Abstract
Background
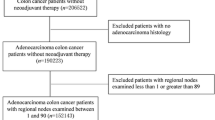
The most important prognostic factor in colonic cancer is the presence or absence of regional lymph nodes metastases. The aim of this study was to evaluate the relationship between 5-year mortality in the New Zealand population, and the number of nodes examined in Stage II and III colon cancers.
Methods
New Zealand Cancer Registry data were retrieved for patients with colonic cancer from January 1995 to July 2003. Patients with incomplete entries, Stage I tumors, and distant metastases were excluded from analysis. Univariate and Cox regression models were used with 5-year mortality as the primary endpoint.
Results
The study identified 4309 patients. Younger age, female gender, Pacific Island descent, and right-sided tumors were associated with significantly higher lymph node retrieval. Cox regression analysis showed that the number of nodes examined was a significant predictor of 5-year mortality when age, sex, ethnicity, and site were controlled for. Five-year survival consistently improved between nodal strata until the 16-node mark, above which survival advantage was minimal. For Stage III cancers, a higher lymph node ratio was associated with a significant increase in mortality.
Conclusions
Increased rates of nodal examination are associated with a significantly lower 5-year mortality for Stage II and III colonic cancer, but this survival advantage appears to be minimal after the 16-node mark. The lymph node ratio has been validated as a powerful predictor of survival in Stage III cancer. Our results support the current practice of harvesting and examining as many nodes as possible during attempted curative resection.








Similar content being viewed by others
References
New Zealand Health Information Service. Cancer: New Registrations and Deaths: 1993. Wellington: Ministry of Health, 1997.
Connolly AB, Frizelle FA, Keating JP, Lynch AC, Thompson-Fawcett MW, Robertson GM. Review of developments in colorectal surgery in New Zealand. N Z Med J. 2002;115:262–5.
Keating J, Pater P, Lolohea S, Wickremesekera K. The epidemiology of colorectal cancer: what can we learn from the New Zealand Cancer Registry? N Z Med J. 2003;116:U437.
Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99:433–41.
Swanson RS, Compton CC, Stewart AK, Bland KI. The prognosis of T3N0 colon cancer is dependent on the number of lymph nodes examined. Ann Surg Oncol. 2003;10:65–71.
Stage III. The classification of cancer of the rectum. J Pathol. 1932;35:323–32.
Simpson WC, Mayo CW. The mural penetration of the carcinoma cell in the colon: anatomic and clinical study. Surg Gynecol Obstet. 1939;68:872–7.
Beart RW, Steele GD, Menck HR, et al. Management and survival of patients with adenocarcinoma of the colon and rectum: a national survey of the Commission on Cancer. J Am Coll Surg. 1995;181:225–36.
Vather R, Sammour T, Zargar K, Metcalf PA, Connolly AB, Hill AG. Lymph node examination as a predictor of long-term outcome in Dukes B colon cancer. Int J Colorectal Dis. 2008 Aug 21 [Epub ahead of print].
Goldstein NS, Sanford W, Coffey M, Layfield LJ. Lymph node recovery from colorectal resection specimens removed for adenocarcinoma. Trends over time and a recommendation for a minimum number of lymph nodes to be recovered. Am J Clin Pathol. 1996;106:209–16.
Hernanz F, Revuelta S, Redondo C, Madrazo C, Castillo J, Gomez-Fleitas M. Colorectal adenocarcinoma: quality of the assessment of lymph node metastases. Dis Colon Rectum. 1994;37:373–7.
Wong JH, Severino R, Honnebier MB, Tom P, Namiki TS. Number of nodes examined and staging accuracy in colorectal carcinoma. J Clin Oncol. 1999;17:2896–900.
Wong S, Ji H, Hollenbeck B, Morris A, Baser O. Hospital lymph node examination rates and survival after resection for colon cancer. JAMA. 2007;298:2149–54.
Fielding LP, Arsenault PA, Chapuis PH, Dent O, Gathright B, Hardcastle JD, et al. Clinicopathological staging for colorectal cancer: an International Documentation System (IDS) and an International Comprehensive Anatomical Terminology (ICAT). J Gastroenterol Hepatol. 1991;6:325–44.
National Quality Forum. Specifications of the national voluntary consensus standards for breast and colon cancer. http://www.qualityforum.org/pdf/cancer/txbreast-colonAppA-Specsvoting01-18-07clean.pdf. Accessed 18 Jan 2007.
Sigurdson ER. Lymph node dissection: is it diagnostic or therapeutic? J Clin Oncol. 2003;21:965–7.
Baxter NN, Virnig DJ, Rothenberger DA, Morris AM, Jessurun J, Virnig BA. Lymph node evaluation in colorectal cancer patients: a population-based study. J Natl Cancer Inst. 2005;97:219–25.
