Abstract
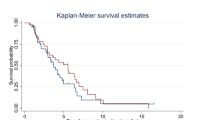
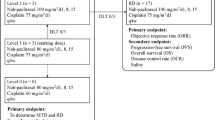
Introduction: The poor prognosis of non small cell lung cancer (NSCLC), as well as the significant toxicity from many conventional cytotoxic regimens warrants the investigation of combinations of new active agents for treatment. This is a phase I–II (dose-finding, efficacy, and toxicity) study of docetaxel + gemcitabine in patients with stage IIIB-IV NSCLC without prior systemic therapy. Patients were treated in cohorts of 3 with alternating increasing doses of docetaxel and gemcitabine at each level. Patients: Fifty patients were entered, of which 49 were eligible including 28 males and 21 females; 15 stage IIIB and 34 stage IV; median age 57 yrs (35–74). Results: The Maximum Tolerated Dose (MTD) was docetaxel 60 mg/m2 day 1 and gemcitabine 750 mg/m2 d1 & 8, every 21 days. The overall response rate is 20%. Eight patients are not formally assessable for response due to early discontinuation or loss to follow-up and are considered to have progressive disease. The median time to progression (TTP) is 3.5 months (1–25), with 11 pts with at least 7 months TTP. There were 10 pts (20%) with a partial response (PR); 18 (37%) maintained stable disease; 21 (43%) had progressive disease (PD) or were not assessable. Toxicity: Forty-nine patients are evaluable for assessment for toxicity: Grade (Gr) 3/4 toxicity was documented thus: 14 with neutropenia, 1 with anemia, 1 with nausea, 2 liver function, 2 dyspnea, 2 fatigue, 1 allergy, 1 neurologic. Conclusion: This regimen is well tolerated and results in phase I–II testing in this patient population warrant further consideration of the study of docetaxel + gemcitabine for advanced NSCLC.
Similar content being viewed by others
References
American Cancer Society Cancer Facts and Figures, 2003
Minna JD, Pass H, Glatstein E, Ihde DC: Cancer of the lung. In Vincent T. De Vita Jr. et al. (eds), Cancer: Principles & Practice: of Oncology, 3rd edition, JB. Lippincott Co., Philadelphia, 1989
Gralla RJ, Kris MG: Chemotherapy in non-small cell lung cancer: Results of recent trials. Semin Oncol 15(suppl 4): 2–5, 1988
Casper ES, Gralla RJ, Kelson DP, Citovic E, Golbey RB: Phase II study of high dose cis-diachlorodiammine-platinum (II) in the treatment of non-small cell lung cancer. Cancer Treat Rep 63: 2107–2109, 1979
De Jager R, Longeval I, Klatersky J: High dose cisplatin with fluid and mannitol induced diuresis in advanced lung cancer: A phase II clinical trial of EORTC lung cancer working party (Belgium). Cancer Treat Rep 64: 1341–1346, 1980
Furnas BE, Williams SD, Einhorn LH, Cobleigh MA: Vindesine, an effective agent in the treatment of non-small cell lung cancer. Cancer Treat Rep 66: 1709–1711, 1982
Luedke SL, Luedke DW, Petruska P, Broun GO, Reed G, Leavitt J: Vindesine (VDS) monochemotherapy for non small cell lung cancer: A report of 45 cases. Cancer Treat Rep 66: 1409–1411, 1982
Gralla RJ, Raphael BG, Golbey RB, Young CW: Phase II evaluation of vindesine in patients with non-small cell carcinoma of the lung. Cancer Treat Rep 63: 1343–1346, 1979
Gralla RJ, Casper ES, Kelsen DP, Braun DW, Dukeman ME et al: Cisplatin and vindesine in combination chemotherapy for advanced carcinoma of the lung: A randomized trial investigating two dosage schedules. Ann Intern Med 95: 414–420, 1981
Woodcock TM, Blumenreich MS, Richman SP, Kubota TT, Gentile PS, Allegra JC: Combination chemotherapy with cis-diammine-dichloroplatinum and vinblastine in advanced non small cell lung cancer. J Clin Oncol 1: 247–250, 1983
Spain RC: Neoadjuvant mitomycin C, cisplatin, and infusion vinblastine in locally and regionally advanced non-small cell lung cancer: Problems and progress from the perspective of long term follow-up. Semin Oncol 15(Suppl 4): 6–15, 1988
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J, Zhu J, Johnson DH: Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. NEJM 346: 92–98, 2002
Dorr RT, Von Hoff DD: Cancer chemotherapy Handbook, 2nd edition. Appleton & Lange, Norwalk CT, 1994, pp. 259–267
Bruno R, Sanderink GJ: Pharmacokinetics and metabolism of docetaxel (Taxotere). Cancer Surveys: Pharmacokinet Cancer Chemother 17: 305–313, 1993
Ringel I, Horwitz SB: Studies with RP 56976 (Taxotere): A semisynthetic analogue of taxol. J Natl Cancer Inst 83: 288–291, 1991
Bissery MC, Guénard D, Guéritte-Voegelein F, Lavelle F: Experimental antitumor activity of taxotere (RP 56976, NCS 628503), a taxol analogue. Cancer Res 51: 4845–4852, 1991
