Abstract
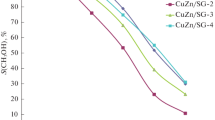
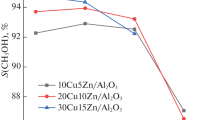
Methanol was synthesized from carbon dioxide and hydrogen over various supported copper catalysts. A support was selected from the viewpoint of its acid-base properties. Acid supports showed high selectivity but low activity. Neutral and basic supports produced only carbon monoxide. Amphoteric supports showed high activities. Among them, TiO2 supports suppressed the reverse water gas shift reaction (CO formation). The role of supports was discussed on the basis of in situ infrared results.
Similar content being viewed by others
References
T. Tagawa, G. Pleizier, and Y. Amenomiya, Applied Catalysis, 18, 285 (1985).
T. Tagawa and Y. Amenomiya, Applied Spectroscopy, 39, 358 (1985).
Y. Amenomiya and T. Tagawa, Proc. 8th Int. Congr. Catalysis, Berlin, vol.2, 557(1984).
T. Tagawa, N. Nomura, and S. Goto, Proc. Int. Conf. Carbon Dioxide Util., Bari, Italy, p.369, (1993).
Y. Amenomiya, Appl. Catal., 30, 57 (1987).
T. Tagawa, N. Chikamatsu, S. Goto and H. Imai, Appl. Catal., 61, 209 (1990).
H. Arakawa, H. Kusama, K. Sayama and K. Okabe, Proc. Int. Conf. Carbon Dioxide Util., Bari, Italy, p.95 (1993) and references therein.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tagawa, T., Nomura, N., Shimakage, M. et al. Effect of supports on copper Catalysts for Methanol Synthesis from CO2 + H2 . Res. Chem. Intermed. 21, 193–202 (1995). https://doi.org/10.1163/156856795X00170
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1163/156856795X00170