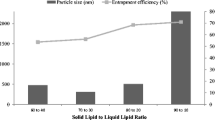
Abstract
Psoriasis is a chronic, autoimmune skin disease affecting approximately 2% of the world's population. Clobetasol propionate which is a superpotent topical corticosteroid is widely used for topical treatment of psoriasis. Conventional dosage forms like creams and ointments are commonly prefered for the therapy. The purpose of this study was to develop a new topical delivery system in order to provide the prolonged release of clobetasol propionate and to reduce systemic absorption and side effects of the drug. Clobetasol propionate loaded-poly(D,L-lactic-co-glycolic acid) (PLGA) microspheres were prepared by oil-in-water emulsion–solvent evaporation technique. Particle size analysis, morphological characterization, DSC and XRD analyses and in vitro drug release studies were performed on the microparticle formulations. Emulgel formulations were prepared as an alternative for topical delivery of clobetasol propionate. In vitro drug release studies were carried out from the emulgel formulations containing pure drug and drug-loaded microspheres. In addition, the same studies were performed to determine the drug release from the commercial cream product of clobetasol propionate. The release of clobetasol propionate from the emulgel formulations was significantly higher than the commercial product. In addition, the encapsulation of clobetasol propionate in the PLGA microspheres significantly delayed the drug release from the emulgel formulation. As a result, the decrease in the side effects of clobetasol propionate by the formulation containing PLGA microspheres is expected.








Similar content being viewed by others
REFERENCES
Bhosle MJ, Kulkarni A, Feldman SR, Balkrishnan R. Quality of life in patients with psoriasis. Health and Quality of Life Outcomes. 2006;4:35.
Fluhr JW, Cavallotti C, Berardesca E. Emollients, moisturizers and keratolytic agents in psoriasis. Clin Dermatol. 2008;26:380–6.
Lebwohl M, Ting PT, Koo JYM. Psoriasis treatment: traditional therapy. Ann Rheum Dis. 2005;64(Suppl II):ii83–6.
Van de Kerkhof PCM. The topical treatment of psoriasis. Clin Exp Dermatol. 2005;30:205–8.
Austad J, Bjerke JR, Gjertsen BT, Helland S, Livden JK, Morken T, et al. Clobetasol propionate followed by calcipotriol is superior to calcipotriol alone in topical treatment of psoriasis. JEADV. 1998;11:19–24.
Tsai J-C, Cheng C-L, Tsai Y-F, Sheu H-M, Chou C-H. Evaluation of in vivo bioequivalence methodology for topical clobetasol 17-propionate based on pharmacodynamic modeling using chinese skin. J Pharm Sci. 2004;93(1):207–17.
Fang J-Y, Fang C-L, Sung KC, Chen H-Y. Effect of low frequency ultrasound on the in vitro percutaneous absorption of clobetasol 17-propionate. Int J Pharm. 1999;191:33–42.
Hu FQ, Yuan H, Zhang HH, Fang M. Preparation of solid lipid nanoparticles with clobetasol propionate by a novel solvent diffusion method in aqueous system and physicochemical characterization. Int J Pharm. 2002;239:121–8.
Campisi G, Giandalia G, De Caro V, Di Liberto C, Arıcò P, Giannola LI. A new delivery system of clobetasol-17-propionate (lipid-loaded microspheres 0.025%) compared with a conventional formulation (lipophilic ointment in a hydrophilic phase 0.025%) in topical treatment of atrophic/erosive oral lichen planus. A phase IV, randomized, observer-blinded, parallel group clinical trial. Br J Dermatol. 2004;150:984–90.
Hu F-Q, Jiang S-P, Du Y-Z, Yuan H, Ye Y-Q, Zeng S. Preparation and characteristics of monostearin nanostructured lipid carriers. Int J Pharm. 2006;314:83–9.
Şenyiğit T, Sonvico F, Barbieri S, Özer Ö, Santi P, Colombo P. Lecithin/chitosan nanoparticles of clobetasol-17-propionate capable of accumulation in pig skin. J Cont Rel. 2010;142:368–73.
Ga De Jalón E, Blanco-Prieto MJ, Ygartua P, Santoyo S. PLGA microparticles: possible vehicles for topical drug delivery. Int J Pharm. 2001;226:181–4.
Haddadi A, Farboud ES, Erfan M, Aboofazeli R. Preparation and characterization of biodegradable urea-loaded microparticles as an approach for transdermal delivery. J Microencapsulation. 2006;23(6):698–712.
Jain RA. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials. 2000;21:2475–90.
Wang J, Ng CW, Win KY, Shoemakers P, Lee TKY, Feng SS, et al. Release of paclitaxel from polylactide-co-glycolide (PLGA) microparticles and discs under irradiation. J Microencapsulation. 2003;20(3):317–27.
Çırpanlı Y, Ünlü N, Çalış S, Hıncal AA. Formulation and in vitro characterization of retinoic acid loaded poly (lactic-co-glycolic acid) microspheres. J Microencapsulation. 2005;22(8):877–89.
Samatlı Y, Yüksel N, Tarımcı N. Preparation and characterization of poly(D, L-lactic-co-glycolic acid) microspheres containing flurbiprofen sodium. Drug Delivery. 2006;13:105–11.
Janoria KG, Mitra AK. Effect of lactide/glycolide ratio on the in vitro release of ganciclovir and its lipophilic prodrug (GCV-monobutyrate) from PLGA microspheres. Int J Pharm. 2007;338:133–41.
Ga De Jalón E, Blanco-Prieto MJ, Ygartua P, Santoyo S. Topical application of acyclovir loaded microparticles: quantification of the drug in porcine skin layers. J Cont Rel. 2001;75:191–7.