Verweij J, Clavel M, Chevalier B: Paclitaxel (Taxol®) and docetaxel (Taxotere®): Not simply two of a kind. Annals of Oncology 5: 495–505, 1994
Extra JM, Rousseau F, Bruno R et al: Phase I and pharmacokinetic study of Taxotere (RP 56976, NSC 628503) given as short I.V. infusion, every 21 days. Cancer Res 53: 1037–102, 1993
Riou JF, Petitgenet O, Combeau C et al: Cellular uptake and efflux of docetaxel (Taxotere) and paclitaxel (Taxol) in P388 cell line. Pro Am Assoc Can Res 35: 385, 1994
Francis PA, Rigas JR, Kris MG, Pisters MG, Orazem JP, Woolley KJ, Heelan RT: Phase II trial of docetaxel in patients with stage III and IV non-small cell lung cancer. J Clin Oncol 12: 1232–1237, 1994
Fossella FV, Lee JS, Murphy WK, Lippman SM, Calayag M, Pang A, Chasen M, Shin DM, Glisson B, Benner S: Phase II study of docetaxel for recurrent or metastatic non-small cell lung cancer. J Clin Oncol 12: 1238–1244, 1994
Burris H, Eckhardt J, Fields S, Rodriguez G, Smith L, Thurman A, Peacock N, Juhn J, Hodges S, Bellet R, Bayssas M, LeBail N, Von Hoff D: Phase II trial of taxotere in patients with non-small cell lung cancer. Proc Am Soc Clin Oncol 12: 335, 1993
Le Chevalier T: Single agent activity of Gemcitabine in advanced non-small cell lung cancer. Sem Oncol 1996; 23(Suppl 10): 36–42
Kaplan E, Meier P: Nonparametric estimation from incomplete observations. J Am Stat Assoc 53: 457–481, 1958
Clinical practice guidelines for the treatment of unresectable non-small-cell lung cancer. J Clin Oncol 15: 2996–3018, 1997
Socinski MA, Morris DE, Masters GA, Lilenbaum R: Chemotherapeutic management of stage IV non-small cell lung cancer. Chest 123: 226S–243S, 2003
Georgoulias V, Kouroussis C, Androulakis N, Kakolyris S, Dimopoulos M-A, Papadakis E, Bouros D, Apostolopoulou F, Papadimitriou C, Agelidou A, Hatzakis K, Kalbakis K, Kotsakis A, Vardakis N, Vlachonicolis J: Front-line treatment of advanced non-small-cell lung cancer with docetaxel and gemcitabine: A multicenter phase II trial. J Clin Oncol 17(3): 914–920, 1999
Georgoulias V, Papadakis E, Alexopoulos A, Tsiafaki X, Veslemes M, Palamidas Ph, Vlachonikolis I: Platinum-based and non-platinum based chemotherapy in advanced non-small cell lung cancer: A randomized multicentre trial. Lancet 357: 1478–1484, 2001
Kosmidis P, Mylonakis N, Nicolaides C, Kalophonos C, Samantas E, Boukovinas J, Fountzilas G, Skarlos D, Econompoulos T, Tsavdaridis D, Papakostas P, Bacoyianis C, Dimopoulos M: Paclitaxel plus carboplatin versus gemcitabine plus paclitaxel in advanced non-small-cell lung cancer: A phase III randomized trial. J Clin Oncol 17: 3578–3585, 2002
Lorusso V, Carpagnano F, Frasci G, Panza N, Di Rienzo G, Cisternino ML, Napoli G, Orlando S, Cinieri S, Brunetti C, Palazzo S, De Lena M: Phase I/II study of gemcitabine plus vinorelbine as first-line chemotherapy of non-small-cell lung cancer. J Clin Oncol 18: 405–411, 2000
Lilenbaum R, Cano R, Schwartz M, Siegel L, Lutzky J, Lewis M, Krill E, Barreras L, Davila E: Gemcitabine and vinorelbine in advanced Nonsmall cell lung carcinoma. Cancer 88: 557–562, 2000
Bajetta E, Chiaria Stani S, De Candis D, Bidoli P, Mariani L, Zilembo N, Pozzi P, Procopio G: Gemcitabine plus vinorelbine as first-line chemotherapy in advanced nonsmall cell lung carcinoma: A phase II trial. Cancer 89: 763–768, 2000
Gridelli C, Frontini L, Peronne F, Gallo C, Gulisano M, Cigolari S, Castiblione F, Robbiat SF, Gasparini G, Ianniello GP, Farris A, Locatelli MC, Felletti R, Piazza E: Gemcitabine plus vinorelbine in advanced non-small cell lung cancer: A phase II study of three different doses. Br J Cancer 83: 707–714, 2000
Beretta GD, Michetti D, Belometti MO et al: Gemcitabine plus vinorelbine in elderly or unfit patients with non-small cell lung cancer. Br J Cancer 83: 573–576, 2000
Frasci G, Lorusso V, Panza N, Comella P, Nicolella G, Bianco A, De Cataldis G, Iannelli A, Bilancia D, Belli M, Massidda B, Piantedosi F, Comella G, De Lena M: Gemcitabine plus vinorelbine versus vinorelbine alone in elderly patients with advanced non-small-cell lung cancer. J Clin Oncol 18: 2529–2536, 2000
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Israel, V., Tagawa, S.T., Snyder, T. et al. Phase I/II trial of gemcitabine plus docetaxel in advanced non small cell lung cancer (NSCLC). Invest New Drugs 22, 291–297 (2004). https://doi.org/10.1023/B:DRUG.0000026255.34988.96
Issue Date:
DOI: https://doi.org/10.1023/B:DRUG.0000026255.34988.96