Abstract—
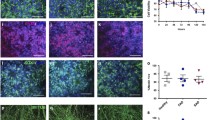
Neurogenesis is a complex process which governs embryonic brain development and is important for brain plasticity throughout the whole life. Postnatal neurogenesis occurs in neurogenic niches that regulate the processes of proliferation and differentiation of stem and progenitor cells driven by stimuli triggering mechanisms of neuroplasticity. In the neurogenic niches cells of glial and endothelial origin are the key regulators of neurogenesis. It is known that physiological neurogeneses is crucial for memory formation, whereas reparative neurogenesis provides partial repair of injured brain structures and compensation of neurological deficits caused by the brain injury. Dysregulation of neurogenesis is a characteristics feature of various neurodevelopmental and neurodegenerative diseases, particularly, Alzheimer’s disease representing a very important medical and social problem. In the in vitro model of the neurogenic niche using hippocampal neurospheres as a source of stem/progenitor cells and astrocytes, we have studied effects of astrocyte activation on the expression of markers of different stages of cell proliferation and differentiation. We found that aberrant mechanisms of development of stem and progenitor cells, caused by the beta-amyloid peptide (Аβ1–42), can be partially restored by targeted activation of GFAP-expressing cells in the neurogenic niche.







Similar content being viewed by others
REFERENCES
Lane, C.A., Hardy, J., and Schott, J.M., Eur. J. Neurol., 2018, vol. 25, no. 1, pp. 59−70. https://doi.org/10.1111/ene.13439
Rodríguez, J.J. and Verkhratsky, A., J. Anat., 2011, vol. 219, no. 1, pp. 78−89.
Hollands, C., Bartolotti, N., and Lazarov, O., Front. Neurosci., 2016, vol. 10, 178. https://doi.org/10.3389/fnins.2016.00178
Komleva, Y.K., Kuvacheva, N.V., Malinovskaya, N.A., Gorina, Ya.V., Lopatina, O.L., Teplyashina, E.A., Pozhilenkova, E.A., Zamay, A.S., Morgun, A.V., and Salmina, A.B., Hum. Physiol., 2016, vol. 42, no. 8, pp. 865−873.
Lin, R., Cai, J., Nathan, C., Wei, X., Schleidt, S., Rosenwasser, R., and Iacovitti, L., Neurobiol. Dis., 2015, vol. 74, pp. 229−239. https://doi.org/10.1016/j.nbd.2014.11.016
Kerr, A.L., Steuer, E.L., Pochtarev, V., and Swain, R.A., Neuroscience, 2010, vol. 171, no. 1, pp. 214−226.
Pozhilenkova, E.A., Lopatina, O.L., Komleva, Y.K., Salmin, V.V., and Salmina, A.B., Rev. Neurosci., 2017, vol. 28, no. 4, pp. 397−415. https://doi.org/10.1515/revneuro-2016-0071
Luo, L., Guo, K., Fan, W., Lu, Y., Chen, L., Wang, Y., Shao, Y., Wu, G., Xu, J., and Lü, L., Exp. Ther. Med., 2017, vol. 13, no. 2, pp. 645−650. https://doi.org/10.3892/etm.2016.4016
Boitsova, E.B., Morgun, A.V., Osipova, E.D., Pozhilenkova, E.A., Martinova, G.P., Frolova, O.V., Olovannikova, R.Y., Tohidpour, A., Gorina, Y.V., Panina, Y.A., and Salmina, A.B., J. Neuroinflammation, 2018, vol. 15, no. 1, 196. https://doi.org/10.1186/s12974-018-1233-2
Khilazheva, E.D., Boitsova, E.B., Pozhilenkova, E.A., Solonchuk, Yu.R., and Salmina, A.B., Tsitologiya, 2015, vol. 57, no. 10, pp. 710–713.