Feinstein AR, Sosin DM, Wells CK. The Will Rogers phenomenon: stage migration and new diagnostic techniques as a source of misleading statistics for survival in cancer. N Engl J Med. 1985;312:1604–8.
Berger AC, Sigurdson ER, LeVoyer T, et al. Colon cancer survival is associated with decreasing ratio of metastatic to examined lymph nodes. J Clin Oncol. 2005;23:8706–12.
De Ridder M, Vinh-Hung V, Van Nieuwenhove Y, et al. Prognostic value of the lymph node ratio in node positive colon cancer. Gut. 2006;55:1681.
Edler D, Ohrling K, Hallström M, Karlberg M, Ragnhammar P. The number of analyzed lymph nodes – a prognostic factor in colorectal cancer. Acta Oncol. 2007;46:975–81.
Wang J, Hassett JM, Dayton MT, Kulaylat MN. Lymph node ratio: role in the staging of node-positive colon cancer. Ann Surg Oncol. 2008;15:1600–8.
Ko CY, Chang JT, Chaudhry S, Kominski G. Are high-volume surgeons and hospitals the most important predictors of in-hospital outcome for colon cancer resection? Surgery. 2002;132:268–73.
Panageas KS, Schrag D, Riedel E, Bach PB, Begg CB. The effect of clustering of outcomes on the association of procedure volume and surgical outcomes. Ann Intern Med. 2003;139:658–65.
Bui L, Rempel E, Reeson D, Simunovic M. Lymph node counts, rates of positive lymph nodes, and patient survival for colon cancer surgery in Ontario, Canada: a population-based study. J Surg Oncol. 2006;93:439–45.
Wright FC, Law CH, Last L, Khalifa M, Arnaout A, Naseer Z, et al. Lymph node retrieval and assessment in stage II colorectal cancer: a population-based study. Ann Surg Oncol. 2003;10:903–9.
Evans MD, Barton K, Rees A, Stamatakis JD, Karandikar SS. The impact of surgeon and pathologist on lymph node retrieval in colorectal cancer and its impact on survival for patients with Dukes’ stage B disease. Colorectal Dis. 2008;10:157–64.
Ratto C, Sofo L, Ippoliti M, Merico M, Bossola M, Vecchio FM, et al. Accurate lymph-node detection in colorectal specimens resected for cancer is of prognostic significance. Dis Colon Rectum. 1999;42:143–58.
Johnson PM, Malatjalian D, Porter GA. Adequacy of nodal harvest in colorectal cancer: a consecutive cohort study. J Gastrointest Surg. 2002;6:883–8.
Hermanek P, Giedl J, Dworak O. Two programs for examination of regional lymph nodes in colorectal carcinoma with regard to the new pN classification. Path Res Pract. 1989;185:867–73.
Calaluce R, Miedema BW, Yesus YW. Micrometastasis in colorectal carcinoma: a review. J Surg Oncol. 1998;67:194–202.
Compton CC, Fielding LP, Burgart LJ, Conley B, Cooper HS, Hamilton SR, et al. Prognostic factors in colorectal cancer: College of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med. 2000;124:979–94.
Sarli L, Bader G, Iusco D, Salvemini C, Mauro DD, Mazzeo A, et al. Number of lymph nodes examined and prognosis of TNM stage II colorectal cancer. Eur J Cancer. 2005;41:272–9.
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Appendix 1
Results of the Cox regression analysis
Regression coefficient (95% CI) | p value | |
|---|---|---|
Age | 1.026 (1.022–1.031) | <.0001 |
Gender | 0.863 (0.794–0.938) | .001 |
Ethnicity | ||
Pacific Islander | <.0001 | |
Asian | 0.527 (0.290–0.959) | .036 |
European | 0.716 (0.465–1.102) | .129 |
Maori | 0.896 (0.548–1.465) | .662 |
Other/not otherwise specified | 0.243 (0.122–0.485) | <.0001 |
Tumor site | ||
Transverse colon | .001 | |
Ascending colon | 0.967 (0.839–1.115) | .648 |
Descending colon | 0.827 (0.715–0.957) | .011 |
Not otherwise specified | 1.183 (0.842–1.663) | .333 |
Number of nodes | ||
>25 | <0.0001 | |
1–4 | 1.659 (1.376–2.000) | <0.0001 |
5–8 | 1.443 (1.229–1.694) | <0.0001 |
9–12 | 1.310 (1.118–1.535) | 0.001 |
13–16 | 1.063 (0.893–1.265) | 0.491 |
17–20 | 1.030 (0.845–1.256) | 0.769 |
21–24 | 1.103 (0.892–1.364) | 0.366 |
Rights and permissions
About this article
Cite this article
Vather, R., Sammour, T., Kahokehr, A. et al. Lymph Node Evaluation and Long-Term Survival in Stage II and Stage III Colon Cancer: A National Study. Ann Surg Oncol 16, 585–593 (2009). https://doi.org/10.1245/s10434-008-0265-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-008-0265-8