Abstract
Background
To address the clinical relevance of molecular detection of occult breast cancer in sentinel lymph nodes and nonsentinel axillary lymph nodes (ALN), we initiated the Minimally Invasive Molecular Staging of Breast Cancer (MIMS) trial, a multi-institutional prospective cohort study. This trial represents the first prospective cohort study in which a multimarker, real-time reverse transcription polymerase chain reaction (RT-PCR) analysis was applied to the detection of breast cancer micrometastases in ALN.
Materials and Methods
Sentinel and/or nonsentinel ALN from 501 breast cancer subjects with T1–T3 primary tumors were analyzed by standard histopathology and multimarker, real-time RT-PCR analysis. Seven breast cancer-associated genes (mam, mamB, PIP, CK19, muc1, PSE, and CEA) known to be overexpressed in metastatic breast cancer compared with control lymph nodes were used. Follow-up data were collected for 5 years.
Results
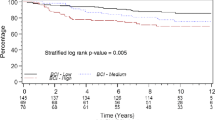
Of the 501 breast cancer subjects enrolled, 348 were node negative and completed the 5-year follow-up. Of these patients (n = 94), 27% demonstrated evidence of molecular overexpression. The 5-year relapse-free survival rate was 95.4% (95% confidence interval [95% CI], 92.4–97.2%). No single gene or combination of study genes was predictive of recurrence.
Conclusions
The genes in this study panel failed to be predictive of clinical relapse. This may be a function of several factors: the low event rate at 5 years, the particular gene set, the methodology used for detection/analysis or that our original hypothesis was wrong and that the presence of positive marker signal by real-time RT-PCR is not associated with a worsened clinical outcome.



Similar content being viewed by others
References
Donegan WL. Tumor-related prognostic factors for breast cancer. CA Cancer J Clin. 1997;47:28–51.
Goldhirsch A, Glick JH, Gelber RD, Coates AS, Senn HJ. Meeting highlights: International Consensus Panel on the Treatment of Primary Breast Cancer. Seventh international conference on adjuvant therapy of primary breast cancer. J Clin Oncol. 2001;19:3817–27.
Woo CS, Silberman H, Nakamura SK, Ye W, Sposto R, Colburn W, et al. Lymph node status combined with lymphovascular invasion creates a more powerful tool for predicting outcome in patients with invasive breast cancer. Am J Surg. 2002;284:337–40.
Cummings MC, Walsh MD, Hohn BG, Bennett IC, Wright RG, McGuckin MA. Occult axillary lymph node metastases in breast cancer do matter: results of 10-year survival analysis. Am J Surg Pathol. 2002;26:1286–95.
Rosen PP, Saigo P, Jr. OB, Weathers E, DePalo A. Predictors of recurrence in stage 1 (T1N1M0) breast carcinoma. Ann Surg. 1981;193:15–20.
de Mascarel I, Bonichon I, Coindre J, Trojani M. Prognostic significance of breast cancer axillary lymph node micrometastases assessed by two special techniques: reevaluation with longer follow-up. Br J Cancer. 1992;66:523–7.
McGuckin MA, Cummings MC, Walsh MD, Hohn BG, Bennett IC, Wright RG. Occult axillary node metastases in breast cancer: their detection and prognostic significance. Br J Cancer. 1996;73:88–95.
Sedmak DD, Meineke TA, Knechtges DS. Detection of metastatic breast carcinoma with monoclonal antibodies to cytokeratins. Arch Pathol Lab Med. 1989;113:786–9.
de Boer M, van Deurzen C, van Dijck J, Borm GF, van Diest PJ, Adang EM, et al. Micrometastases or isolated tumor cells and the outcome of breast cancer. N Engl J Med. 2009;361:653–63.
de Boer M, van Dijck JAAM, Bult P, Borm GF, Tjan-Heijnen VC. Breast cancer prognosis and occult lymph node metastases, isolated tumor cells, and micrometastases. J Natl Cancer Inst. 2010;102:410–25.
Lockett MA, Baron PL, O’Brien PH, Elliott BM, Robison JG, Maitre N, et al. Detection of occult breast cancer micrometastases in axillary lymph nodes using a multi-marker reverse transcriptase-polymerase chain reaction panel. J Am Coll Surg. 1998;187:9–16.
Lockett MA, Metcalf JS, Baron PL, O’Brien PH, Elliott BM, Robison JG, et al. Efficacy of reverse transcriptase-polymerase chain reaction screening for micrometastatic disease in axillary lymph nodes of breast cancer patients. Am Surg. 1998;64:539–43.
