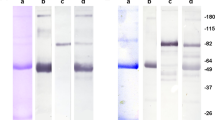
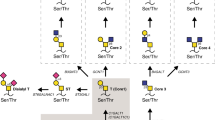
Abstract
Tn antigen is a tumor-associated antigen that appears on cancer cells as a result of aberrant O-glycosylation. The most studied form of Tn antigen is found in mucins, in particular, in mucin 1 (MUC1). Antibodies against this form of Tn antigen are used to diagnose tumors, as well as to generate T-killers with a chimeric receptor. Some carcinomas do not carry MUC1 and antibodies of a different specificity are required to detect Tn antigen on these cells. In our work, we searched for anti-Tn antibodies without preliminary assumptions about the proteins that may be carriers of the Tn antigen. For this purpose, we obtained several pairs of isogenic cell lines with the wild type and knockout of the Cosmc gene, which is essential for correct protein O-glycosylation. Using the created lines as immunogens, we generated a monoclonal antibody AKC3, which reacted with the Cosmc-deficient A549 lung adenocarcinoma cells and did not bind to the wild-type cells. Using mass spectrometry, as well as co-immunoprecipitation, it was shown that the AKC3 antibody recognized the Tn antigen in the context of CD44 protein – a protein important for tumor growth. The AKC3 antibody can be used for tumor diagnosis, and to generate T cells with a chimeric receptor for treatment of tumors that do not express mucins.





Similar content being viewed by others
Abbreviations
- GalNAc:
-
N-acetyl-D-galactosamine
- LRG1:
-
leucine rich alpha-2-glycoprotein 1; MUC1, mucin 1
- MW:
-
molecular weight
- PAGE:
-
polyacrylamide gel electrophoresis
- PBS:
-
phosphate-buffered saline
- SDS:
-
sodium dodecyl sulfate
- WB:
-
Western blotting
References
Ju, T., Otto, V. I., and Cummings, R. D. (2011) The Tn antigen-structural simplicity and biological complexity, Angew. Chem. Int. Ed., 50, 1770-1791, doi: https://doi.org/10.1002/anie.201002313 .
Ju, T., Lanneau, G. S., Gautam, T., Wang, Y., Xia, B., Stowell, S. R., Willard, M. T., Wang, W., Xia, J. Y., Zuna, R. E., Laszik, Z., Benbrook, D. M., Hanigan, M. H., and Cummings, R. D. (2008) Human tumor antigens Tn and sialyl Tn arise from mutations in Cosmc, Cancer Res., 68, 1636-1646, doi: https://doi.org/10.1158/0008-5472.CAN-07-2345 .
Springer, G. F. (1997) Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy, J. Mol. Med., 75, 594-602, doi: https://doi.org/10.1007/s001090050144 .
Schietinger, A., Philip, M., Yoshida, B. A., Azadi, P., Liu, H., Meredith, S. C., and Schreiber, H. (2006) A mutant chaperone converts a wild-type protein into a tumor-specific antigen, Science, 314, 304-308, doi: https://doi.org/10.1126/science.1129200 .
Tarp, M. A., and Clausen, H. (2008) Mucin-type O-glycosylation and its potential use in drug and vaccine development, Biochim. Biophys. Acta, 1780, 546-563, doi: https://doi.org/10.1016/j.bbagen.2007.09.010 .
Persson, N., Stuhr-Hansen, N., Risinger, C., Mereiter, S., Polónia, A., Polom, K., Kovács, A., Roviello, F., Reis, C. A., Welinder, C., Danielsson, L., Jansson, B., and Blixt, O. (2017) Epitope mapping of a new anti-Tn antibody detecting gastric cancer cells, Glycobiology, 27, 635-645, doi: https://doi.org/10.1093/glycob/cwx033 .
