Abstract
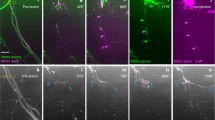
Schwann cells and axons labeled by transgene-encoded, fluorescent proteins can be repeatedly imaged in living mice to observe the reinnervation of neuromuscular junctions. Axons typically return to denervated junctions by growing along Schwann cells contained in the old nerve sheaths or “Schwann cell tubes”. These axons then commonly “escape” the synaptic sites by growing along the Schwann cell processes extended during the period of denervation. These “escaped fibers” grow to innervate adjacent synaptic sites along Schwann cells bridging these sites. Within the synaptic site, Schwann cells, originally positioned above the synaptic site continue to cover the acetylcholine receptors (AChRs) immediately following denervation, but gradually vacate portions of this site. When regenerating axons return, they first deploy along the Schwann cells and ignore sites of AChRs vacated by Schwann cells. In many cases these vacated sites are never reinnervated and are ultimately lost. Following partial denervation, Schwann cells grow in an apparently tropic fashion from denervated to nearby innervated synaptic sites and serve as the substrates for nerve sprouting. These experiments show that Schwann cells provide pathways that stimulate axon growth and insure the rapid reinnervation of denervated or partially denervated muscles.
Similar content being viewed by others
References
ANTON, E. S., WESKAMP, G., REICHARDT, L. F. & MATTHEW, W. D. (1994) Nerve growth factor and its low-affinity receptor promote Schwann cell migration. Proc. Natl. Acad. Sci. USA 91, 279–2799.
ASTROW, S. H., QIANG, H. & KO, C. P. (1998) Perisynaptic Schwann cells at neuromuscular junctions revealed by a novel monoclonal antibody. J. Neurocytol. 27, 66–681.
ASTROW, S. H., SON, Y.-J. & THOMPSON, W. J. (1994) Differential neural regulation of a neuromuscular junction-associated antigen in muscle fibers and Schwann cells. J. Neurobiol. 25, 93–952.
BALICE-GORDON, R. J. & LICHTMAN, J. W. (1994) Long-term synapse loss induced by focal blockade of postsynaptic receptors. Nature 372, 51–524.
BIRKS, R., HUXLEY, H. E. & KATZ, B. (1960a) The fine structure of the neuromuscular junction of the frog. J. Physiol. 150, 13–144.
BIRKS, R., KATZ, B. & MILEDI, R. (1960b) Physiological and structural changes at the amphibian myoneural junction, in the course of nerve degeneration. J. Physiol. 150, 14–168.
BIXBY, J. L., LILIEN, J. & REICHARDT, L. F. (1988) Identification of the major proteins that promote neuronal process outgrowth on Schwann cells in vitro. J. Cell Biol. 107, 35–361.
BRITSCH, S., GOERICH, D. E., RIETHMACHER, D., PEIRANO, R. I., ROSSNER, M., NAVE, K. A., BIRCHMEIER, C. & WEGNER, M. (2001) The transcription factor Sox10 is a key regulator of peripheral glial development. Genes Dev. 15, 6–78.
BROCKES, J. P. (1984) Assays for cholinergic properties in cultured rat Schwann cells. Proc.Roy. Soc. Lond. B 222, 12–134.
BROOKE, M. H., WILLIAMSON, E. & KAISER, K. K. (1971) The behavior of four fiber types in developing and reinnervated muscle. Arch. Neurol. 25, 36–366.
BROWN, M. C., HOLLAND, R. L. & HOPKINS, W. G. (1981) Motor nerve sprouting. Ann. Rev. Neurosci. 4, 1–42.
CASTETS, F., GRIFFIN, W. S. T., MARKS, A. & VAN ELDIK, L. J. (1997) Transcriptional regulation of the human S100? gene. Mol. Brain Res. 46, 20–216.
COUTEAUX, R. (1960) Motor end-plate structure. In The Structure and Function of Muscle (edited by BOURNE, G. H.) pp. 33–380. New York: Academic Press.