Schoenfeld A, Luqmani Y, Smith D, O’Reilly S, Shousha S, Sinnett HD, et al. Detection of breast cancer micrometastases in axillary lymph nodes by using polymerase chain reaction. Cancer Res. 1994;54:2986–90.
Mori M, Mimori K, Inoue H, Barnard GF, Tsuji K, Nanbara S, et al. Detection of cancer micrometastases in lymph nodes by reverse transcriptase-polymerase chain reaction. Cancer Res. 1995;74:3417–20.
Datta YH, Adams PT, Drobyski WR, Ethier SP, Terry VH, Roth MS. Sensitive detection of occult breast cancer by the reverse-transcriptase polymerase chain reaction. J Clin Oncol. 1994;12:475–82.
Bostick PJ, Chatterjee S, Chi DD, Huynh KT, Giuliano AE, Cote R, et al. Limitations of specific reverse-transcriptase polymerase chain reaction markers in the detection of metastases in the lymph nodes and blood of breast cancer patients. J Clin Oncol. 1998;16:2632–40.
Eltahir EM, Mallinson DS, Birnie GD, Hagan C, George WD, Purushotham AD. Putative markers for the detection of breast carcinoma cells in blood. Br J Cancer. 1998;77:1203–7.
Giesing M, Austrup F, Bockmann B, Driesel G, Eder C, Kusiak I, et al. Independent prognostication and therapy monitoring of breast cancer patients by DNA/RNA typing of minimal residual cancer cells. Int J Biol Markers. 2000;15:94–9.
Goeminne JC, Guillaume T, Salmon M, Machiels JP, D’Hondt V, Symann M. Unreliability of carcinoembryonic antigen (CEA) reverse transcriptase-polymerase chain reaction (RT-PCR) in detecting contaminating breast cancer cells in peripheral blood stem cells due to induction of CEA by growth factors. Bone Marrow Transplant. 1999;24:769–75.
Lopez-Guerrero JA, Gilabert PB, Gonzalez EB, Sanz Alonso MA, Perez JP, Talens AS, et al. Use of reverse-transcriptase polymerase chain reaction (RT-PCR) for carcinoembryonic antigen, cytokeratin 19, and maspin in the detection of tumor cells in leukapheresis products from patients with breast cancer: comparison with immunocytochemistry. J Hematother. 1999;8:53–61.
Gasparini G, Toi M, Gion M, Verderio P, Dittadi R, Hanatani M, et al. Prognostic significance of vascular endothelial growth factor protein in node-negative breast carcinoma. J Natl Cancer Inst. 1997;89:139–47.
Mitas M, Mikhitarian K, Walters C, Baron PL, Elliott BM, Brothers TE, et al. Quantitative real-time RT-PCR detection of breast cancer micrometastasis using a multigene marker panel. Int J Cancer. 2001;93:162–71.
Mitas M, Mikhitarian K, Hoover L, Lockett MA, Kelley L, Hill A, Gillanders WE, et al. Prostate-specific Ets (PSE) factor: a novel marker for detection of metastatic breast cancer in axillary lymph nodes. Br J Cancer. 2002;86:899–904.
Veys I, Durbecq V, Majjaj S, Schobbens JC, Noterman D, Sirtaine N, et al. Eighteen months clinical experience with the GeneSearch breast lymph node assay. Am J Surg. 2009;198:203–9.
Tafe L, Schwab M, Lefferts JA, Wells WA, Tsongalis GJ. A validation study of a new molecular diagnostic assay: The Dartmouth-Hitchcock Medical Center experience with the GeneSearch BLN assay in breast sentinel lymph nodes. Exp Mol Pathol. 2010;88:1–6.
Gillanders WE, Mikhitarian K, Hebert R, Mauldin PD, Palesch Y, Walters C, et al. Molecular detection of micrometastatic breast cancer in histopathology-negative axillary lymph nodes correlates with traditional predictors of prognosis. Ann Surg. 2004;239:828–40.
Noguchi S, Aihara R, Motomura K, Inaji H, Imaoka S, Koyama H. Histological characteristics of breast cancer patients with occult lymph node metastases detected by keratin 19 mRNA reverse transcriptase-polymerase chain reaction. Cancer. 1996;78:1235–40.
