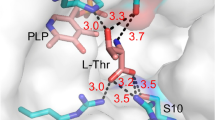
Abstract
l-Threonine transaldolase could catalyze the transaldolation of l-threonine and aldehyde to generate β-hydroxy-α-amino acids with high diastereoselectivity. A novel l-threonine transaldolase (PmLTTA) was identified from Pseudomonas sp. through genome mining. PmLTTA exhibited high activity in the synthesis of l-threo-phenylserine from l-threonine and benzaldehyde, with specific activity of 5.48 U mg–1. However, the application of PmLTTA was impeded by the low conversion ratio and variable diastereoselectivity, which were caused by the toxicity of aldehydes and kinetic/thermodynamic controls in the transaldolation reaction. To solve these issues, alcohol dehydrogenase was used to remove the by-product acetaldehyde, and then carboxylic acid reductase was introduced to alleviate the inhibition of benzaldehyde and toxicity of DMSO. Finally, a multi-enzyme cascade reaction, comprising of PmLTTA, carboxylic acid reductase, alcohol dehydrogenase and glucose dehydrogenase, was constructed to prepare l-threo-phenylserine from cheap benzoic acid, in which alleviated inhibition of aldehydes and desirable diastereoselectivity were achieved. Under the optimized conditions, the conversion ratio of 57.1% and de value of 95.3% were reached. This study provides an efficient and green approach for the synthesis of chiral l-threo-phenylserine from industrial byproduct, and provides guidance for the development of cascade reactions influenced by the toxic intermediates and complicated kinetic/thermodynamic controls.






Similar content being viewed by others
References
Barbie P, Kazmaier U. Total synthesis of cyclomarin A, a marine cycloheptapeptide with anti-tuberculosis and anti-malaria activity. Org Lett. 2016;18(2):204–7. https://doi.org/10.1021/acs.orglett.5b03292.
Gwon HJ, Baik SH. Diastereoselective synthesis of L: -threo-3,4-dihydroxyphenylserine by low-specific L: -threonine aldolase mutants. Biotechnol Lett. 2010;32(1):143–9. https://doi.org/10.1007/s10529-009-0125-z.
Zhao W, Yang B, Zha R, Zhang Z, Tang S, Pan Y, Qi N, Zhu L, Wang B. A recombinant L-threonine aldolase with high diastereoselectivity in the synthesis of L-threo-dihydroxyphenylserine. Biochem Eng J. 2020. https://doi.org/10.1016/j.bej.2020.107852.
Lamotte G, Holmes C, Sullivan P, Goldstein DS. Substantial renal conversion of L-threo-3,4-dihydroxyphenylserine (droxidopa) to norepinephrine in patients with neurogenic orthostatic hypotension. Clin Auton Res. 2019;29(1):113–7. https://doi.org/10.1007/s10286-018-0564-5.
Zhao GH, Li H, Liu W, Zhang WG, Zhang F, Liu Q, Jiao QC. Preparation of optically active beta-hydroxy-alpha-amino acid by immobilized Escherichia coli cells with serine hydroxymethyl transferase activity. Amino Acids. 2011;40(1):215–20. https://doi.org/10.1007/s00726-010-0637-9.
Chen Q, Chen X, Feng J, Wu Q, Zhu D, Ma Y. Improving and inverting Cβ-stereoselectivity of threonine aldolase via substrate-binding-guided mutagenesis and a stepwise visual screening. ACS Catal. 2019;9(5):4462–9. https://doi.org/10.1021/acscatal.9b00859.
Ashford PA, Bew SP. Recent advances in the synthesis of new glycopeptide antibiotics. Chem Soc Rev. 2012;41(3):957–78. https://doi.org/10.1039/c1cs15125h.
Masruri WAC, McLeod MD. Osmium-catalyzed vicinal oxyamination of alkenes by N-(4-toluenesulfonyloxy)carbamates. J Org Chem. 2012;77(19):8480–91. https://doi.org/10.1021/jo301372y.
