2a resistance gene evolved through recombination and confers immunity to the seed-borne barley leaf stripe pathogen in the absence of hypersensitive cell death. Springer, Dordrecht
Caplan J, Padmanabhan M, Dineshkumar SP (2008) Plant nb-lrr immune receptors: From recognition to transcriptional reprogramming. Cell Host Microbe 3(3):126–135
Cizmeci D, Dempster EL, Champion OL, Wagley S, Akman OE, Prior JL, Soyer OS, Mill J, Titball RW (2016) Mapping epigenetic changes to the host cell genome induced by burkholderia pseudomallei reveals pathogen-specific and pathogen-generic signatures of infection. Sci Rep 6:30861
Coleman C, Copetti D, Cipriani G, Hoffmann S, Kozma P, Kovács L, Morgante M, Testolin R, Gaspero GD (2009) The powdery mildew resistance gene ren1 co-segregates with an nbs-lrr gene cluster in two central asian grapevines. BMC Genet 10(1):89
Ea VDB, Freddie CT, Kahn K, Parker JE, Jones JD (2010) Arabidopsis rpp4 is a member the rpp5 multigene family of tir-nb-lrr genes and confers downy mildew resistance through multiple signaling components. Plant J 29(4):439–451
Feinberg AP (2007) Phenotypic plasticity and the epigenetics of human disease. Nature 447(7143):433–440
Fga B (2010) Resistance to powdery mildew in wheat: A review of its use in agriculture and breeding programmes. Plant Pathol 33(3):279–300
Freymark G, Diehl T, Miklis M, Romeis T, Panstruga R (2007) Antagonistic control of powdery mildew host cell entry by barley calcium-dependent protein kinases (cdpks). Molecular plant-microbe interactions: MPMI, 20(10):1213
Gómezdíaz E, Jordà M, Peinado MA, Rivero A (2012) Epigenetics of host–pathogen interactions: The road ahead and the road behind. PLoS Pathog 8(11):e1003007
Herrera CM, Medrano M, Bazaga P (2016) Comparative spatial genetics and epigenetics of plant populations: Heuristic value and a proof of concept. Mol Ecol 25(8):1653–1664
Hunt BG, Ometto L, Wurm Y, Shoemaker D, Yi SV, Keller L, Goodisman MA (2011) Relaxed selection is a precursor to the evolution of phenotypic plasticity. Proc Natl Acad Sci USA 108(38):15936–15941
Jaligot E, Rival A (2011) Applying epigenetics in plant breeding: Balancing genome stability and phenotypic plasticity
Jong GD (2010) Evolution of phenotypic plasticity: Patterns of plasticity and the emergence of ecotypes. New Phytol 166(1):101–118
Kleinmanns JA, Schubert D (2014) Polycomb and Trithorax group protein-mediated control of stress responses in plants. Biol Chem 395(11):1291–1300. https://doi.org/10.1515/hsz-2014-0197
Lämke J, Bäurle I (2017) Epigenetic and chromatin-based mechanisms in environmental stress adaptation and stress memory in plants. Genome Biol 18(1):124
Qian J, Jiang S, Su W, Ping G (2009) Characteristics of oil from hulless barley (hordeum vulgare l.) bran from tibet. J Am Oil Chem Soc 86(12):1175–1179
Rapp RA, Wendel JF (2010) Epigenetics and plant evolution. New Phytol 168(1):81–91
Ridout CJ, Skamnioti P, Porritt O, Sacristan S, Jones JD, Brown JK (2006) Multiple avirulence paralogues in cereal powdery mildew fungi may contribute to parasite fitness and defeat of plant resistance. Plant Cell 18(9):2402–2414
Sikkink KL, Reynolds RM, Ituarte CM, Cresko WA, Phillips PC (2014) Rapid evolution of phenotypic plasticity and shifting thresholds of genetic assimilation in the nematode caenorhabditis remanei. G3 (Bethesda, Md.) 4(6):1103–1112
Spanu PD, Abbott JC, Amselem J, Burgis TA, Soanes DM, Stüber K, Ver LvTE, Brown JK, Butcher SA, Gurr SJ (2010) Genome expansion and gene loss in powdery mildew fungi reveal tradeoffs in extreme parasitism. Science 330(6010):1543–1546
Uchida N, Tasaka M (2011) Regulation of nb-lrr-type uni and its related signaling pathway: Signaling crosstalk and methodology for quick identification of related factors. Plant Signal Behav 6(8):1219–1222
Wissler, Josef H (2012) Tuberculosis and epigenetics: Functional noncoding rna in the host-pathogen interaction. FASEB J 26(12)
Yuan HJ (2014) Identification of powdery mildew resistance of tibet hulless barley germplasm. Barley & Cereal Sciences
Yuan H, Zeng X, Yang Q, Xu Q, Wang Y, Jabu D, ... Tashi N (2018) Gene coexpression network analysis combined with metabonomics reveals the resistance responses to powdery mildew in Tibetan hulless barley. Sci Rep 8(1):14928. https://doi.org/10.1038/s41598-018-33113-7
Zeng XQ, Luo XM, Wang YL, Xu QJ, Bai LJ, Yuan HJ, Tashi N (2014) Transcriptome sequencing in a tibetan barley landrace with high resistance to powdery mildew. Sci World J 2014(4):594579
Zeng X, Wang Y, Xu Q, Yuan H, Wei Z, Ni M (2015) Cloning and characterization of hbsnrk2.4 in tibetan hulless barley (Hordeum vulgare L. var. Nudum hk.F.). Biotechnol Bull 31(2):116–121
Acknowledgements
The work was supported grants from the National Key R&D Program of China (2018YFD1000703) and the Tibet Financial Special Fund(XZNKY-2019-C-051,2017CZZX001).
Author information
Authors and Affiliations
Contributions
Qijun Xu ,Sang Zha and Chunbao Yang conceived and supervised the work. Xingquan Zeng analyzed the RNA-seq data. Zeqing Li and Yulin Wang performed the ChIP-seq experiments. Zeqing Li analyzed the ChIP-seq data. Hongjun Yuan and Mingzhai Yu planted the hulless barley and collected the samples. Qijun Xu ,Sang Zha and Chunbao Yang wrote the article with contributions from all other authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zha, S., Yang, C., Zeng, X. et al. Comparative analysis of H3K4 and H3K27 trimethylations in two contrasting Tibetan hulless barely varieties on powdery mildew infection. J Plant Pathol 103, 117–126 (2021). https://doi.org/10.1007/s42161-020-00673-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-020-00673-5