Abstract
Purpose. We have previously demonstrated that celecoxib, a selective COX-2 inhibitor, reaches the retina following repeated oral administrations and inhibits diabetes-induced vascular endothelial growth factor (VEGF) mRNA expression and vascular leakage in a rat model. The aim of this study was to quantify the relative retinal bioavailability of celecoxib from the subconjunctival route compared to a systemic route.
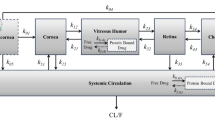
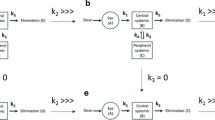
Methods. The plasma and ocular tissue distribution of celecoxib was determined in male Sprague-Dawley rats following subconjunctival and intraperitoneal administrations of drug suspension at a dose of 3 mg/rat. The animals were sacrificed at 0.5, 1, 2, 3, 4, 8, and 12 h post-dosing, the blood was collected, and the eyes were enucleated and frozen. The plasma, sclera, retina, vitreous, lens, and the cornea were isolated and celecoxib levels were determined using an HPLC method. The tissue exposure of the drug was measured as the area under the curve (AUC0-≈) of the concentration vs. time profiles. The relative bioavailability was estimated as the AUC0-≈ ratio between subconjunctival and intraperitoneal groups.
Results. For the subconjunctivally dosed (ipsilateral) eye, the AUC0-≈ ratios between subconjunctival and intraperitoneal groups were 0.8 ± 0.1, 53 ± 4, 54 ± 8, 145 ± 21, 61 ± 16, and 52 ± 6 for plasma, sclera, retina, vitreous, lens, and cornea, respectively. For the contralateral ocular tissues, the AUC0-≈ ratios were 1.2 ± 0.3, 1.1 ± 0.3, 1.1 ± 0.4, 1.0 ± 0.3, and 1.2 ± 0.3 in the sclera, retina, vitreous, lens, and the cornea, respectively, between the subconjunctival and the intraperitoneal groups. Assuming that the drug AUCs in contralateral eye were equal to the systemic pathway contribution to AUCs in the ipsilateral eye, the percent contribution of local pathways as opposed to systemic circulation for celecoxib delivery to the ipsilateral eye tissues was estimated to be 98% or greater.
Conclusions. The retinal delivery of celecoxib was substantially higher following subconjunctival administration compared to the intraperitoneal route. The transscleral pathway almost completely accounts for the retinal celecoxib delivery following subconjunctival administration.
Similar content being viewed by others
REFERENCES
S. P. Ayalasomayajula and U. B. Kompella. Celecoxib, a selective cyclooxygenase-2 inhibitor, inhibits retinal vascular endothelial growth factor expression and vascular leakage in a streptozoto-cin-induced diabetic rat model. Eur. J. Pharmacol. 458:283–289 (2003).
L. P. Aiello, R. L. Avery, P. G. Arrigg, B. A. Keyt, H. D. Jampel, S. T. Shah, L. R. Pasquale, H. Thieme, M. A. Iwamoto, J. E. Park, H. V. Nguyen, L. M. Aiello, N. Ferrara, G. L. King. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N. Engl. J. Med. 331: 1480–1487 (1994).
A. P. Adamis, J. W. Miller, M. T. Bernal, D. J. D'Amico, J. Folkman, T. K. Yeo, and K. T. Yeo. Increased vascular endothe-lial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am. J. Ophthalmol. 118:445–450 (1994).
R. P. Danis, T. A. Ciulla, M. Criswell, and L. Pratt. Anti-angiogenic therapy of proliferative diabetic retinopathy. Expert Opin. Pharmacother.2:395–407 (2001).
N. Patel, L. Sun, D. Moshinsky, H. Chen, K. M. Leahy, P. Le, K. G. Moss, X. Wang, A. Rice, D. Tam, A. D. Laird, X. Yu, Q. Zhang, C. Tang, G. McMahon, and A. Howlett. A selective and oral small molecule inhibitor of vascular epithelial growth factor receptor (VEGFR)-2 and VEGFR-1 inhibits neovascularization and vascular permeability. J. Pharmacol. Exp. Ther. 306:838–845 (2003).
H. Ozaki, M. S. Seo, K. Ozaki, H. Yamada, E. Yamada, N. Oka-moto, F. Hofmann, J. M. Wood, and P. A. Campochiaro. Block-ade of vascular endothelial cell growth factor receptor signaling is sufficient to completely prevent retinal neovascularization. Am. J. Pathol. 156:697–707 (2000).
