Abstract
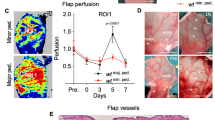
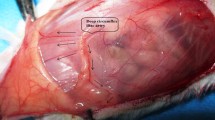
A functional skin-flap model of angiogenesis in the mouse was utilized to investigate ischaemic flap survival/angiogenesis whilst under pharmacological or genetic inhibition of nitric oxide synthase (NOS). In this model, the epigastric artery was cauterized. Following a five-day angiogenic period an abdominal skin-flap supplied by the pre-existing epigastric artery was raised and resutured. After a further six days the outcome was determined by measuring the area of living skin-flap that was sustained by new vessel growth around the cauterized artery. Both pharmacological [S-methyl-isothiourea (SMT) given via i.p. injection for the five-day angiogenic period] and genetic inhibition of NOS 2 (using NOS 2−/− mice) caused a similar and significant fall in flap survival compared to saline-treated wild-type mice (P < 0.05). Delaying pharmacological NOS 2 inhibition for two days post-arterial cauterization increased flap survival in wild-type mice to that of saline-treated controls, whilst treating wild-type mice with SMT for only the first three days of angiogenesis produced a significant decrease in flap survival, similar to that of five-day SMT-treated wild types. Immunoreactivity for NOS 2 in NOS 2−/− knockout mice was absent in both pedicles and skin-flaps, whilst mast cells from both the pedicle and the skin-flap stained positively for NOS 2 in wild-type mice. Vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (FGF 2) immunoreactivity was also strongly evident in mast cells of both wild-type and NOS 2−/− mice at both sites. These results point to a significant role for NOS 2 in promoting wound healing/angiogenesis in its early stages.
Similar content being viewed by others
References
Kane AJ, Barker JE, Mitchell GM et al. Inducible nitric oxide synthase (iNOS) activity promotes ischaemic skin flap survival. Br J Pharmacol 2001; 132 (8): 1631–8.
Stewart AG, Phan LH, Grigoriadis G. Physiological and pathophysiological roles of nitric oxide. Microsurgery 1994; 15 (10): 693–702.
Pipili-Synetos E, Sakkoula E, Haralabopoulos G et al. Evidence that nitric oxide is an endogenous antiangiogenic mediator. Br J Pharmacol 1994; 111: 894–902.
Pipili-Synetos E, Papageorgiou A, Sakkoula E et al. Inhibition of angiogenesis, tumour growth and metastasis by the NO-releasing vasodilators, isosorbide mononitrate and dinitrate. Br J Pharmacol 1995; 116 (2): 1829–34.
Sakkoula E, Pipili-Synetos E, Maragoudakis ME. Involvement of nitric oxide in the inhibition of angiogenesis by interleukin-2. Br J Pharmacol 1997; 122 (5): 793–5.
Ziche M, Morbidelli L, Masini E et al. Nitric oxide promotes DNA synthesis and cyclic GMP formation in endothelial cells from postcapillary venules. Biochem Biophys Res Commun 1993; 192 (3): 1198–203.
Ziche M, Morbidelli L, Parenti A et al. Substance P increases cyclic GMP levels on coronary postcapillary venular endothelial cells. Life Sci 1993; 53 (14): PL229–34.
Gallo O, Masini E, Morbidelli L et al. Role of nitric oxide in angiogenesis and tumor progression in head and neck cancer. J Natl Cancer Inst 1998; 90 (8): 587–96.
Konopka TE, Barker JE, Bamford TL et al. Nitric oxide synthase II gene disruption: Implications for tumor growth and vascular endothelial growth factor production. Cancer Res 2001; 61 (7): 3182–7.
Ziche M, Parenti A, Ledda F et al. Nitric oxide promotes proliferation and plasminogen activator production by coronary venular endothelium through endogenous bFGF. Circ Res 1997; 80 (6): 845–52.
Yamasaki K, Edington HD, McClosky C et al. Reversal of impaired wound repair in iNOS-deficient mice by topical adenoviral-mediated iNOS gene transfer. J Clin Invest 1998; 101 (5): 967–71.
Ruetten H, Thiemermann C. Prevention of the expression of inducible nitric oxide synthase by aminoguanidine or aminoethylisothiourea in macrophages and in the rat. Biochem Biophys Res Commun 1996; 225 (2): 525–30.
Southan GJ, Szabo C, Thiemermann C. Isothioureas: Potent inhibitors of nitric oxide synthases with variable isoform selectivity. Br J Pharmacol 1995; 114 (2): 510–6.
MacMicking JD, Nathan C, Hom G et al. Altered responses to bacterial infection and endotoxic shock in mice lacking inducible nitric oxide synthase. Cell 1995; 81 (4): 641–50.
Nathan C. Inducible nitric oxide synthase: What difference does it make? J Clin Invest 1997; 100 (10): 2417–23.
Reichner JS, Meszaros AJ, Louis CA et al. Molecular and metabolic evidence for the restricted expression of inducible nitric oxide synthase in healing wounds. Am J Pathol 1999; 154 (4): 1097–104.
Frank S, Madlener M, Pfeilschifter J et al. Induction of inducible nitric oxide synthase and its corresponding tetrahydrobiopterincofactor-synthesizing enzyme GTP-cyclohydrolase I during cutaneous wound repair. J Invest Dermatol 1998; 111 (6): 1058–64.
Niwa K, Takizawa S, Kawaguchi C et al. Expression of inducible nitric oxide synthase immunoreactivity in rat brain following chronic hypoxia: Effect of aminoguanidine. Neurosci Lett 1999; 271 (2): 109–12.
Shimizu K, Naito S, Urata Y et al. Inducible nitric oxide synthase is expressed in granuloma pyogenicum. Br J Dermatol 1998; 138 (5): 769–73.
