Abstract
The previously described algorithm for constructing isobaric–isothermal subsolidus sections of four-component systems comprising stoichiometric compounds is used as the base for developing an approach to the enumeration of melting diagrams of such the systems. The underlying idea of the approach is the placement of different types of invariant points over the tetrahedration elements of the phase diagram. Next, adjacent invariant points are connected by lines on which arrows are placed to indicate the decreasing temperature direction. This procedure is carried out within the limits imposed by the properties of the phase diagrams. Exemplary schematic melting diagrams constructed with the use of the described approach are provided. The results of the work can be used to design an optimization algorithm for an experimental study of phase diagrams, as well as to develop databases for such the diagrams.


Similar content being viewed by others
Notes
Kosyakov, Shestakov, Grachev, and Komarov [28] suggested, just as we suggest here, that these phases have small homogeneity areas, so they can be regarded as stoichiometric phases.
The program provides imaging of the results of calculations in the form of WRL files.
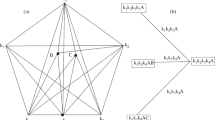
The temperature decreases along a monovariant line as the line thickness decreases.
A diotectic point is an invariant point from which two monovariant lines originate as temperature lowers; a tritectic point is an invariant point from which three monovariant lines originate as temperature lowers.
REFERENCES
Y. Liu, Therm. Acta 555, 53 (2013). https://doi.org/10.1016/j.tca.2012.12.022
V. I. Kosyakov, V. A. Shestakov, and E. V. Grachev, Russ. J. Inorg. Chem. 55, 611 (2010). https://doi.org/10.1134/S0036023610040194
P. P. Fedorov, Russ. J. Inorg. Chem. 50, 1933 (2005).
V. I. Kosyakov, Dokl. Chem. 374, 199 (2000).
V. Lutsyk and V. Vorob’eva, Proceedings of the International Conference on Phase Diagram Calculations and Computational Thermochemistry Calphad XXXVIII, Prague, 2009, p. 66.
L. A. Serafimov, Russ. J. Phys. Chem. A 76, 1211 (2002).
N. A. Charykov, A. V. Rumyantsev, and M. V. Charykova, Russ. J. Phys. Chem. A 72, 1761 (1998).
K. A. Khaldoyanidi, J. Struct. Chem. 44, 116 (2003).
L. S. Palatnik, Russ. J. Phys. Chem. A 65, 3208 (1991).
V. I. Kosyakov, Russ. J. Phys. Chem. A 93, 1635 (2019). https://doi.org/10.1134/S0036024419090085
V. I. Lutsyk and V. P. Vorob’eva, Russ. J. Inorg. Chem. 59, 956 (2014). https://doi.org/10.7868/S0044457X14090128
A. V. Frolkova, T. E. Ososkova, and A. K. Frolkova, Theor. Found. Chem. Eng. 54, 407 (2020). https://doi.org/10.1134/S0040579520020049
V. I. Lutsyk and V. P. Vorob’eva, Russ. J. Inorg. Chem. 59, 1123 (2014). https://doi.org/10.1134/S0036023614100106
V. I. Kosyakov, N. A. Pylneva, Z. G. Bazarova, and A. M. Yurkin, Mater. Res. Bull. 36, 573 (2001). https://doi.org/10.1016/S0025-5408(01)00519-0
A. M. Gasanaliev, B. Y. Gamataeva, and D. E. Bekova, Russ. J. Inorg. Chem. 50, 1080 (2005).
V. I. Kosyakov, Russ. J. Inorg. Chem. 53, 946 (2008). https://doi.org/10.1134/S0036023608060223
P. A. Akhmedova, B. Yu. Gamataeva, and A. M. Gasanaliev, Russ. J. Inorg. Chem. 54, 779 (2009). https://doi.org/10.1134/S0036023609050179
Zh. A. Kochkarov and I. V. Shogenov, Russ. J. Inorg. Chem. 53, 1517 (2008). https://doi.org/10.1134/S0036023608090283
D. G. Cherkasov, V. F. Kurskii, and K. K. Il’in, Russ. J. Inorg. Chem. 53, 139 (2008). https://doi.org/10.1134/S0036023608010208
D. G. Cherkasov, V. F. Kurskii, S. I. Sinegubova, and K. K. Il’in, Russ. J. Inorg. Chem. 54, 969 (2009). https://doi.org/10.1134/S0036023609060217
D. G. Cherkasov, M. P. Smotrov, and K. K. Il’in, Russ. J. Phys. Chem. A 84, 922 (2010). https://doi.org/10.1134/S0036024410060063
D. G. Cherkasov, K. K. Il’in, and V. F. Kurskii, Russ. J. Inorg. Chem. 56, 787 (2011). https://doi.org/10.1134/S0036023611050068
V. F. Kurskii, D. G. Cherkasov, and K. K. Il’in, Izv. Sarat. Univ. Ser. Khim. Biol. Ekol. 6, 9 (2006).
A. Smirnov, A. Samarov, and M. Toikka, J. Chem. Eng. Data 66, 1466 (2021). https://doi.org/10.1021/acs.jced.0c01066
A. F. Martínez, C. A. Sánchez, A. Orjuela, and G. Rodríguez, Fluid Phase Equilib. 516, 112612 (2020). https://doi.org/10.1016/j.fluid.2020.112612
A. V. Basko, K. V. Pochivalov, A. V. Bazanov, et al., Thermochim. Acta. 684, 178499 (2020). https://doi.org/10.1016/j.tca.2019.178499
V. L. Solozhenko and V. Z. Turkevich, J. Phys. Chem. 122, 8505 (2018). https://doi.org/10.1021/acs.jpcc.8b00102
V. I. Kosyakov, V. A. Shestakov, E. V. Grachev, and V. Yu. Komarov, Russ. J. Inorg. Chem. 61, 1274 (2016). https://doi.org/10.1134/S0036023616100119
V. A. Shestakov, E. V. Grachev, and V. I. Kosyakov, Russ. J. Phys. Chem. A 94, 1083 (2020). https://doi.org/10.1134/S0036024420060205
V. A. Shestakov and V. I. Kosyakov, Russ. J. Inorg. Chem. 66, 401 (2021). https://doi.org/10.1134/S0036023621030165
ACKNOWLEDGMENTS
The authors are grateful to V.I. Kosyakov for formulating the task and for valuable comments.
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation (project No. 121031700314-5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Shestakov, V.A., Grachev, E.V. Enumeration of Melting Diagrams for Four-Component Systems Comprising Stoichiometric Compounds. Russ. J. Inorg. Chem. 67, 488–491 (2022). https://doi.org/10.1134/S0036023622040179
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622040179