L. P. Aiello and J. S. Wong. Role of vascular endothelial growth factor in diabetic vascular complications. Kidney Int. Suppl. 77:S113–S119 (2000).
S. H. Lee, E. Soyoola, P. Chanmugam, S. Hart, W. Sun, H. Zhong, S. Liou, D. Simmons, and D. Hwang. Selective expression of mitogen-inducible cyclooxygenase in macrophages stimulated with lipopolysaccharide. J. Biol. Chem. 267:25934–25938 (1992).
X. H. Liu, A. Kirschenbaum, M. Lu, S. Yao, A. Dosoretz, J. F. Holland, and A. C. Levine. Prostaglandin E2 induces hypoxia-inducible factor-1alpha stabilization and nuclear localization in a human prostate cancer cell line. J. Biol. Chem. 277:50081–50086 (2002).
J. Pe'er, D. Shweiki, A. Itin, I. Hemo, H. Gnessin, and E. Keshet. Hypoxia-induced expression of vascular endothelial growth fac-tor by retinal cells is a common factor in neovascularizing ocular diseases. Lab. Invest. 72:638–645 (1995).
P. Needleman and P. C. Isakson. The discovery and function of COX-2. J. Rheumatol. 24:6–8 (1997).
T. Okamoto and O. Hino. Expression of cyclooxygenase-1 and-2 mRNA in rat tissues: tissue-specific difference in the expression of the basal level of mRNA. Int. J. Mol. Med. 6:455–457 (2000).
S. R. Ahmad, C. Kortepeter, A. Brinker, M. Chen and J. Beitz. Renal failure associated with the use of celecoxib and rofecoxib. Drug Saf. 25:537–544 (2002).
M. Epstein. Cardiovascular and renal effects of COX-2-specific inhibitors: recent insights and evolving clinical implications. Am. J. Ther. 8:81–83 (2001).
G. A. Peyman and G. J. Ganiban. Delivery systems for intraoc-ular routes. Adv. Drug Deliv. Rev. 16:107–123 (1995).
C. P. S. Cheruvu, S. P. Ayalasomayajula, and U. B. Kompella. Retinal delivery of sodium fluorescein, budesonide, and cele-coxib following subconjunctival injection. Drug Delivery Tech-nology 3:62–67 (2003).
G. Gudauskas, C. Kumi, C. Dedhar, N. Bussanich and J. Root-man. Ocular pharmacokinetics of subconjunctivally versus intra-venously administered 6-mercaptopurine. Can. J. Ophthalmol. 20:110–113 (1985).
G. S. Kalsi, H. K. Silver and J. Rootman. Ocular pharmacokinet-ics of dacarbazine following subconjunctival versus intravenous administration in the rabbit. Can. J. Ophthalmol. 26:247–251 (1991).
D. Maurice. Review: practical issues in intravitreal drug delivery. J. Ocul. Pharmacol. Ther. 17:393–401 (2001).
A. Hay, H. W. Flynn Jr., J. I. Hoffman, and A. H. Rivera. Needle penetration of the globe during retrobulbar and peribulbar injec-tions. Ophthalmology 98:1017–1024 (1991).
T. W. Lee and J. R. Robinson. Drug delivery to the posterior segment of the eye: some insights on the penetration pathways after subconjunctival injection. J. Ocul. Pharmacol. Ther. 17:565–572 (2001).
O. Weijtens, R. C. Schoemaker, E. G. Lentjes, F. P. Romijn, A. F. Cohen, and J. C. van Meurs. Dexamethasone concentration in the subretinal fluid after a subconjunctival injection, a peribulbar in-jection, or an oral dose. Ophthalmology 107:1932–1938 (2000).
M. S. Guirguis, S. Sattari, and F. Jamali. Pharmacokinetics of celecoxib in the presence and absence of interferon-induced acute inflammation in the rat: application of a novel HPLC assay. J. Pharm. Pharm. Sci. 4:1–6 (2001).
U. B. Kompella, N. Bandi, and S. P. Ayalasomayajula. Subcon-junctival nano-and microparticles sustain retinal delivery of budesonide, a corticosteroid capable of inhibiting VEGF expres-sion. Invest. Ophthalmol. Vis. Sci. 44:1192–1201 (2003).
U. B. Kompella and V. H. L. Lee. Barriers to drug transport in ocular epithelia. In G. L. Amidon, P. I. Lee, and E. M. Topp (eds.), Transport Processes in Pharmaceutical Systems,Marcel Dekker, New York, 1999, pp. 317–376.
D. H. Geroski and H. F. Edelhauser. Transscleral drug delivery for posterior segment disease. Adv. Drug Deliv. Rev. 52:37–48 (2001).
