Abstract
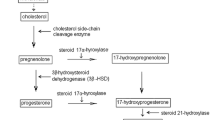
Study of changes in the proteome of extracellular body fluids under conditions of simulated weightlessness of medium duration (21 days) remains relevant for clarifying the physiological mechanisms of homeostasis regulation and is important for gravitational physiology and medicine. Plasma samples of 8 healthy volunteers participating in head down bed rest (HDBR –6°) experiment were studied using semi-quantitative proteomics methods. Each volunteer participated both in the control session and in the session with physical training for negative changes prevention. By the end of HDBR, significant changes in the concentration of proteins involved in platelet degranulation, blood coagulation, fibrinolysis, proteolysis regulation, complement activation, and the immune response were detected in both sessions. The following changes in biochemical parameters of carbohydrate metabolism and regulation of circulating blood volume were shown: a significant increase in renin concentration and a tendency to an increase in aldosterone; an increase in fasting insulin concentration and a tendency to an increase in the insulin resistance index. Physical training did not have a significant effect on biochemical parameters, except for the representation of cholesterol fractions, un which a significant decrease in the concentration of high-density lipoprotein cholesterol observed in HDBR became statistically insignificant in session with physical training. However, the protein composition in blood plasma of volunteers in the prophylaxis session changed less compared to HDBR. Physical training resulted in increase in the concentration of proteins involved in the normalization of carbohydrate and lipid metabolism, such as a macrophage stimulating protein and phosphatidylinositol-glycan-specific phospholipase D, which indicates the effectiveness of preventive measures.




Similar content being viewed by others
REFERENCES
Larina, I.M., Popova, I.A., Mikhailov, V.M., et al., The hormonal mechanisms supporting muscle work during prolonged head-down tile hypokinesia, Fiziol. Chel., 1999, vol. 25, no. 3, p. 117.
Grigor’ev, A.I., Water-salt metabolism and renal function in humans with prolonged hypokinesia, Nefrologiya, 2001, no. 3, p. 7.
Mikhailov, V.M., Hypokinesia as a risk factor in extreme conditions, Aviakosm. Ekol. Med., 2001, vol. 35, no. 2, p. 26.
Batchu, S.N., Smolock, E.M., Dyachenko, I.A., et al., Autonomic dysfunction determines stress-induced cardiovascular and immune complications in mice, J. Am. Heart Assoc., 2015, vol. 4, no. 5, p. e001952.
Baranov, M.V., Katuntsev, V.P., Shpakov, A.V., and Baranov, V.M., A method of ground simulation of physiological effects of hypogravity on humans, Bull. Exp. Biol. Med., 2016, vol. 160, no. 3, p. 401.
Coupé, M., Yuan, M., Demiot, C., et al., Low-magnitude whole body vibration with resistive exercise as a countermeasure against cardiovascular deconditioning after 60 days of head-down bed rest, Am. J. Physiol.: Regul. Integr. Comp. Physiol., 2011, vol. 301, no. 6, p. R1748.
Greaves, D., Arbeille, P., Guillon, L., et al., Effects of exercise countermeasure on myocardial contractility measured by 4D speckle tracking during a 21-day head-down bed rest, Eur. J. Appl. Physiol., 2019, vol. 119, nos. 11–12, p. 2477.
Afonin, B.V., Effect of space flight antiorthostatic hypokinesia of various durations on blood insulin level, Kosm. Biol. Aviakosm. Med., 1989, vol. 23, no. 3, p. 77.
Kermorgant, M., Nasr, N., Custaud, M.A., et al., Effects of resistance exercise and nutritional supplementation on dynamic cerebral autoregulation in Head-Down Bed Rest, Front. Physiol., 2019, vol. 10, p. 1114.
Brzhozovskiy, A.G., Kononikhin, A.S., Pastushkova, L.Kh., et al., The effects of spaceflight factors on the human plasma proteome, including both real space missions and ground-based experiments, Int. J. Mol. Sci., 2019, vol. 20, no. 13, p. 3194.
Prieto, D., Contreras, C., and Sánchez, A., Endothelial dysfunction, obesity and insulin resistance, Curr. Vasc. Pharmacol., 2014, vol. 12, no. 3, p. 412.
Wang, M.H., Zhou, Y.Q., and Chen, Y.Q., Macrophage-stimulating protein and RON receptor tyrosine kinase: potential regulators of macrophage inflammatory activities, Scand. J. Immunol., 2002, vol. 56, no. 6, p. 545.
Chanda, D., Li, T., Song, K.H., et al., Hepatocyte growth factor family negatively regulates hepatic gluconeogenesis via induction of orphan nuclear receptor small heterodimer partner in primary hepatocytes, J. Biol. Chem., 2009, vol. 284, no. 42, p. 28510.
Bezerra, J.A., Carrick, T.L., Degen, J.L., et al., Biological effects of targeted inactivation of hepatocyte growth factor-like protein in mice, J. Clin. Invest., 1998, vol. 101, no. 5, p. 1175.