Cartwright JE, Johnstone AP, Whitley GS. Endogenously produced nitric oxide inhibits endothelial cell growth as demonstrated using novel antisense cell lines. Br J Pharmacol 2000; 131 (1): 131–7.
Sennlaub F, Courtois Y, Goureau O. Nitric oxide synthase-II is expressed in severe corneal alkali burns and inhibits neovascularization. Invest Ophthalmol Vis Sci 1999; 40 (12): 2773–9.
Pipili-Synetos E, Kritikou S, Papadimitriou E et al. Nitric oxide synthase expression, enzyme activity and NO production during angiogenesis in the chick chorioallantoic membrane. Br J Pharmacol 2000; 129 (1): 207–13.
Murohara T, Asahara T, Silver M et al. Nitric oxide synthase modulates angiogenesis in response to tissue ischemia. J Clin Invest 1998; 101 (11): 2567–78.
Lee PC, Salyapongse AN, Bragdon GA et al. Impaired wound healing and angiogenesis in eNOS-deficient mice. Am J Physiol 1999; 277 (4 Pt 2): H1600–8.
Smith Jr, RS, Lin KF, Agata J et al. Human endothelial nitric oxide synthase gene delivery promotes angiogenesis in a rat model of hindlimb ischemia. Arterioscler Thromb Vasc Biol 2002; 22 (8): 1279–85.
Ziche M, Morbidelli L, Choudhuri R et al. Nitric oxide synthase lies downstream from vascular endothelial growth factor-induced but not basic fibroblast growth factor-induced angiogenesis. J Clin Invest 1997; 99 (11): 2625–34.
Shukla A, Rasik AM, Shankar R. Nitric oxide inhibits wounds collagen synthesis. Mol Cell Biochem 1999; 200 (1-2): 27–33.
Naslund I, Norrby K. NO and de novo mammalian angiogenesis: Further evidence that NO inhibits bFGF-induced angiogenesis while not influencing VEGF165-induced angiogenesis. Apmis 2000; 108 (1): 29–37.
Fukumura D, Gohongi T, Kadambi A et al. Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factor-induced angiogenesis and vascular permeability. Proc Natl Acad Sci USA 2001; 98 (5): 2604–9.
Papapetropoulos A, Desai KM, Rudic RD et al. Nitric oxide synthase inhibitors attenuate transforming-growth-factor-beta 1-stimulated capillary organization in vitro. Am J Pathol 1997; 150 (5): 1835–44.
Joshi M, Fuller LR, Batchelor GC. L-arginine metabolites regulate DNA synthesis and nitric oxide synthase activity in cultured human dermal microvascular endothelial cells-potential positive and negative regulators of angiogenesis derived from L-arginine. Cancer Invest 1999; 17 (4): 235–44.
Messina A, Knight KR, Dowsing BJ et al. Localization of inducible nitric oxide synthase to mast cells during ischemia/reperfusion injury of skeletal muscle. Lab Invest 2000; 80 (3): 423–31.
Bidri M, Feger F, Varadaradjalou S et al. Mast cells as a source and target for nitric oxide. Int Immunopharmacol 2001; 1 (8): 1543–58.
Grutzkau A, Kruger-Krasagakes S, Baumeister H et al. Synthesis, storage, and release of vascular endothelial growth factor/vascular permeability factor (VEGF/VPF) by human mast cells: Implications for the biological significance of VEGF206. Mol Biol Cell 1998; 9 (4): 875–84.
Qu Z, Liebler JM, Powers MR et al. Mast cells are a major source of basic fibroblast growth factor in chronic inflammation and cutaneous hemangioma. Am J Pathol 1995; 147 (3): 564–73.
Barker JE, Stewart AG. Differing effects of nitric oxide compared with peroxynitrite on mast cell degranulation in vitro. Br J Pharmacol 1998; 123: 305P.
Parenti A, Morbidelli L, Cui XL et al. Nitric oxide is an upstream signal of vascular endothelial growth factor-induced extracellular signal-regulated kinase 1/2 activation in postcapillary endothelium. J Biol Chem 1998; 273 (7): 4220–6.
Bouloumie A, Schini-Kerth VB, Busse R. Vascular endothelial growth factor up-regulates nitric oxide synthase expression in endothelial cells. Cardiovasc Res 1999; 41 (3): 773–80.
Kroll J, Waltenberger J. VEGF-A induces expression of eNOS and iNOS in endothelial cells via VEGF receptor-2 (KDR). Biochem Biophys Res Commun 1998; 252 (3): 743–6.
Kostyk SK, Kourembanas S, Wheeler EL et al. Basic fibroblast growth factor increases nitric oxide synthase production in bovine endothelial cells. Am J Physiol 1995; 269 (5 Pt 2): H1583–9.
Babaei S, Teichert-Kuliszewska K, Monge JC et al. Role of nitric oxide in the angiogenic response in vitro to basic fibroblast growth factor. Circ Res 1998; 82 (9): 1007–15.
Bussolati B, Dunk C, Grohman M et al. Vascular endothelial growth factor receptor-1 modulates vascular endothelial growth factor-mediated angiogenesis via nitric oxide. Am J Pathol 2001; 159 (3): 993–1008.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Furuta, S., Vadiveloo, P., Romeo-Meeuw, R. et al. Early Inducible Nitric Oxide Synthase 2 (NOS 2) Activity Enhances Ischaemic Skin Flap Survival. Angiogenesis 7, 33–43 (2004). https://doi.org/10.1023/B:AGEN.0000037328.86329.8b
Issue Date:
DOI: https://doi.org/10.1023/B:AGEN.0000037328.86329.8b