Katsuki T, Sharpless KB. The first practical method for asymmetric epoxidation. J Am Chem Soc. 1980;102(18):5974–6. https://doi.org/10.1021/ja00538a077.
Levitskiy OA, Grishin YK, Magdesieva TV. Stereoselective electrosynthesis of β-hydroxy-α-amino acids in the form of NiII-schiff-base complexes. Eur J Org Chem. 2019;2019(20):3174–82. https://doi.org/10.1002/ejoc.201900466.
Xu L, Wang L-C, Xu X-Q, Lin J. Characteristics of l-threonine transaldolase for asymmetric synthesis of β-hydroxy-α-amino acids. Catal Sci Technol. 2019;9(21):5943–52. https://doi.org/10.1039/c9cy01608b.
Xu L, Wang LC, Su BM, Xu XQ, Lin J. Multi-enzyme cascade for improving beta-hydroxy-alpha-amino acids production by engineering L-threonine transaldolase and combining acetaldehyde elimination system. Bioresour Technol. 2020;310: 123439. https://doi.org/10.1016/j.biortech.2020.123439.
Česnik M, Sudar M, Roldan R, Hernandez K, Parella T, Clapés P, Charnock S, Vasić-Rački Đ, Findrik Blažević Z. Model-based optimization of the enzymatic aldol addition of propanal to formaldehyde: a first step towards enzymatic synthesis of 3-hydroxybutyric acid. Chem Eng Res Des. 2019;150:140–52. https://doi.org/10.1016/j.cherd.2019.06.025.
Xu G, Dai W, Wang Y, Zhang L, Sun Z, Zhou J, Ni Y. Molecular switch manipulating Prelog priority of an alcohol dehydrogenase toward bulky-bulky ketones. Mol Catal. 2020. https://doi.org/10.1016/j.mcat.2019.110741.
Gong L, Xiu Y, Dong J, Han R, Xu G, Ni Y. Sustainable one-pot chemo-enzymatic synthesis of chiral furan amino acid from biomass via magnetic solid acid and threonine aldolase. Bioresour Technol. 2021;337: 125344. https://doi.org/10.1016/j.biortech.2021.125344.
Kumar P, Meza A, Ellis JM, Carlson GA, Bingman CA, Buller AR. L-Threonine transaldolase activity is enabled by a persistent catalytic intermediate. ACS Chem Biol. 2021;16(1):86–95. https://doi.org/10.1021/acschembio.0c00753.
Dombrowski N, Seitz KW, Teske AP, Baker BJ. Genomic insights into potential interdependencies in microbial hydrocarbon and nutrient cycling in hydrothermal sediments. Microbiome. 2017;5(1):106–106. https://doi.org/10.1186/s40168-017-0322-2.
Zhou Z, Liu Y, Xu W, Pan J, Luo Z-H, Li M. Genome- and community-level interaction insights into carbon utilization and element cycling functions of hydrothermarchaeota in hydrothermal sediment. mSystems. 2020;5(1):e00795-e1719. https://doi.org/10.1128/mSystems.00795-19.
Scott TA, Heine D, Qin Z, Wilkinson B. An L-threonine transaldolase is required for L-threo-beta-hydroxy-alpha-amino acid assembly during obafluorin biosynthesis. Nat Commun. 2017;8:15935. https://doi.org/10.1038/ncomms15935.
Wang S, Deng H. Peculiarities of promiscuous L-threonine transaldolases for enantioselective synthesis of beta-hydroxy-alpha-amino acids. Appl Microbiol Biotechnol. 2021;105(9):3507–20. https://doi.org/10.1007/s00253-021-11288-w.
Wu L, Tong MH, Raab A, Fang Q, Wang S, Kyeremeh K, Yu Y, Deng H. An unusual metal-bound 4-fluorothreonine transaldolase from Streptomyces sp. MA37 catalyses promiscuous transaldol reactions. Appl Microbiol Biotechnol. 2020;104(9):3885–96. https://doi.org/10.1007/s00253-020-10497-z.