Yu, S., Allen, J.N., Dey, A., et al., The Ron receptor tyrosine kinase regulates macrophage heterogeneity and plays a protective role in diet-induced obesity, atherosclerosis, and hepatosteatosis, J. Immunol., 2016, vol. 197, no. 1, p. 256.
Tsukagawa, E., Adachi, H., Hirai, Y., et al., Independent association of elevated serum hepatocyte growth factor levels with development of insulin resistance in a 10-year prospective study, Clin. Endocrinol., 2013, vol. 79, no. 1, p. 43.
O’Neill, L.A. and Hardie, D.G., Metabolism of inflammation limited by AMPK and pseudo-starvation, Nature, 2013, vol. 493, no. 7432, p. 346.
Zhang, B.B., Zhou, G., and Li, C., AMPK: an emerging drug target for diabetes and the metabolic syndrome, Cell Metab., 2009, vol. 9, no. 5, p. 407.
Deeg, M.A., Bierman, E.L., and Cheung, M.C., GPI-specific phospholipase D associates with an apoA-I- and apoA-IV-containing complex, J. Lipid Res., 2001, vol. 42, no. 3, p. 442.
Hoener, M.C. and Brodbeck, U., Phosphatidylinositol-glycanspecific phospholipase D is an amphiphilic glycoprotein that in serum is associated with high-density lipoproteins, Eur. J. Biochem., 1992, vol. 206, no. 3, p. 747.
von Toerne, C., Huth, C., de las Heras Gala, T., et al., MASP1, THBS1, GPLD1 and ApoA-IV are novel biomarkers associated with prediabetes: the KORA F4 study, Diabetologia, 2016, vol. 59, no. 9, p. 1882.
Schofield, J.N., Stephens, J.W., Hurel, S.J., et al., Insulin reduces serum glycosylphosphatidylinositol phospholipase D levels in human type I diabetic patients and streptozotocin diabetic rats, Mol. Genet. Metab., 2002, vol. 75, no. 2, p. 154.
Suzuki, S., Suzuki, C., Hinokio, Y., et al., Insulin-mimicking bioactivities of acylated inositol glycans in several mouse models of diabetes with or without obesity, PLoS One, 2014, vol. 9, no. 6, p. e100466.
Kristiansen, S. and Richter, E.A., GLUT4-containing vesicles are released from membranes by phospholipase D cleavage of a GPI anchor, Am. J. Physiol.: Endocrinol. Metab., 2002, vol. 283, no. 2, p. 374.
Deeg, M.A., Bowen, R.F., Williams, M.D., et al., Increased expression of GPI-specific phospholipase D in mouse models of type 1 diabetes, Am. J. Physiol.: Endocrinol. Metab., 2001, vol. 281, no. 1, p. 147.
Schofield, J.N., Stephens, J.W., Hurel, S.J., et al., Insulin reduces serum glycosylphosphatidylinositol phospholipase D levels in human type I diabetic patients and streptozotocin diabetic rats, Mol. Genet. Metab., 2002, vol. 75, no. 2, p. 154.
Qin, W., Liang, Y.Z., Qin, B.Y., et al., The clinical significance of glycoprotein phospholipase D levels in distinguishing early stage latent autoimmune diabetes in adults and type 2 diabetes, PLoS One, 2016, vol. 11, no. 6, p. 1.
Raikwar, N.D., Bowen-Deeg, R.F., Du, X.S., et al., Glycosylphosphatidylinositol-specific phospholipase D improves glucose tolerance, Metabolism, 2010, vol. 59, no. 10, p. 1413.
Ganse, B., Limper, U., Bühlmeier, J., et al., Reproducible pattern of distribution and increased appearance after bed rest, Aviat. Space Environ. Med., 2013, vol. 84, no. 8, p. 864.
Matczuk, J., Zalewska, A., Łukaszuk, B., et al., Insulin resistance and obesity affect lipid profile in the salivary glands, J. Diabetes Res., 2016, vol. 2016, p. 8 163 474.
Funding
This work was supported by the Russian Foundation for Basic Research, project no. 18-34-00524.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare no apparent or potential conflicts of interest related to the publication of this article.
Statement of compliance with standards of research involving humans as subjects. All studies were carried out in accordance with the principles of biomedical ethics formulated in the Helsinki Declaration of 1964 and its subsequent updates and approved by the ethics committee of the Central University Hospital of Toulouse (France). Each study participant submitted voluntary written informed consent, signed by him after explaining to him the potential risks and benefits, as well as the nature of the forthcoming study.
Additional information
Translated by I. Shipounova
Rights and permissions
About this article
Cite this article
Kashirina, D.N., Pastushkova, L.K., Brzhozovskiy, A.G. et al. Research of the Plasma Protein Profile in Comparison with the Biochemical Parameters of Blood of Volunteers in a 21-Day Head Down Bed Rest. Hum Physiol 46, 423–431 (2020). https://doi.org/10.1134/S0362119720040064
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0362119720040064