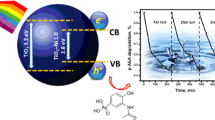
High band-gap energy and large contribution of Ti–O–Si linkage were found on the catalyst synthesized with acetyl acetone as a chelating agent in a sol–gel process. Meanwhile, when acetic acid as a chelating agent was employed, it promoted the formation of TiO2–SiO2 mixed oxide with relatively high specific surface area and high photocurrent density. Low charge transfer resistance as well as wide space charge region in TiO2–SiO2 particles prepared with acetic acid gave rise to high photocurrent density, which might result in the superior photocatalytic water decomposition for hydrogen production.
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Nguyen, TV., Choi, DJ. & Yang, OB. Effect of chelating agents on the properties of TiO2–SiO2 mixed oxide for photocatalytic water decomposition. Res Chem Intermediat 31, 483–491 (2005). https://doi.org/10.1163/1568567053956662
Published:
Issue Date:
DOI: https://doi.org/10.1163/1568567053956662