Abstract
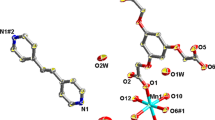
New Mn(III) complexes with Schiff bases and dicyanamide are synthesized: [Mn(Salpn)N(CN)2] n (two polymorphous modifications, Ia and Ib), {[Mn(5-BrSalen)N(CN)2] · CH3OH} n (II), and [Mn(3-MeOSalen)N(CN)2(H2O)] (III), where SalpnH2 = N,N′-bis(salicylidene)-1,3-diaminopropane, 5-BrSalenH2 = N,N′-bis(5-bromosalicylidene)-1,2-diaminoethane, and 3-MeOSalenH2 = N,N′-bis(3-methoxysalicylidene)-1,2-diaminoethane. Complexes Ia, Ib, and II have the polymer structure in which the dicyanamide anion binds the paramagnetic Mn(III) complexes with the Schiff bases into one-dimensional chains. Unlike them, in complex III the monomer units containing water and the dicyanamide anion as terminal ligands form dimers due to hydrogen bonds. The study of the magnetic properties of complexes Ia and II shows a weak antiferromagnetic interaction between the Mn3+ ions through the dicyanamide bridges in these complexes.
Similar content being viewed by others
References
Ashmawy, F.M., McAuliffe, C.A., (Dick) Parish, R.V., and Tames, J., Dalton Trans., 1985, no. 7, p. 1391.
Hulme, Ch.E., Watkinson, M., Haynes, M., et al., Dalton Trans., 1997, no. 11, p. 1805.
Sakamoto, F., Sumiya, T., Fujita, M., et al., Chem. Lett., 1998, vol. 27, no. 11, p. 1127.
Garcia-Deibe, A., Sousa, A., Bermejo, M.R., et al., Chem. Commun., 1991, no. 8, p. 728.
Miyasaka, H., Clerac, R., Ishii, T., et al., Dalton Trans., 2002, no. 7, p. 1528.
Lecren, L., Wernsdorfer, W., Lindigni, Y.-G., et al., J. Am. Chem. Soc., 2007, vol. 129, no. 16, p. 5045.
Shyu, H.-L., Wei, H.-H., and Wang, Yu., Inorg. Chim. Acta, 1999, vol. 290, no. 1, p. 8.
Reddy, K.R., Rajasekharan, M.V., and Tuchagues, J.-P., Inorg. Chem., 1998, vol. 37, no. 23, p. 5978.
Li, H., Zhong, Zh.J., Duan, Ch.-Y., et al., Inorg. Chim. Acta, 1998, vol. 271, nos. 1–2, p. 99.
Khalaji, A.D. and Ng, S.W., Russ. Chem. Coord. Chem., 2011, vol. 37, no. 1, p. 26.
Matsumoto, N., Sunatsuki, Y., Miyasaka, H., et al., Angew. Chem., Int. Ed. Engl., 1999, vol. 38, no. 12, p. 171.
Natase, S., Tuna, F., Maxim, C., et al., Cryst. Growth Des., 2007, vol. 7, no. 9, p. 1825.
Lu, Z., Yuan, M., Pan, F., et al., Inorg. Chem., 2006, vol. 45, no. 9, p. 3538.
Sailaja, S., Reddy, K.R., Rajasekharan, M.V., et al., Inorg. Chem., 2003, vol. 42, no. 1, p. 180.
Choi, H.-L., Sokol, J.J., and Long, J.R., Inorg. Chem., 2004, vol. 43, no. 5, p. 1606.
Zhou, H.-B., Wang, J., Wang, H.-Sh., et al., Inorg. Chem., 2011, vol. 50, no. 15, p. 6868.
Ababei, R., Li, Y.-G., Roubeau, O., et al., New J. Chem., 2009, vol. 33, no. 6, p. 1237.
Ferbinteanu, M., Miyasaka, H., Wernsdorfer, W., et al., J. Am. Chem. Soc., 2005, vol. 127, no. 9, p. 3090.
Pan, F., Wang, Z.-M., and Gao, S., Inorg. Chem., 2007, vol. 46, no. 24, p. 10221.
Sun, H.-L., Wang, Z.-M., and Gao, S., Coord. Chem. Rev., 2010, vol. 254, nos. 1–2, p. 1081.
Manson, J.L., Kmety, C.R., Huang, Q.Z., et al., Chem. Mater., 1998, vol. 10, no. 9, p. 2552.
Kurmoo, M. and Kepert, C., New J. Chem., 1998, vol. 22, no. 12, p. 1515.
Batten, S.R., Jensen, P., Kepert, C.J., et al., Dalton Trans., 1999, no. 24, p. 2987.
Van der Werff, P., Batten, S.R., Jensen, P., et al., Polyhedron, 2001, vol. 20, nos. 11–14, p. 1129.
Van der Werff, P.M., Batten, S.R., Jensen, P., et al., Cryst. Growth Des., 2004, vol. 4, no. 3, p. 503.
Schlueter, J.A., Manson, J.L., and Geiser, U., Inorg. Chem., 2005, vol. 44, no. 9, p. 3194.
Shi, Q., Cao, R., Li, X., et al., New J. Chem., 2002, vol. 26, no. 10, p. 1397.
Price, D.J., Batten, S.R., Moubaraki, B., and Murray, K.S., Indian J. Chem., Sect. A: Inorg., Bioinorg., Phys., Theor. Anal. Chem., 2003, vol. 42, p. 2256.
Batten, S.R., Jensen, P., Moubaraki, B., et al., Chem. Commun., 1998, no. 3, p. 439.
Kmety, C.R.Q., Huang, Q., Jeffrey, W., et al., Phys. Rev., 2000, vol. 62, no. 9, p. 5576.
Nakamoto, K., Infrared Spectra and Raman Spectra of Inorganic and Coordination Compounds, New York: Wiley, 1986.
CrysAlisPro. Agilent Technologies. Version 1.171.35.19. 2011.
Burla, M.C., Caliandro, R., Camalli, M., et al., J. Appl. Crystallogr., 2005, vol. 38, p. 381.
Palatinus, L. and Chapuis, G., J. Appl. Crystallogr., 2007, vol. 40, p. 786.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.A. Kopotkov, S.V. Simonov, A.V. Sadakov, E.B. Yagubskii, 2013, published in Koordinatsionnaya Khimiya, 2013, Vol. 39, No. 2, pp. 112–119.
Rights and permissions
About this article
Cite this article
Kopotkov, V.A., Simonov, S.V., Sadakov, A.V. et al. Manganese(III) complexes with tetradentate (N2O2) Schiff bases and dicyanamide. Russ J Coord Chem 39, 201–208 (2013). https://doi.org/10.1134/S107032841302005X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107032841302005X