Santoyo S, Ga De Jalón E, Ygartua P, Renedo MJ, Blanco-Prieto MJ. Optimization of topical cidofovir penetration using microparticles. Int J Pharm. 2002;242:107–13.
Sánchez A, Vila-Jato JL, Alonso MJ. Development of biodegradable microspheres and nanospheres for the controlled release of cyclosporin A. Int J Pharm. 1993;99:263–73.
Ghaderi R, Sturesson C, Carlfors J. Effect of preparative parameters on the characteristics of poly(D, L-lactide-co-glycolide) microspheres made by the double emulsion method. Int J Pharm. 1996;141:205–16.
Sehra S, Dhake AS. Formulation and evaluation of sustained release microspheres of poly-lactide-co-glycolide containing tamoxifen citrate. J Microencapsulation. 2005;22(5):521–8.
Zhang H, Gao S. Temozolomide/PLGA microparticles and antitumor activity against glioma C6 cancer cells in vitro. Int J Pharm. 2007;329:122–8.
Gabor F, Ertl B, Wirth M, Mallinger R. Ketoprofen- poly(D, L-lactic-co-glycolic acid) microspheres: influence of manufacturing parameters and type of polymer on the release characteristics. J Microencapsulation. 1999;16(1):1–12.
Mao S, Xu J, Cai C, Germershaus O, Schaper A, Kissel T. Effect of WOW process parameters on morphology and burst release of FITC-dextran loaded PLGA microspheres. Int J Pharm. 2007;334:137–48.
Freitas S, Merkle HP, Gander B. Microencapsulation by solvent extraction/evaporation: reviewing the state of the art of microsphere preparation process technology. J Cont Rel. 2005;102:313–32.
Yang Y-Y, Chung T-S, Ng NP. Morphology, drug distribution, and in vitro release profiles of biodegradable polymeric microspheres containing protein fabricated by double-emulsion solvent extraction/evaporation method. Biomaterials. 2001;22:231–41.
Schlicher EJAM, Postma NS, Zuidema J, Talsma H, Hennink WE. Preparation and characterization of poly(D, L-lactic-co-glycolic acid) microspheres containing desferrioxamine. Int J Pharm. 1997;153:235–45.
Arıca B, Kaş HS, Orman MN, Hıncal AA. Biodegredable bromocryptine mesylate microspheres prepared by a solvent evaporation technique. I: evaluation of formulation variables on microspheres characteristics for brain delivery. J Microencapsulation. 2002;19(4):473–84.
Choi HS, Seo S-A, Khang G, Rhee JM, Lee HB. Preparation and characterization of fentanyl-loaded PLGA microspheres: in vitro release profiles. Int J Pharm. 2002;234:195–203.
Jagadeesh HG, Devi VK. Tamoxifen loaded poly (ε-caprolactone) based injectable microspheres for breast cancer. Int J Pharm Pharmaceut Sci. 2010;2(4):189–95.
Fernandez-Carballido A, Herrero-Vanrell R, Molina-Martinez IT, Pastoriza P. Biodegradable ibuprofen-loaded PLGA microspheres for intraarticular administration: effect of labrafil addition on release in vitro. Int J Pharm. 2004;279:33–41.
Mahajan HS, Gattani SG. Gellan gum based microparticles of metoclopromide hydrochloride for intranasal delivery: development and evaluation. Chem Pharm Bull. 2009;57(4):388–92.
Barakat NS, Radwan MA. In vitro performance of carbamazepine loaded to various molecular weights of poly(D, L-lactide-co-glycolide). Drug Delivery. 2006;13:9–18.
Heya T, Okada H, Ogawa Y, Toguchi H. Factors influencing the profiles of TRH release from copoly(d, l-lactic/glycolic acid) microspheres. Int J Pharm. 1991;72:199–205.
Zolnik BS, Leary PE, Burgess DJ. Elevated temperature accelerated release testing of PLGA microspheres. J Cont Rel. 2006;112:293–300.
Bezemer JM, Radersma R, Grijpma DW, Dijkstra PJ, Van Blitterswijk CA, Feijen J. Microspheres for protein delivery prepared from amphiphilic multiblock copolymers 2. modulation of release rate. J Cont Rel. 2000;67:249–60.
Fu X, Ping Q, Gao Y. Effects of formulation factors on encapsulation efficiency and release behaviour in vitro of huperzine A-PLGA microspheres. J Microencapsulation. 2005;22(7):705–14.
Klose D, Siepmann F, Elkharraz K, Krenzlin S, Siepmann J. How porosity and size affect the drug release mechanisms from PLGA-based microparticles. Int J Pharm. 2006;314:198–206.
Mao S, Shi Y, Li L, Xu J, Schaper A, Kissel T. Effects of process and formulation parameters on characteristics and internal morphology of poly(D, L-lactide-co-glycolide) microspheres formed by the solvent evaporation method. Eur J Pharm Biopharm. 2008;68:214–23.
Yen S-Y, Sung KC, Wang J-J, Hu OY-P. Controlled release of nalbuphine propionate from biodegradable microspheres: in vitro and in vivo studies. Int J Pharm. 2001;220:91–9.
DECLARATION OF INTEREST
This study was supported by the Management of Scientific Research Projects of Ankara University (Project Number: 07B3336002). The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
ANSWERS TO REVIEWER 3
1. The use of English in this manuscript was thoroughly checked by a native speaker.
2. The explanation (“The different thermodynamic activities between formulations could also have accounted for these results”) was added into the “RESULTS AND DISCUSSION” section on the advice of the reviewer.
Rights and permissions
About this article
Cite this article
Badıllı, U., Şen, T. & Tarımcı, N. Microparticulate Based Topical Delivery System of Clobetasol Propionate. AAPS PharmSciTech 12, 949–957 (2011). https://doi.org/10.1208/s12249-011-9661-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9661-7