Abstract
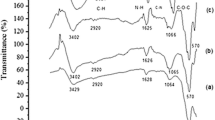
Enzyme immobilization is an operative method for improving enzyme properties. There are various supports to enzyme immobilization. In the present study chitosan nanoparticles have been applied to alkaline phosphatase (ALP) immobilize due to their special properties. Firstly, the Ionic Gelation method was used to synthesize Chitosan nanoparticles, and were characterized using XRD and Zetasizer Nano. Then, chitosan nanoparticles were activated with 1-ethyl-3-(3-dimethylaminopropyl carbodiimide (EDC) and ALP was such- cessfully immobilized on them. The influences of time, pH, and concentration of enzyme, and EDC on the efficiency of immobilization have been investigated. The stability studied against organic solvents and high temperature, which has indicated the stability of the immobilized enzyme, was increased during the immobilization. The surface topography of the bare and immobilized enzyme nanoparticles was studied by AFM. The calculation of binding efficiency showed that the immobilized ALP has conserved 71% of its native activity. The improvement of the enzyme stability by immobilization has indicated that chitosan nanoparticles were suitable support for immobilization.
Similar content being viewed by others
References
Muginova, S.V., Zhavoronkova, A.M., Polyakov, A.E., and Shekhovtsova, T.N., Anal. Sci., 2007, vol. 23, pp. 357–63.
Joseph, E.C., Annu Rev. Biophys. Biomol. Struct., 1992, vol. 21, pp. 441–483.
Montornes, A.E. and Osullivan, J.M., J. Electroanal. Chem., 2006, vol. 587, pp. 140–147.
Kreuzer, M.P., Sullivan, C.K., and Guilbault G.G., Analytica Chimica Acta, 1999, vol. 393, pp. 95–102.
Rankin, S.A., Christiansen, A., Lee, W., Banavara, D.S., Lopez-Hernandez, A., J. Dairy Sci., 2010, vol. 93, pp. 5538–51.
Soares, C.F., Fonseca, L.M., Leite, M.O., and Oliveira, M.C.P.P., Arq. Bras. Med. Vet. Zoo, 2010, pp. 1223–1230.
Martin, D., Linxweiler, W., Tanzer, D., Vormbrock, R., Olt, R., Kiesner, C., and Meisel, H., Deut Lebensm Rundsch, 2005, vol. 101, pp. 281–286.
Rittie, L. and Perbal, B., J. Cell Commun. Signal, vol. 2, pp. 25–45.
Mateo, C., Palomo, J.M., Fernandez-Lorente, G, Guisan, J.M., and Fernandez-Lafuente, R., Enzyme Microbiol. Technol., 2007, vol. 40, pp. 1451–1463.
Nisha, S., Karthick, A., and Gobi, N., Chem. Sci. Rev. Lett., 2012, vol. 1, pp. 148–155.
Kuhlmeyer, C. and Klein, J., Enzyme Microbiol. Technol., 2003, vol. 32, pp. 99–106.
Gomez, J.L., Bodalo, A., Gomez, E., Bastida, J., Hidalgo, A.M., and Gomez, M., Enzyme Microbial Technol., 2006, vol. 39, pp. 1016–1022.
Temocin, Z. and Yigitoglu, M., Bioprocess Biosyst. Eng., 2008. vol. 32, pp. 467–474.
Liu, W., Wang, L., and Jiang, R., Top. Catal., 2012, vol. 55, pp. 1146–1156.
Sorlier, P., Denuziere. A., Viton, C., and Domard, A., Biomacromol., 2001. vol. 2, pp. 765–772.
Yang, K., Xu, N.S., and Su, W.W., J. Biotechnol., 2010, vol. 148, pp. 119–127.
Wang, J.J., Zeng, Z.W., Xiao, R.Z., Xie, T., Zhou, G.L., Zhan, X.R., and Wang S.L., Int. J. Nanomedicine, vol. 6, pp. 765–774.
Grenha, A., Seijo, B., and Remunan-Lopez, C., Eur. J. Pharmaceutical Sci., 2005, vol. 25, pp. 427–437.
Kong, M., Chen, X.G., Xing, K., and Park, H.J., Int. J. Food Microbial., 2010, vol. 144, pp. 51–63.
RaviKumar, M.N.V., React. Funct. Polym., 2000, vol. 46, pp. 1–27.
Kim, I.Y., Seo, S.J., Moon, H.S., Yoo, M.K., Park, I.Y., Kim, B.C., and Cho, C.S., Biotechnol. Adv., 2008, vol. 26, pp. 1–21.
Venkatesan, J. and Kim, S.K., Mar. Drugs, 2010, vol. 8, pp. 2252–2266.
Xing, L., Lie, M., Zhengwei, M., and Changyou, G., Adv. Polym. Sci., 2011. vol. 244, pp. 81–128.
Rodrigues, S., Dionisio, M., Lopez, C.R., and Grenha, A., J. Funct. Biomater., 2011. vol. 3, pp. 615–641.
Jayakumar, R., Menon, D., Manzoor, K., Nair, S.V., and Tamura, H., Carbohydrate Polym., 2010, vol. 82, pp. 227–232.
Krajewska, B., Enzyme Microbial. Technol., 2004, vol. 35, pp. 126–139.
Nakorn, P., J. Metals Mater. Minerals, 2008, vol. 18, pp. 73–77.
Tang, Z.X., Qian, J.Q., and Shi L.E., Process Biochem., 2006, vol. 41, pp. 1193–1197.
Bindhu, L.V. and Abraham, E.T., J. Appl. Polymer Sci., 2003, vol. 88, pp. 1456–1464.
Fernandes, K.F., Lima, C.S., Pinho, H., and Collins, C.H., Process Biochem., 2003, vol. 38, pp. 1379–138.
Vaezifar, S., Golozar, M.A., Morshed, M., Razavi, S.H., Karbasi, S., and Kamali, M., J. Clust. Sci., vol. 24, 2013, pp. 891–903.
Zubriene, A., Budriene, S., Lubiene, J., and Dienys, G., Biocatal. Biotransform., 2002, vol. 20, pp. 423–427.
Hirano S, Miur O., Biotechnol. Bioeng, 1979, vol. 21, pp. 711–714.
Zubriene, A., Budriene, S., Gorochovceva, N., Romaskevic, T., Matulionis, E., and Dienys, G., Chemija, 2003, vol. 14, pp. 226–230.
Zhang, P. and Tan, W., J. Analytic Chem., 2001, vol. 369, pp. 302–307.
Masato, T, Akihiro, O, Yoshihisa, Y, and Masashi, K., J. Electroanal. Chem., 2003, vol. 566, pp. 323–329.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Jafary, F., Varshosaz, J., Panjehpour, M. et al. Immobilization of alkaline phosphatase using chitosan nanoparticles. Russ J Appl Chem 88, 891–897 (2015). https://doi.org/10.1134/S1070427215050262
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427215050262