Y. Yanagi, Y. Tamaki, R. Obata, K. Muranaka, N. Homma, H. Matsuoka, and H. Mano. Subconjunctival administration of bu-cillamine suppresses choroidal neovascularization in rat. Invest. Ophthalmol. Vis. Sci. 43:3495–3499 (2002).
Y. Yanagi, Y. Tamaki, Y. Inoue, R. Obata, K. Muranaka, and N. Homma. Subconjunctival doxifluridine administration suppresses rat choroidal neovascularization through activated thymidine phosphorylase. Invest. Ophthalmol. Vis. Sci. 44:751–754 (2003).
E. Sakurai, A. Anand, B. K. Ambati, N. van Rooijen and J. Ambati. Macrophage depletion inhibits experimental choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 44:3578–3585 (2003).
S. F. Wen, Z. Chen, J. Nery, and B. Faha. Characterization of adenovirus p21 gene transfer, biodistribution, and immune re-sponse after local ocular delivery in New Zealand white rabbits. Exp. Eye Res. 77:355–365 (2003).
T. W. Kim, J. D. Lindsey, M. Aihara, T. L. Anthony, and R. N. Weinreb. Intraocular distribution of 70-kDa dextran after sub-conjunctival injection in mice. Invest. Ophthalmol. Vis. Sci. 43: 1809–1816 (2002).
S. O. Hung, A. Patterson, D. I. Clark, and P. J. Rees. Oral acy-clovir in the management of dendritic herpetic corneal ulceration. Br. J. Ophthalmol. 68:398–400 (1984).
P. A. Thomas, C. M. Kalavathy, D. J. Abraham and J. Rajas-ekaran. Oral ketoconazole in Keratomycosis. Indian J. Ophthal-mol. 35:197–203 (1987).
A. M. Avunduk, R. W. Beuerman, E. D. Warnel, H. E. Kaufman, and D. Greer. Comparison of efficacy of topical and oral flucona-zole treatment in experimental Aspergillus keratitis. Curr. Eye Res. 26:113–117 (2003).
D. M. Meisler, M. B. Raizman, and E. I. Traboulsi. Oral eryth-romycin treatment for childhood blepharokeratitis. J. AAPOS 4:379–380 (2000).
G. M. Grass and J. R. Robinson. Mechanisms of corneal drug penetration. I: In vivo and in vitro kinetics. J. Pharm. Sci. 77:3–14 (1988).
I. Ahmed and T. F. Patton. Importance of the noncorneal ab-sorption route in topical ophthalmic drug delivery. Invest. Oph-thalmol. Vis. Sci. 26:584–587 (1985).
R. D. Schoenwald, G. S. Deshpande, D. G. Rethwisch, and C. F. Barfknecht. Penetration into the anterior chamber via the con-junctival/ scleral pathway. J. Ocul. Pharmacol. Ther. 13:41–59 (1997).
E. F. Erkin, U. Gunenc, F. H. Oner, A. Gelal, Y. Erkin, and H. Guven. Penetration of amikacin into aqueous humor of rabbits. Ophthalmologica 215:299–302 (2001).
O. Weijtens, E. J. Feron, R. C. Schoemaker, A. F. Cohen, E. G. Lentjes, F. P. Romijn, and J. C. van Meurs. High concentration of dexamethasone in aqueous and vitreous after subconjunctival in-jection. Am. J. Ophthalmol. 128:192–197 (1999).
J. Rootman, N. Bussanich, G. Gudauskas, and C. Kumi. Effects of subconjunctivally injected antineoplastic agents on three mod-els of corneal inflammation. Can. J. Ophthalmol. 20:142–146 (1985).
J. Ambati, C. S. Canakis, J. W. Miller, E. S. Gragoudas, A. Edwards, D. J. Weissgold, I. Kim, F. C. Delori, and A. P. Adamis. Diffusion of high molecular weight compounds through sclera. Invest. Ophthalmol. Vis. Sci. 41:1181–1185 (2000).
N. Unlu and J. R. Robinson. Scleral permeability to hydrocorti-sone and mannitol in the albino rabbit eye. J. Ocul. Pharmacol. Ther. 14:273–281 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ayalasomayajula, S.P., Kompella, U.B. Retinal Delivery of Celecoxib Is Several-Fold Higher Following Subconjunctival Administration Compared to Systemic Administration. Pharm Res 21, 1797–1804 (2004). https://doi.org/10.1023/B:PHAM.0000045231.51924.e8
Issue Date:
DOI: https://doi.org/10.1023/B:PHAM.0000045231.51924.e8