Fesko K, Suplatov D, Švedas V. Bioinformatic analysis of the fold type I PLP-dependent enzymes reveals determinants of reaction specificity in L-threonine aldolase from Aeromonas jandaei. FEBS Open Bio. 2018. https://doi.org/10.1002/2211-5463.12441.
Steffen-Munsberg F, Vickers C, Kohls H, Land H, Mallin H, Nobili A, Skalden L, van den Bergh T, Joosten HJ, Berglund P, Höhne M, Bornscheuer UT. Bioinformatic analysis of a PLP-dependent enzyme superfamily suitable for biocatalytic applications. Biotechnol Adv. 2015;33(5):566–604. https://doi.org/10.1016/j.biotechadv.2014.12.012.
Eliot AC, Kirsch JF. Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations. Annu Rev Biochem. 2004;73:383–415. https://doi.org/10.1146/annurev.biochem.73.011303.074021.
Fesko K, Reisinger C, Steinreiber J, Weber H, Schürmann M, Griengl H. Four types of threonine aldolases: similarities and differences in kinetics/thermodynamics. J Mol Catal B. 2008;52–53:19–26. https://doi.org/10.1016/j.molcatb.2007.10.010.
Steinreiber J, Schurmann M, Wolberg M, van Assema F, Reisinger C, Fesko K, Mink D, Griengl H. Overcoming thermodynamic and kinetic limitations of aldolase-catalyzed reactions by applying multienzymatic dynamic kinetic asymmetric transformations. Angew Chem Int Ed Engl. 2007;46(10):1624–6. https://doi.org/10.1002/anie.200604142.
Park J, Lee H-S, Oh J, Joo JC, Yeon YJ. A highly active carboxylic acid reductase from Mycobacterium abscessus for biocatalytic reduction of vanillic acid to vanillin. Biochem Eng J. 2020;161: 107683. https://doi.org/10.1016/j.bej.2020.107683.
He A, Li T, Daniels L, Fotheringham I, Rosazza JP. Nocardia sp. carboxylic acid reductase: cloning, expression, and characterization of a new aldehyde oxidoreductase family. Appl Environ Microbiol. 2004;70(3):1874–81. https://doi.org/10.1128/aem.70.3.1874-1881.2004.
Schwendenwein D, Ressmann AK, Doerr M, Höhne M, Bornscheuer UT, Mihovilovic MD, Rudroff F, Winkler M. Random mutagenesis-driven improvement of carboxylate reductase activity using an amino benzamidoxime-mediated High-throughput assay. Adv Synth Catal. 2019. https://doi.org/10.1002/adsc.201900155.
Thomas A, Cutlan R, Finnigan W, van der Giezen M, Harmer N. Highly thermostable carboxylic acid reductases generated by ancestral sequence reconstruction. Commun Biol. 2019;2:429. https://doi.org/10.1038/s42003-019-0677-y.
Steinreiber J, Fesko K, Reisinger C, Schürmann M, van Assema F, Wolberg M, Mink D, Griengl H. Threonine aldolases—an emerging tool for organic synthesis. Tetrahedron. 2007;63(4):918–26. https://doi.org/10.1016/j.tet.2006.11.035.
Acknowledgements
We are grateful to the National Key Research and Development Program (2021YFC2102700), the National Natural Science Foundation of China (22077054, 22078127), the National First-Class Discipline Program of Light Industry Technology and Engineering (LITE2018-07), and Program of Introducing Talents of Discipline to Universities (111-2-06) for the financial support of this research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiu, Y., Xu, G. & Ni, Y. Multi-enzyme cascade for sustainable synthesis of l-threo-phenylserine by modulating aldehydes inhibition and kinetic/thermodynamic controls. Syst Microbiol and Biomanuf 2, 705–715 (2022). https://doi.org/10.1007/s43393-022-00102-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43393-022-00102-x