Coates AS, Keshaviah A, Thurlimann B, Mouridsen H, Mauriac L, Forbes JF, et al. Five years of letrozole compared with tamoxifen as initial adjuvant therapy for postmenopausal women with endocrine-responsive early breast cancer: update of study BIG 1-98. J Clin Oncol. 2007;25:486–92.
Coombes RC, Kilburn LS, Snowdon CF, Paridaens R, Coleman RE, Jones SE, et al. Survival and safety of exemestane versus tamoxifen after 2-3 years’ tamoxifen treatment (intergroup exemestane study): a randomised controlled trial. Lancet. 2007;369:559–70.
Howell A, Cuzick J, Baum M, Buzdar A, Dowsett M, Forbes JF, et al. Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvant treatment for breast cancer. Lancet. 2005;365:60–2.
Abdul-Rasool S, Kidson S, Panieri E, Dent D, Pillay K, Hanekom GS. An evaluation of molecular markers for improved detection of breast cancer metastases in sentinel nodes. J Clin Pathol. 2006;59:289–97.
Hughes SJ, Xi L, Raja S, Gooding W, Cole DJ, Gillanders WE, et al. A rapid, fully automated, molecular-based assay accurately analyzes sentinel lymph nodes for the presence of metastatic breast cancer. Ann Surg. 2006;243:389–98.
Martin Martinez MDM, Veys I, Majjaj S, Lespagnard L, Schobbens JC, Rouas G, et al. Clinical validation of a molecular assay for intra-operative detection of metastases in breast sentinel lymph nodes. Eur J Surg Oncol. 2009;35:387–92.
Backus J, Laughlin T, Wang Y, Belly R, White R, Baden J, et al. Identification and characterization of optimal gene expression markers for detection of breast cancer metastasis. J Mol Diagn. 2005;7:327–36.
Zehentner BK, Dillon DC, Jiang Y, Xu J, Bennington A, Molesh DA, et al. Application of a multigene reverse transcription-PCR assay for detection of mammaglobin and complementary transcribed genes in breast cancer lymph nodes. Clin Chem. 2002;48:1225–31.
Manzotti M, Dell’Orto P, Maisonneuve P, Zurrida S, Mazzarol G, Viale G. Reverse transcription-polymerase chain reaction assay for multiple mRNA markers in the detection of breast cancer metastases in sentinel lymph nodes. Int J Cancer. 2001;95:307–12.
Min CJ, Tafra L, Verbanac KM. Identification of superior markers for PCR detection of breast cancer metastases in sentinel lymph nodes. Cancer Res. 1998;58:4581–4.
Marchetti A, Buttitta F, Bertacca G, Zavaglia K, Bevilacqua G, Angelucci D, et al. mRNA markers of breast cancer nodal metastases: comparison between mammaglobin and carcinoembryonic antigen in 248 patients. J Pathol. 2001;195:186–90.
Watson MA, Dintzis S, Darrow CM, Voss LE, DiPersio J, Jensen R, et al. Mammaglobin expression in primary, metastatic, and occult breast cancer. Cancer Res. 1999;59:3028–31.
Leygue E, Snell L, Dotzlaw H, Hole K, Troup S, Hiller-Hitchcock T, et al. Mammaglobin, a potential marker of breast cancer nodal metastasis. J Pathol. 1999;189:28–33.
Viale G, Dell’Orto P, Biasi M, Stufano V, De Brito Lima LN, et al. Comparative evaluation of an extensive histopathologic examination and a real-time reverse-transcription-polymerase chain reaction assay for mammaglobin and cytokeratin 19 on axillary sentinel lymph nodes of breast carcinoma patients. Ann Surg. 2008;247:136–42.
Blumencranz P, Whitworth P, Deck K, Rosenberg A, Reintgen D, Beitsch P, et al. Scientific Impact Recognition Award. Sentinel node staging for breast cancer: intraoperative molecular pathology overcomes conventional histologic sampling errors. Am J Surg. 2007;194:426–32.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was support by the grant from Department of Defense N00014-99-1-0784.
Rights and permissions
About this article
Cite this article
Fisher, C.S., Cole, D.J., Mitas, M. et al. Molecular Detection of Micrometastatic Breast Cancer in Histopathology—Negative Axillary Lymph Nodes Fails to Predict Breast Cancer Recurrence: A Final Analysis of a Prospective Multi-Institutional Cohort Study. Ann Surg Oncol 17 (Suppl 3), 312–320 (2010). https://doi.org/10.1245/s10434-010-1258-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1258-y