Abstract
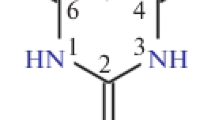
The crystal and molecular structures of thallium(I) thiobarbiturate C4H3N2O2STl (C4H4N2O2S is 2-thiobarbituric acid, Н2ТВА) have been determined. Crystallographic data for Tl(НТВА) are a = 11.2414(7) Å, b = 3.8444(3) Å, с = 14.8381(9) Å, β = 99.452(2)°, V = 649.00(7) Å3, space group P2/с, Z = 4. Each of the two independent thallium ions is bonded to four oxygen and two sulfur atoms to form a distorted tetrahedron. N‒H…O and C‒H…S hydrogen bonds form a branched three-dimensional network. The structure is also stabilized by π‒π interaction between heterocyclic НТВА- ions. The IR spectra of Tl(НТВА) agree with X-ray powder diffraction data. The compound is also stable below 280°C, and Tl2SO4 is one of the thermolysis products in an oxidative medium in the region of 500‒650°C.
Similar content being viewed by others
References
Biological Inorganic Chemistry, Ed. by I. Bertini, H. B. Gray, E. I. Stiefel, and J. Valentine (BINOM. Laboratoriya Znanii, Moscow, 2013), Vol.1.
K. M. Coup and P. J. Swedlund, Chem. Geol. 398, 97 (2015).
A. Turner, D. Turner, and C. Braungardt, Marine Pollut. Bull 69, 172 (2013).
M. A. McGuire, T. K. Reynolds, and F. J. DiSalvo, Chem. Mater. 17, 2875 (2005).
S. Bondock, T. A. El-Gaber, and A. A. Fadda, Phosphorus Sulfur Silicon Relat. Elem. 182, 1915 (2007).
R. Ya. Levina and F. K. Velichko, Russ. Chem. Rev. 29, 437 (1960).
V. K. Ahluwalia and R. Aggarwal, Proc. Ind. Nat. Acad. Sci. 5, 369 (1996).
J. H. Lee, S. Lee, M. Y. Park, and H. J. Myung, Virology 8, 18 (2011).
N. R. Penthala, P. R. Ponugoti, V. Kasam, and P. A. Crooks, Bioorg. Med. Chem. Lett. 23, 1442 (2013).
M. A. El-Gahami, S. A. Ibrahim, and H. M. A. Salman, Synth. React. Inorg. Met.-Org. Chem. 21, 1497 (1991).
V. I. Balas, I. I. Verginadis, G. D. Geromichalos, et al., Eur. J. Med. Chem. 46, 2835 (2011).
N. N. Golovnev and M. S. Molokeev, 2-Thiobarbituric Acid and Its Complexes with Metals: Synthesis, Structure, Properties (Siberian Federal Univ., Krasnoyarsk, 2014) [in Russian].
M. Kubicki, A. Owczarzak, V. I. Balas, et al., J. Coord. Chem. 65, 1107 (2012).
N. N. Golovnev, M. S. Molokeev, and M. Y. Belash, J. Struct. Chem. 54, 566 (2013).
Bruker AXS TOPAS V4: General Profile and Structure Analysis Software for Powder Diffraction Data—User’s Manual (Bruker, Karlsruhe, 2008).
Cambridge Structural Database, Ver. 5.34 (Univ. of Cambridge, Cambridge, UK, 2012).
PLATON: A Multipurpose Crystallographic Tool (Utrecht Univ. Utrecht, The Netherlands, 2008).
J. V. Steed and J. L. Atwood, Supramolecular Chemistry (Wiley, Chichester, 2000; Akademkniga, Moscow, 2007).
N. N. Golovnev and M. S. Molokeev, Zh. Strukt. Khim. 55, 122 (2014).
M. R. Chierotti, L. Ferrero, N. Garino, et al., Chem. Eur. J. 16, 4347 (2010).
N. A. Smorygo and B. A. Ivin, Khim. Geterotsikl. Soedin., No. 10, 1402 (1975).
F. Ramondo, A. Pieretti, L. Gontrani, and L. Bencivenni, Chem. Phys. 271, 293 (2001).
E. Mendez, M. F. Cerda, J. S. Gancheff, et al., J. Phys. Chem. C 111, 3369 (2007).
N. N. Golovnev, M. S. Molokeev, S. N. Vereshchagin, et al., Polyhedron 70, 71 (2014).
V. A. Rabinovich and Z. Ya. Khavin, The Concise Handbook of Chemistry (Khimiya, Leningrad, 1978).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.N. Golovnev, M.S. Molokeev, 2016, published in Zhurnal Neorganicheskoi Khimii, 2016, Vol. 61, No. 4, pp. 463–467.
Rights and permissions
About this article
Cite this article
Golovnev, N.N., Molokeev, M.S. Crystal structure of thallium(I) 2-thiobarbiturate. Russ. J. Inorg. Chem. 61, 442–446 (2016). https://doi.org/10.1134/S0036023616040100
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023616040100