DENNIS, M. J. & MILEDI, R. (1974) Electrically induced release of acetylcholine from denervated Schwann cells. J. Physiol. 237, 43–452.
DONG, Z., BRENNAN, A., LIU, N., YARDEN, Y., LEFKOWITZ, G., MIRSKY, R. & JESSEN, K. R. (1995) Neu differentiation factor is a neuron-glia signal and regulates survival, proliferation, and maturation of rat Schwann cell precursors. Neuron 15, 58–596.
FALLON, J. R. (1985) Neurite guidance by non-neuronal cells in culture: Preferential outgrowth of peripheral neurites on glial as compared to nonglial cell surfaces. J. Neurosci. 5, 316–3177.
FENG, G., MELLOR, R. H., BERNSTEIN, M., KELLERPECK, C., NGUYEN, Q. T., WALLACE, M., NERBONNE, J. M., LICHTMAN, J. W. & SANES, J. R. (2000) Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28, 4–51.
GUTMANN, E. & YOUNG, J. Z. (1944) The re-innervation of muscle after various periods of atrophy. J. Anat. 78, 1–43.
HENDERSON, C. E., PHILLIPS, H. S., POLLOCK, R. A., DAVIES, A. M., LEMEULLE, C., ARMANINI, M., SIMPSON, L. C., MOFFET, B., VANDLEN, R. A., KOLIATSOS, V. E. & ROSENTHAL, A. (1994) GDNF:Apotent survival factor for motoneurons present in peripheral nerve and muscle. Science 266, 106– 1064.
HOLMES, W. & YOUNG, J. Z. (1942) Nerve regeneration after immediate and delayed suture. J. Anat. 77, 6–96.
JAHROMI, B. S., ROBITAILLE, R. & CHARLTON, M. P. (1992) Transmitter release increases intracellular calcium in perisynaptic Schwann cells in situ. Neuron 8, 106–1077.
JIRMANOVÁ, I. (1975) Ultrastructure of motor end-plates during pharmacologically-induced degeneration and subsequent regeneration of skeletal muscle. J. Neurocytol. 4, 14–155.
KANG, H., LUBISCHER, J. L., NEWMAN, C. S., THOMPSON, W. J. & KRIEG, P. A. (2000) In vivo imaging of terminal Schwann cells using GFP expressing transgenic mice. Soc. Neurosci. Abs. 26, 703.2.
KANG, H. & THOMPSON, W. J. (2002) Schwann cell processes guide axons reinnervating the neuromuscular junction. Soc. Neurosci. Abs. 28, 234.16.
KO, C.-P. & CHEN, L. (1996) Synaptic remodeling revealed by repeated in vivo observations and electron microscopy of identified frog neuromuscular junctions. J. Neurosci. 16, 178–1790.
KOIRALA, S., QIANG, H. & KO, C. P. (2000) Reciprocal interactions between perisynaptic Schwann cells and regenerating nerve terminals at the frog neuromuscular junction. J. Neurobiol. 44, 34–360.
KUFFLER, S. W. & NICHOLLS, J. G. (1966) The physiology of neuroglial cells. Ergeb. Physiol. 57, –90.
LICHTMAN, J. W., MAGRASSI, L. & PURVES, D. (1987) Visualization of neuromuscular junctions over periods of several months in living mice. J. Neurosci. 7, 121–1222.
LOVE, F. M. & THOMPSON, W. J. (1999) Glial cells promote muscle reinnervation by responding to activity-dependent postsynaptic signals. J. Neurosci. 19, 1039–10396.
LUBISCHER, J. L. & THOMPSON, W. J. (1999) Neonatal partial denervation results in nodal but not terminal sprouting and a decrease in efficacy of remaining neuromuscular junctions in rat soleus muscle. J. Neurosci. 19, 893–8944.
