Abstract
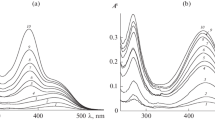
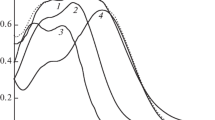
It has been shown that adsorption of 3-phenylpropenic (cinnamic), 3-(4-hydroxyphenyl)propenic (coumaric), and 3-(3-methoxy-4-hydroxyphenyl)propenic (ferulic) acids on the surface of highly dispersed alumina is characterized by L-type isotherms according to the Giles classification. The maximum adsorption values (~3.3 × 10–4 mol/g), as well as dissociation constants (pKCOOH = 4.5 ± 0.1), are almost equal for all acids, thereby suggesting that their molecules are identically bound to the adsorbent surface via carboxyl groups. The pH dependences of the adsorption of cinnamic, coumaric, and ferulic acids are described by dome-shaped curves, and the positions of their maxima in the pH scale coincide with the thermodynamic constants of carboxyl group dissociation. A linear correlation found between the adsorption values of 3-phenylpropenic acids and their hydrophobicity parameters log P indicates a noticeable contribution of hydrophobic interactions to the adsorption of the acids on the surface of highly dispersed alumina.





Similar content being viewed by others
REFERENCES
El-Seedi, H.R., El-Said, A.M., Khalifa, S.A., et al., J. Agric. Food Chem., 2012, vol. 60, p. 10877.
Balasundram, N., Sundram, K., and Samman, S., Food Chem., 2006, vol. 99, p. 191.
Razzaghi-Asl, N., Garrido, J., Khazraei, H., et al., Curr. Med. Chem., 2013, vol. 20, p. 4436.
Yashin, Ya.I., Ryzhnev, V.Yu., Yashin, A.Ya., and Chernousova, N.I., Prirodnye antioksidanty. Soderzhanie v pishchevykh produktakh i vliyanie ikh na zdo-rov’e i starenie cheloveka (Natural Antioxidants. Content in Food and Their Impact on Human Health and Aging), Moscow: TransLit, 2009.
Yashin, A.Ya., Sorbts. Khromatogr. Protsessy, 2014, vol. 14, p. 419.
Fizicheskaya khimiya lignina (Physical Chemistry of Lignin), Bogolitsyn, K.G. and Lunin, V.V., Eds., Arkhangelsk: Arkhang. Gos. Tekh. Univ., 2009.
Arceusz, A., Wesolowski, M., and Konieczynski, P., Nat. Prod. Commun., 2013, vol. 8, p. 1821.
Zeng, H., Liu, Z., Zhao, S., et al., J. Sep. Sci., 2016, vol. 39, p. 3806.
Shil’ko, E.A., Milevskaya, V.V., Temerdashev, Z.A., and Kiseleva, N.V., Anal. Kontrol, 2018, vol. 22, p. 303.
Silva, M., Castellanos, L., and Ottens, M., Ind. Eng. Chem. Res., 2018, vol. 57, p. 5359.
Dávila-Guzman, N.E., Cerino-Córdova, F.J., Diaz-Flores, P.E., et al., Chem. Eng. J., 2012, vol. 183, p. 112.
Simon, V., Thuret, A., Candy, L., et al., Chem. Eng. J., 2015, vol. 280, p. 748.
Thiel, A., Tippkötter, N., Suck, K., et al., Eng. Life Sci., 2013, vol. 13, p. 239.
Anastas, P.T. and Warner, J.C., Green Chemistry: Theory and Practice, Oxford: Oxford Univ. Press, 2000.
Eldin, A.B., Ismaiel, O.A., Hassan, W.E., and Shalaby, A.A., J. Anal. Chem., 2016, vol. 71, p. 861.
Pogorelyi, V.K., Barvinchenko, V.N., Pakhlov, E.M., and Smirnova, O.V., Colloid J., 2005, vol. 67, p. 172.
Dovbii, O.A., Kazakova, O.A., and Lipkovskaya, N.A., Colloid J., 2006, vol. 68, p. 707.
Pogorelyi, V.K., Kazakova, O.A., Barvinchenko, V.N., et al., Colloid J., 2007, vol. 69, p. 203.
Chukin, G.D., Stroenie oksida alyuminiya i katalizato-rov gidroobesserivaniya. Mekhanizmy reaktsii (Structure of Aluminum Oxide and Hydrodesulfurization Catalysts), Moscow: Printa, 2010.
Tikhomirova, T.I., Kubyshev, S.S, and Ivanov, A.V., Russ. J. Phys. Chem. A, 2013, vol. 87, p. 1357.
Jiang, L., Gao, L., and Liu, Y., Colloids Surf. A, 2002, vol. 211, p. 165.
Bernshtein, I.Ya. and Kaminskii, Yu.L., Spektrofotometricheskii analiz v organicheskoi khimii (Spectrophotometric Analysis in Organic Chemistry), Leningrad: Khimiya, 1986.
Barvinchenko, V.N., Lipkovskaya, N.A., Kulik, T.V., and Kartel’, N.T., Colloid J., 2019, vol. 81, p. 1.
Westall, J.C. and Hohl, H., Adv. Colloid Interface Sci., 1980, vol. 12, p. 265.
Tombácz, E. and Szekeres, M., Langmuir, 2001, vol. 17, p. 1411.
Tombácz, E., Szekeres, M., and Klumpp, E., Langmuir, 2001, vol. 17, p. 1420.
Giles, C.H., MacEwan, T.H., Nakhwa, S.N., and Smith, D., J. Chem. Soc., 1960, vol. 10, p. 3973.
Chemical Information Resources from the National Library of Medicine. http://sis.nlm.nih.gov/chemical.html.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Additional information
Translated by A. Kirilin
Rights and permissions
About this article
Cite this article
Barvinchenko, V.N., Lipkovskaya, N.A. Adsorption of Natural 3-Phenylpropenic Acids on Highly Dispersed Alumina Surface. Colloid J 83, 183–188 (2021). https://doi.org/10.1134/S1061933X21020022
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X21020022