Abstract
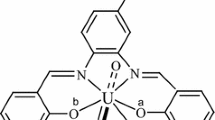
A new Schiff base complex of [UO2L(C2H5OH)(H2O)3]NO3 (HL=4-(2-hydroxy-naphthalen-1-ylmethyleneamino)-antipyrin) composition has been synthesized and characterized by elemental analysis, IR, UV and molar conductance. The thermodecomposition kinetics of the complex was investigated under non-isothermal conditions. TG and DTG curves indicate that the complex decomposes in five steps. The kinetics of the first step was calculated by a combination of Achar differential and Coats-Redfern integral methods. The kinetic equation was determined.
Similar content being viewed by others
References
G. Preetam, Synth. React. Inorg. Met-Org. Chem., 31 (2001) 247.
B. Caifeng, F. Yuhua, S. Guoxin, C. Jun, J. Radioanal. Nucl. Chem., 246 (2000) 221.
F. Yuhua, B. Caifeng, L. Jiying, J. Radioanal. Nucl. Chem., 254 (2002) 641.
R. K. Agarwal, K. Arora, P. Dutt, Synth. React. Inorg. Met-Org. Chem., 24 (1994) 301.
E. M. Nour, A. M. Al-Kority, S. A. Sadeek, S. M. Teleb, Synth. React. Inorg. Met-Org. Chem., 23 (1993) 39.
W. J. Geary, Coord. Chem. Rev., 7 (1971) 81.
R. E. Hester, E. M. Nour, J. Raman Spectrosc., 11 (1981) 49.
L. Yuzheng, Thermal Analysis, 1 st ed., Qinghua University Press, Beijing, 1987, p. 94.
B. N. Achar, Proc. Intern. Clay Conference, Book 1, Jerusalem, 1966, p. 67.
A. W. Coats, J. P. Redfern, Nature (London), 201 (1964) 68.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caifeng, B., Yuhua, F. Synthesis and thermodecomposition kinetics of the UO2 (II) complex with 4-(2-hydroxy-naphthalen-1-ylmethyleneamino)-antipyrin. Journal of Radioanalytical and Nuclear Chemistry 262, 497–500 (2004). https://doi.org/10.1023/B:JRNC.0000046785.85970.af
Issue Date:
DOI: https://doi.org/10.1023/B:JRNC.0000046785.85970.af