MACLEOD, G. T., DICKENS, P. A. & BENNETT, M. R. (2001) Formation and function of synapses with respect to Schwann cells at the end of motor nerve terminal branches on mature amphibian (Bufo marinus) muscle. J. Neurosci. 21, 238–2392.
MILEDI, R. & SLATER, C. R. (1968) Electrophysiology and electron-microscopy of rat neuromuscular junctions after nerve degeneration. Proc. Roy. Soc. Lond. B. 169, 28– 306.
MILEDI, R. & SLATER, C. R. (1970) On the degeneration of rat neuromuscular junctions after nerve section. J. Physiol. 207, 50–528.
MORRIS, J. K., LIN, W., HAUSER, C., MARCHUK, Y., GETMAN, D. & LEE, K. F. (1999) Rescue of the cardiac defect in ErbB2 mutant mice reveals essential roles of ErbB2 in peripheral nervous system development. Neuron 23, 27–283.
NEWMAN, E. A. (1984) Regional specialization of retinal glial cell membrane. Nature 309, 15–157.
NGUYEN, Q. T., SANES, J. R. & LICHTMAN, J. W. (2002) Pre-existing pathways promote precise projection patterns. Nat. Neurosci. 5, 86–867.
O'MALLEY, J. P., WARAN, M. T. & BALICE-GORDON, R. J. (1999) In vivo observations of terminal Schwann cells at normal, denervated, and reinnervated mouse neuromuscular junctions. J. Neurobiol. 38, 27–286.
PALADE, G. E. (1954) Electron microscope observations of interneuronal and neuromuscular synapses. Anat. Rec. 118, 33–336.
PENG, H. B., YANG, J. F., DAI, Z., LEE, C. W., HUNG, H. W., FENG, Z. H. & KO, C. P. (2003) Differential effects of neurotrophins and Schwann cell-derived signals on neuronal survival/growth and synaptogenesis. J. Neurosci. 23, 505–5060.
PETERS, A. & MUIR, A. R. (1959) The relationship between axons and Schwann cells during development of peripheral nerves in the rat. J. Exp. Physiol. 44, 11–130.
RAMON Y CAJAL, S. (1928) Degeneration and Regeneration of the Nervous System. New York: Hafner, translated by R. M., 1959.
RANVIER, L. (1878) Le¸cons Sur l'histologies du Syst´eme Nerveux. Volume 2, Paris: Savy.
REIST, N. E. & SMITH, S. J. (1992) Neurally evoked calcium transients in terminal Schwann cells at the neuromuscular junction. Proc. Natl. Acad. Sci. USA 89, 762– 7629.
REYNOLDS, M. L. & WOOLF, C. J. (1992) Terminal Schwann cells elaborate extensive processes following denervation of the motor endplate. J. Neurocytol. 21, 5–66.
RICH, M. M. & LICHTMAN, J. W. (1989) In vivo visualization of pre-and postsynaptic changes during synapse elimination in reinnervated mouse muscle. J. Neurosci. 9, 178–1805.
RIETHMACHER, D., SONNENBERG-RIETHMACHER, E., BRINKMAN, V., YAMAAI, T., LEWIN, G. R. & BIRCHMEIER, C. (1997) Severe neuropathies in mice with targeted mutations in the ErbB3 receptor. Nature 389, 72–730.
ROBERTSON, J. D. (1956) The ultrastructure of a reptilian myoneural junction. J. Biophys. Biochem. Cytol. 2, 38–394.
ROBITAILLE, R. (1995) Purinergic receptors and their activation by endogenous purines at perisynaptic glial cells of the frog neuromuscular junction. J. Neurosci. 15, 712–7131.
ROBITAILLE, R. (1998) Modulation of synaptic efficacy and synaptic depression by glial cells at the frog neuromuscular junction. Neuron 21, 84–855.
ROCHEL, S. & ROBBINS, N. (1988) Effect of partial denervation and terminal field expansion on neuromuscular transmitter release and nerve terminal structure. J. Neurosci. 8, 33–338.