Blixt, O., Lavrova, O. I., Mazurov, D. V., Clo, E., Kracun, S. K., Bovin, N. V., and Filatov, A. V. (2012) Analysis of Tn antigenicity with a panel of new IgM and IgG1 monoclonal antibodies raised against leukemic cells, Glycobiology, 22, 529-542, doi: https://doi.org/10.1093/glycob/cwr178 .
Mazurov, D., Ilinskaya, A., Heidecker, G., and Filatov, A. (2012) Role of O-glycosylation and expression of CD43 and CD45 on the surfaces of effector T cells in human T cell leukemia virus type 1 cell-to-cell infection, J. Virol., 86, 2447-2458, doi: https://doi.org/10.1128/JVI.06993-11 .
Hsu, P. D., Scott, D. A., Weinstein, J. A., Ran, F. A., Konermann, S., Agarwala, V., Li, Y., Fine, E. J., Wu, X., Shalem, O., Cradick, T. J., Marraffini, L. A., Bao, G., and Zhang, F. (2013) DNA targeting specificity of RNA-guided Cas9 nucleases, Nat. Biotechnol., 31, 827-832, doi: https://doi.org/10.1038/nbt.2647 .
Stolfa, G., Mondal, N., Zhu, Y., Yu, X., Buffone, A., and Neelamegham, S. (2016) Using CRISPR-Cas9 to quantify the contributions of O-glycans, N-glycans and Glycosphingolipids to human leukocyte-endothelium adhesion, Sci. Rep., 6, 30392, doi: https://doi.org/10.1038/srep30392 .
Tarasevich, A., Filatov, A., Pichugin, A., and Mazurov, D. (2015) Monoclonal antibody profiling of cell surface proteins associated with the viral biofilms on HTLV-1 transformed cells, Acta Virol., 59, 247-256, doi: https://doi.org/10.4149/av_2015_03_247 .
Dobrochaeva, K., Khasbiullina, N., Shilova, N., Antipova, N., Obukhova, P., Ovchinnikova, T., Galanina, O., Blixt, O., Kunz, H., Filatov, A., Knirel, Y., LePendu, J., Khaidukov, S., and Bovin, N. (2020) Specificity of human natural antibodies referred to as anti-Tn, Mol. Immunol., 120, 74-82, doi: https://doi.org/10.1016/j.molimm.2020.02.005 .
Filatov, A. V., Krotov, G. I., Zgoda, V. G., and Volkov, Y. (2007) Fluorescent immunoprecipitation analysis of cell surface proteins: a methodology compatible with mass-spectrometry, J. Immunol. Methods, 319, 21-33, doi: https://doi.org/10.1016/j.jim.2006.09.014 .
Azevedo, R., Gaiteiro, C., Peixoto, A., Relvas-Santos, M., Lima, L., Santos, L. L., and Ferreira, J. A. (2018) CD44 glycoprotein in cancer: a molecular conundrum hampering clinical applications, Clin. Proteomics, 15, 22, doi: https://doi.org/10.1186/s12014-018-9198-9 .
Gasbarri, A., Del Prete, F., Girnita, L., Martegani, M. P., Natali, P. G., and Bartolazzi, A. (2003) CD44s adhesive function spontaneous and PMA-inducible CD44 cleavage are regulated at post-translational level in cells of melanocytic lineage, Melanoma Res., 13, 325-337, doi: https://doi.org/10.1097/00008390-200308000-00001 .
Ponta, H., Sherman, L., and Herrlich, P. A. (2003) CD44: from adhesion molecules to signalling regulators, Nat. Rev. Mol. Cell Biol., 4, 33-45, doi: https://doi.org/10.1038/nrm1004 .
Naor, D., Sionov, R. V., and Ish-Shalom, D. (1997) CD44: structure, function and association with the malignant process, Adv. Cancer Res., 71, 241-319, doi: https://doi.org/10.1016/S0065-230X(08)60101-3 .
