Abstract
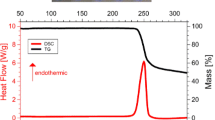
Rates of thermal decomposition of glycine, alanine, and serine are described by the equation of first order reaction in the temperature range 200–300°C. Apparent rate constants and apparent activation energies of decomposition of α-amino acids were evaluated. It was found that the main gaseos reaction product is carbon dioxide.
Similar content being viewed by others
References
Yunusov, D.Kh., Beglov, B.M., Zakirov, B.S., Milova, D.A., and Narkulov, A.N., USSR Author’s Certificate no. 1244136, 1984; Byull. Izobret., 1986, no. 326.
UK Patent no. 8631038, 1986; Ref. Zh. Khim., 1989, 7N191.
Food Process (USA), 1983, vol. 44, no. 7, p. 90; Ref. Zh. Khim., 1984, 1P368.
Skoblik, T.I., Markin, A.Yu., Zaborina, T.G., Fillipovich, E.I., Zheltuzhina, G.A., Evstigneeva, R.P., Babizhaeva, M.A., and Sharkov, V.Ya., Russian Patent no. 2030422, 1995, 4817453/04.
Tyukavkina, R.A. and Baukov, Yu.I., Bioorganicheskaya khimiya (Bioorganic Chemistry), Moscow: Drofa, 2005, p. 542.
Rabinovich, V.A. and Khavin, Z.Ya., Kratkii khimicheskii spravochnik (Brief Chemical Handbook), Leningrad: Khimiya, 1991.
Yablokov, V.A., Smeltsova, I.L., Zelyaev, I.A., and Mitrofanova, S.V., Privolzhskii Nauchn. Zh., 2007, no. 4, p. 115.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.Ya. Yablokov, I.L. Smel’tsova, I.A. Zelyaev, S.V. Mitrofanova, 2009, published in Zhurnal Obshchei Khimii, 2009, Vol. 79, No. 8, pp. 1344–1346.
Rights and permissions
About this article
Cite this article
Yablokov, V.Y., Smel’tsova, I.L., Zelyaev, I.A. et al. Studies of the rates of thermal decomposition of glycine, alanine, and serine. Russ J Gen Chem 79, 1704–1706 (2009). https://doi.org/10.1134/S1070363209080209
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363209080209