ROCHON, D., ROUSSE, I. & ROBITAILLE, R. (2001) Synapse-glia interactions at the mammalian neuromuscular junction. J. Neurosci. 21, 381–3829.
SCHERER, S. S. & SALZER, J. L. (1996) Axon-Schwann cell interactions during peripheral nerve degeneration and regeneration. In Glial Cell Development (edited by JESSEN, K. R. & RICHARDSON, W. D.) pp. 16–196. Oxford: BIOS Scientific Publishers.
SCHWANN, T. (1839) Microscopical Researches Into the Accordance in the Structure and Growth of Animals and Plants. London: Sydenham Society.
SLACK, J. R. & HOPKINS, W. G. (1982) Neuromuscular transmission at terminals of sprouted mammalian motor neurones. Brain Res. 237, 12–135.
SON, Y.-J. & THOMPSON, W. J. (1995a) Schwann cell processes guide regeneration of peripheral axons. Neuron 14, 12–132.
SON, Y.-J. & THOMPSON, W. J. (1995b) Nerve sprouting in muscle is induced and guided by processes extended by Schwann cells. Neuron 14, 13–141.
SPEIDEL, C. C. (1932) Studies of living nerves. I. The movements of individual sheath cells and nerve sprouts correlated with the process of myelin-sheath formation in amphibian larvae. J. Exp. Zool. 61, 27–331.
TELLO, F. (1907) Degeneration et regeneration des plagues motrices. Travaux du Laboratorire de Recherches Biologiques de l'Universit'e de Madrid 5, 11–149.
THOMPSON, W. (1978) Reinnervation of partially denervated rat soleus muscle. Acta Physiol. Scand. 103, 8–91.
TIAN, L. & THOMPSON, W. J. (2000) Neuromuscular synaptic instability is produced by long-term partial denervation in adult rat muscles. Soc. Neurosci. Abs. 26, 703.3.
TIAN, L. & THOMPSON, W. J. (2002) Schwann cells induce and guide nerve growth during sprouting in reinnervated soleus muscles in vivo. Soc. Neurosci. Abs. 28, 234.14.
TRACHTENBERG, J. T. & THOMPSON, W. J. (1996) Schwann cell apoptosis at developing neuromuscular junctions is regulated by glial growth factor. Nature 379, 17–177.
TRACHTENBERG, J. T. & THOMPSON, W. J. (1997) Nerve terminal withdrawal from rat neuromuscular junctions induced by neuregulin and Schwann cells. J. Neurosci. 17, 624–6255.
VERDI, J. M., GROVES, A. K., FARIÑAS, I., JONES, K., MARCHIONNI, M. A., REICHARDT, L. F. & ANDERSON, D. J. (1996) A reciprocal cell-cell interaction mediated by NT-3 and neuregulins controls the early survival and development of sympathetic neuroblasts. Neuron 16, 51–527.
WALSH, M. K. & LICHTMAN, J. W. (2003) In vivo timelapse imaging of synaptic takeover associated with naturally occurring synapse elimination. Neuron 37, 6– 73.
WOLPOWITZ, D., MASON, T. B., DIETRICH, P., MENDELSOHN, M., TALMAGE, D. A. & ROLE, L. W. (2000) Cysteine-rich domain isoforms of the neuregulin-1 gene are required for maintenance of peripheral synapses. Neuron 25, 7–91.
YOUNG, J. Z. (1942) The functional repair of nervous tissue. Physiol. Rev. 22, 31–374.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kang, H., Tian, L. & Thompson, W. Terminal Schwann cells guide the reinnervation of muscle after nerve injury. J Neurocytol 32, 975–985 (2003). https://doi.org/10.1023/B:NEUR.0000020636.27222.2d
Issue Date:
DOI: https://doi.org/10.1023/B:NEUR.0000020636.27222.2d