Hu, B., Ma, Y., Yang, Y., Zhang, L., Han, H., and Chen, J. (2018) CD44 promotes cell proliferation in non-small cell lung cancer, Oncol. Lett., 15, 5627-5633 doi: https://doi.org/10.3892/ol.2018.8051 .
Du, T., Jia, X., Dong, X., Ru, X., Li, L., Wang, Y., Liu, J., Feng, G., and Wen, T. (2020) Cosmc disruption-mediated aberrant O-glycosylation suppresses breast cancer cell growth via impairment of CD44, Cancer Manag. Res., 12, 511-522, doi: https://doi.org/10.2147/CMAR.S234735 .
O’Donnell, L. C., Druhan, L. J., and Avalos, B. R. (2002) Molecular characterization and expression analysis of leucine-rich alpha2-glycoprotein, a novel marker of granulocytic differentiation, J. Leukoc. Biol., 72, 478-485.
Fang, X. J., Jiang, H., Zhu, Y. Q., Zhang, L. Y., Fan, Q. H., and Tian, Y. (2014) Doxorubicin induces drug resistance and expression of the novel CD44st via NF-κB in human breast cancer MCF-7 cells, Oncol. Rep., 31, 2735-2742, doi: https://doi.org/10.3892/or.2014.3131 .
Campos, D., Freitas, D., Gomes, J., Magalhães, A., Steentoft, C., Gomes, C., Vester-Christensen, M. B., Ferreira, J. A., Afonso, L. P., Santos, L. L., Pinto de Sousa, J., Mandel, U., Clausen, H., Vakhrushev, S. Y., and Reis, C. A. (2015) Probing the O-glycoproteome of gastric cancer cell lines for biomarker discovery, Mol. Cell. Proteomics, 14, 1616-1629, doi: https://doi.org/10.1074/mcp.M114.046862 .
Steentoft, C., Fuhrmann, M., Battisti, F., Van Coillie, J., Madsen, T. D., Campos, D., Adnan Halim, A., Vakhrushev, S. Y., Joshi, H. J., Schreiber, H., Mandel, U., and Narimatsu, Y. (2019) A strategy for generating cancer-specific monoclonal antibodies to aberrant O-glycoproteins: identification of a novel dysadherin-Tn antibody, Glycobiology, 29, 307-319, doi: https://doi.org/10.1093/glycob/cwz004 .
Welinder, C., Baldetorp, B., Borrebaeck, C., Fredlund, B.-M., and Jansson, B. (2011) A new murine IgG1 anti-Tn monoclonal antibody with in vivo anti-tumor activity, Glycobiology, 21, 1097-1107, doi: https://doi.org/10.1093/glycob/cwr048 .
Posey, A. D., Schwab, R. D., Boesteanu, A. C., Steentoft, C., Mandel, U., Engels, B., Stone, J. D., Madsen, T. D., Schreiber, K., Haines, K. M., Cogdill, A. P., Chen, T. J., Song, D., Scholler, J., Kranz, D. M., Feldman, M. D., Young, R., Keith, B., Schreiber, H., Clausen, H., Johnson, L. A., and June, C. H. (2016) Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma, Immunity, 44, 1444-1454, doi: https://doi.org/10.1016/j.immuni.2016.05.014 .
Gorchakov, A. A., Kulemzin, S. V., Kochneva, G. V., and Taranin, A. V. (2020) Challenges and prospects of chimeric antigen receptor T-cell therapy for metastatic prostate cancer, Eur. Urol., 77, 299-308, doi: https://doi.org/10.1016/j.eururo.2019.08.014 .
Funding
This work was financially supported by the Russian Foundation for Basic Research (project no. 18-29-07025 mk).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest in financial or any other sphere. This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Shuvalova, M.L., Kopylov, A.T., Mazurov, D.V. et al. CD44-Associated Tn Antigen as a New Biomarker of Tumor Cells with Aberrant Glycosylation. Biochemistry Moscow 85, 1064–1071 (2020). https://doi.org/10.1134/S0006297920090060
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297920090060