Ehret, F., Vogler, S., and Kempermann, G., Stem Cell Res., 2015, vol. 15, no. 3, pp. 514−521. https://doi.org/10.1016/j.scr.2015.09.010
Salmin, V.V., Komleva, Y.K., Kuvacheva, N.V., Morgun, A.V., Khilazheva, E.D., Lopatina, O.L., Pozhilenkova, E.A., Shapovalov, K.A., Uspen-skaya, Y.A., and Salmina, A.B., Front. Aging Neurosci., 2017, vol. 9, 245. https://doi.org/10.3389/fnagi.2017.00245
Khan, J., Das, G., Gupta, V., Mohapatra, S., Ghosh, S., and Ghosh, S., ACS Chem. Neurosci., 2018, vol. 9, no. 11, pp. 2870−2878. https://doi.org/10.1021/acschemneuro.8b00414
Beccari, S., Valero, J., Maletic-Savatic, M., and Sierra, A., Sci. Rep., 2017, vol. 7, no. 1, 16528. https://doi.org/10.1038/s41598-017-16466-3
Falk, S. and Götz, M., Curr. Opin. Neurobiol., 2017, vol. 47, pp. 188−195. https://doi.org/10.1016/j.conb.2017.10.025
Wilhelmsson, U., Faiz, M., de Pablo, Y., Sjöqvist, M., Andersson, D., Widestrand, A., Potokar, M., Stenovec, M., Smith, P.L., Shinjyo, N., Pekny, T., Zorec, R., Ståhlberg, A., Pekna, M., Sahlgren, C., and Pekny, M., Stem Cells., 2012, vol. 30, no. 10, pp. 2320−2329. https://doi.org/10.1002/stem.1196
Robinson, C., Apgar, C., and Shapiro, L.A., Neural Plasticity, 2017, vol. 26, no. 7, pp. 1314−1318. https://doi.org/10.1155/2016/1347987
Gourine, A.V., Kasymov, V., Marina, N., Tang, F., Figueiredo, M.F., Lane, S., Teschemacher, A.G., Spyer, K.M., Deisseroth, K., and Kasparov, S., Science, 2010, vol. 329, no. 5991, pp. 571−575. https://doi.org/10.1126/science.1190721
Figueiredo, M.F., Lane, S., Stout, R.F., Liu, B., Parpura, V., Teschemacher, A.G., and Kasparov, S., Cell Calcium, 2014, vol. 56, no. 3, pp. 208−214. https://doi.org/10.1016/j.ceca.2014.07.007
Liu, Y., Xue, Q., Tang, Q., Hou, M., Qi, H., Chen, G., Chen, W., Zhang, J., Chen, Y., and Xu, X., Microvasc. Res., 2013, vol. 90, pp. 199−205. https://doi.org/10.1016/j.mvr.2013.08.004
Scrace, S., O’Neill, E., Hammond, E.M., and Pires, I.M., Methods in Molecular Biology (Clifton, N.J.), 2013, vol. 1046, pp. 295−306. https://doi.org/10.1007/978-1-62703-538-5_17
Figueiredo, M., Figueiredo, M., Lane, S., Stout, R.F., Jr., Liu, B., Parpura, V., Teschema-cher, A.G., and Kasparov, S., Cell Calcium, 2014, vol. 56, no. 3, pp. 208−214. https://doi.org/10.1016/j.ceca.2014.07.007
Itokazu, Y. and Yu, R.K., Mol. Neurobiol., 2014, vol. 50, no. 1, pp. 186−196. https://doi.org/10.1007/s12035-014-8634-8
Hack, M., Sugimori, M., Lundberg, C., Nakafuku, M., and Götz, M., Mol. Cell. Neurosci., 2004, vol. 25, no. 4, pp. 664−678.
Sakurai, K. and Osumi, N., J. Neurosci., 2008, vol. 28, no. 18, pp. 4604−4612. https://doi.org/10.1523/JNEUROSCI.5074-07.2008
Gao, Z., Ure, K., Ables, J.L., Lagace, D.C., Nave, K.A., Goebbels, S., Eisch, A.J., and Hsieh, J., Nat. Neurosci., 2009, vol. 12, no. 9, pp. 1090–1092. https://doi.org/10.1038/nn.2385
Boutin, C., Hardt, O., de Chevigny, A., Coré, N., Goebbels, S., Seidenfaden, R., Bosio, A., and Cremeret, H., Proc. Natl. Acad. Sci. USA, 2010, vol. 107, no. 3, pp. 1201−1206. https://doi.org/10.1073/pnas.0909015107
Roybon, L., Hjalt, T., Stott, S., Guillemot, F., Li, J.Y., and Brundin, P., PLoS One, 2009, vol. 4, no. 3, e4779. https://doi.org/10.1371/journal.pone.0004779
Thakurela, S., Tiwari, N., Schick, S., Garding, A., Ivanek, R., Berninger, B., and Tiwari, V.K., Cell Discov., 2016, vol. 9, no. 2, 15045. https://doi.org/10.1038/celldisc.2015.45
Song, H., Stevens, C.F., and Gage, F.H., Nature, 2002, vol. 417, no. 6884, pp. 39−44.
Cao, X., Li, L.P., Qin, X.H., Li, S.J., Zhang, M., Wang, Q., Hu, H.H., Fang, Y.Y., Gao, Y.B., Li, X.W., Sun, L.R., Xiong, W.C., Gao, T.M., and Zhu, X.H., Stem Cells, 2013, vol. 31, no. 8, pp. 1633−1643. https://doi.org/10.1002/stem.1408
Mohn, T.C. and Koob, A.O., J. Exp. Neurosci., 2015, vol. 9, no. 2, pp. 25−34. https://doi.org/10.4137/JEN.S25520
ACKNOWLEDGMENTS
The authors thank Professor S. Kasparov (University of Bristol, UK) for providing adenoviral vectors.
Funding
This work was performed within the State Assignment of the Ministry of Public Health of the Russian Federation (2018–2020) “New Technologies for the Management of Neurogenesis and Angiogenesis in the Brain.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Animal experiments were carried out in accordance with generally accepted ethical international standards and the principles of humanity stated in the European Community Directive (2010/63/EC) and the requirements of the order of the Ministry of Public Health of the Russian Federation no. 267 of June 19, 2003 “On the Approval of Laboratory Practice in the Russian Federation.”
The authors declare that they have no conflict of interest.
Additional information
Translated by A. Medvedev
Rights and permissions
About this article
Cite this article
Morgun, A.V., Osipova, E.D., Boytsova, E.B. et al. Astrocyte-Mediated Regulation of Cell Development in the Model of Neurogenic Niche in Vitro Treated with Aβ1-42. Biochem. Moscow Suppl. Ser. B 14, 6–14 (2020). https://doi.org/10.1134/S1990750820010114
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990750820010114