Abstract
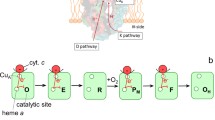
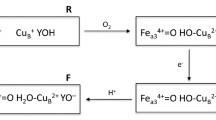
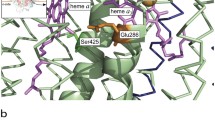
Kinetics of the reduction of the hemes in cytochrome c oxidase in the presence of high concentration of ruthenium(III)hexaammine chloride was examined using a stopped-flow spectrophotometer. Upon mixing of the oxidized enzyme with dithionite and Ru(NH3) 3+6 , three well-resolved phases were observed: heme a reduction reaching completion within a few milliseconds is followed by two slow phases of heme a 3 reduction. The difference spectrum of heme a 3 reduction in the visible region is characterized by a maximum at ∼612 nm, rather than at 603 nm as was believed earlier. It is shown that in the case of bovine heart cytochrome c oxidase containing a special cation-binding site in which reversible binding of calcium ion occurs, heme a 3 reduction is slowed down by low concentrations of Ca2+. The effect is absent in the case of the bacterial cytochrome oxidase in which the cation-binding site contains a tightly bound Ca2+ ion. The data corroborate the inhibition of the cytochrome oxidase enzymatic activity by Ca2+ ions discovered earlier and indicate that the cation affects intramolecular electron transfer.
Similar content being viewed by others
Abbreviations
- CBS:
-
cation-binding site
- COX:
-
cytochrome c oxidase
- RuAm:
-
hexaammineruthenium (Ru(NH3) 3+6 )
References
Ferguson-Miller, S., and Babcock, G. T. (1996) Chem. Rev., 7, 2889–2907.
Belevich, I., and Verkhovsky, M. I. (2008) Antiox. Redox Signal., 10, 1–29.
Yoshikawa, S., Muramoto, K., and Shinzawa-Itoh, K. (2011) Annu. Rev. Biophys., 40, 205–223.
Tsukihara, T., Aoyama, H., Yamashita, E., Takashi, T., Yamaguichi, H., Shinzawa-Itoh, K., Nakashima, R., Yaono, R., and Yoshikawa, S. (1996) Science, 272, 1136–1144.
Yoshikawa, S., Shinzawa-Itoh, K., Nakashima, R., Yaono, R., Inoue, N., Yao, M., Fei, M. J., Libeu, C. P., Mizushima, T., Yamaguchi, H., Tomizaki, T., and Tsukihara, T. (1998) Science, 280, 1723–1729.
Ostermeier, C., Harrenga, A., Ermler, U., and Michel, H. (1997) Proc. Natl. Acad. Sci. USA, 94, 10547–10553.
Pfitzner, U., Kirichenko, A., Konstantinov, A. A., Mertens, M., Wittershagen, A., Kolbesen, B. O., Steffens, G. C. M., Harrenga, A., Michel, H., and Ludwig, B. (1999) FEBS Lett., 456, 365–369.
Lee, A., Kirichenko, A., Vygodina, T., Siletsky, S. A., Das, T. K., Rousseau, D. L., Gennis, R. A., and Konstantinov, A. A. (2002) Biochemistry, 41, 8886–8898.
Kirichenko, A., Vygodina, T. V., Mkrtchyan, H. M., and Konstantinov, A. A. (1998) FEBS Lett., 423, 329–333.
Kirichenko, A. V., Pfitzner, U., Ludwig, B., Soares, C. M., Vygodina, T. V., and Konstantinov, A. A. (2005) Biochemistry, 44, 12391–12401.
Riistama, S., Laakkonen, L., Wikstrom, M., Verkhovsky, M. I., and Puustinen, A. (1999) Biochemistry, 38, 10670–10677.
Wikstrom, M., and Saari, H. (1975) Biochim. Biophys. Acta, 408, 170–179.
Saari, H., Pentilla, T., and Wikstrom, M. (1980) J. Bioenerg. Biomembr., 12, 325–338.
Mkrtchyan, H., Vygodina, T., and Konstantinov, A. A. (1990) Biochem. Int., 20, 183–190.
Vygodina, T. V., Ptushenko, V. V., and Konstantinov, A. A. (2008) Biochim. Biophys. Acta, 1777, S110–S111.
Konstantinov, A. A. (2010) Biochim. Biophys. Acta, 1797 (Suppl.), 92.
Vygodina, T. V., and Konstantinov, A. A. (2010) Biochim. Biophys. Acta, 1797, 102.
Scott, R. A., and Gray, H. B. (1980) J. Am. Chem. Soc., 102, 3219–3224.
Hochman, J. H., Partridge, B., and Ferguson-Miller, S. (1981) J. Biol. Chem., 256, 8693–8698.
Reichardt, J. K. V., and Gibson, Q. H. (1982) J. Biol. Chem., 257, 9268–9270.
Tsofina, L. M., Liberman, E. A., Vygodina, T. V., and Konstantinov, A. A. (1986) Biochem. Int., 12, 103–110.
Fowler, L. R., Richardson, S. H., and Hatefi, Y. (1962) Biochim. Biophys. Acta, 64, 170–173.
Mitchell, D. M., and Gennis, R. B. (1995) FEBS Lett., 368, 148–150.
Halaka, F. G., Babcock, G. T., and Dye, J. L. (1981) J. Biol. Chem., 256, 1084–1087.
Orii, Y. (2008) J. Bioenerg. Biomembr., 30, 47–53.
Lambeth, D. O., Campbell, K. L., Zand, R., and Palmer, G. (1973) J. Biol. Chem., 248, 8130–8136.
Nilsson, T. (1992) Proc. Natl. Acad. Sci. USA, 89, 6497–6501.
Zaslavsky, D., Kaulen, A., Smirnova, I. A., Vygodina, T. V., and Konstantinov, A. A. (1993) FEBS Lett., 336, 389–393.
Jancura, D., Antalik, M., Berka, V., Palmer, G., and Fabian, M. (2006) J. Biol. Chem., 281, 20003–20010.
Jancura, D., Berka, V., Antalik, M., Bagelova, J., Gennis, R. B., Palmer, G., and Fabian, M. (2006) J. Biol. Chem., 281, 30319–30325.
Moody, A. J. (1996) Biochim. Biophys. Acta, 1276, 6–20.
Wikstrom, M., Krab, K., and Saraste, M. (1981) Cytochrome Oxidase — a Synthesis, Academic Press, N. Y.
Zimmermann, B. H., Nitsche, C. I., Fee, J. A., Rusnak, F., and Munck, E. (1988) Proc. Natl. Acad. Sci. USA, 85, 5779–5783.
Oertling, W. A., Surerus, K. K., Einarsdottir, O., Fee, J. A., Dyer, R. B., and Woodruff, W. H. (1994) Biochemistry, 33, 3128–3141.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2012, Vol. 77, No. 8, pp. 1095–1104.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM12-049, June 17, 2012.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Vygodina, T.V., Dyuba, A.V. & Konstantinov, A.A. Effect of calcium ions on electron transfer between hemes a and a 3 in cytochrome c oxidase. Biochemistry Moscow 77, 901–909 (2012). https://doi.org/10.1134/S0006297912080111
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297912080111