Abstract
Background
Randomized trials have demonstrated a benefit associated with adjuvant therapy for pancreatic cancer, and retrospective studies have demonstrated improvements in postoperative mortality. The purpose of this study was to evaluate whether these improvements could be identified in a cohort of patients who underwent resection for pancreatic cancer at a single institution over three decades.
Methods
Short- (30 days), intermediate- (1 year), and long-term survival were compared between decades. Long-term survival focused on patients who survived at least 1 year to minimize the effects of perioperative mortality and patient selection.
Results
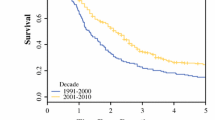
Between 1983 and 2009, 1147 pancreatic resections were performed for ductal adenocarcinoma, including 123 resections in the 1980s, 399 in the 1990s, and 625 in the 2000s. The 30-day mortality rates were 4.9%, 1.5% (P = 0.03 vs. 1980s), and 1.3% (P = 0.007 vs. 1980s). The 1-year mortality rates were 42%, 31% (P < 0.001 vs. 1980s), and 24% (P < 0.001 vs. 1980s and 1990s). In the group of patients who survived 1 year, the overall survivals were 23.2 months, 25.6 months (P = 0.6 vs. 1980s), and 24.5 months (P = 0.2 vs. 1980s). In a multivariate analysis adjusted for pathologic features, the decade of resection was not a significant predictor of long-term survival (hazard ratio = 1.1, P = 0.3).
Conclusions
Patients who underwent resection for pancreatic cancer between 2000 and 2009 experienced improved operative mortality and 1-year survival compared to those who underwent resection in the 1980s, while the long-term survival was similar over all three decades. These results underscore the need for early detection strategies and more effective adjuvant therapies for patients with pancreatic cancer.




Similar content being viewed by others
References
Kalser MH, Ellenberg SS. Pancreatic cancer. Adjuvant combined radiation and chemotherapy following curative resection. Arch Surg. 1985;120:899–903.
Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350:1200–10.
Oettle H, Post S, Neuhaus P, et al. Adjuvant chemotherapy with gemcitabine vs. observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297:267–77.
Begg CB, Cramer LD, Hoskins WJ, Brennan MF. Impact of hospital volume on operative mortality for major cancer surgery. JAMA. 1998;280:1747–51.
Winter JM, Cameron JL, Campbell KA, et al. 1423 pancreaticoduodenectomies for pancreatic cancer: a single-institution experience. J Gastrointest Surg. 2006;10:1199–210.
Grobmyer SR, Pieracci FM, Allen PJ, Brennan MF, Jaques DP. Defining morbidity after pancreaticoduodenectomy: use of a prospective complication grading system. J Am Coll Surg. 2007;204:356–64.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225–49.
Thorson AG. Progress in cancer care: a rational call to do better. CA Cancer J Clin. 2010;60:7–11.
Riall TS, Nealon WH, Goodwin JS, et al. Pancreatic cancer in the general population: improvements in survival over the last decade. J Gastrointest Surg. 2006;10:1212–23.
Simons JP, Ng SC, McDade TP, Zhou Z, Earle CC, Tseng JF. Progress for resectable pancreatic [corrected] cancer?: a population-based assessment of US practices. Cancer. 2010;116:1681–90.
Winter JM, Cameron JL, Lillemoe KD, et al. Periampullary and pancreatic incidentaloma: a single institution’s experience with an increasingly common diagnosis. Ann Surg. 2006;243:673–80.
Balcom JH 4th, Rattner DW, Warshaw AL, Chang Y, Fernandez-del Castillo C. Ten-year experience with 733 pancreatic resections: changing indications, older patients, and decreasing length of hospitalization. Arch Surg. 2001;136:391–8.
Kosuge T, Kiuchi T, Mukai K, Kakizoe T, Japanese Study Group of Adjuvant Therapy for Pancreatic Cancer (JSAP). A multicenter randomized controlled trial to evaluate the effect of adjuvant cisplatin and 5-fluorouracil therapy after curative resection in cases of pancreatic cancer. Jpn J Clin Oncol. 2006;36:159–65.
Smeenk HG, van Eijck CH, Hop WC, et al. Long-term survival and metastatic pattern of pancreatic and periampullary cancer after adjuvant chemoradiation or observation: long-term results of EORTC trial 40891. Ann Surg. 2007;246:734–40.
Boeck S, Ankerst DP, Heinemann V. The role of adjuvant chemotherapy for patients with resected pancreatic cancer: systematic review of randomized controlled trials and meta-analysis. Oncology. 2007;72:314–21.
NCCN Clinical practice guidelines in oncology. Pancreatic adenocarcinoma. http://www.nccn.org/professionals/physician_gls/PDF/pancreatic.pdf (2010).
Bilimoria KY, Bentrem DJ, Ko CY, et al. Multimodality therapy for pancreatic cancer in the U.S.: utilization, outcomes, and the effect of hospital volume. Cancer. 2007;110:1227–34.
Bilimoria KY, Bentrem DJ, Tomlinson JS, et al. Quality of pancreatic cancer care at Veterans Administration compared with non–Veterans Administration hospitals. Am J Surg. 2007;194:588–93.
Takada T, Amano H, Yasuda H, et al.; Study Group of Surgical Adjuvant Therapy for Carcinomas of the Pancreas and Biliary Tract. Is postoperative adjuvant chemotherapy useful for gallbladder carcinoma? A phase III multicenter prospective randomized controlled trial in patients with resected pancreaticobiliary carcinoma. Cancer. 2002;95:1685–95.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winter, J.M., Brennan, M.F., Tang, L.H. et al. Survival after Resection of Pancreatic Adenocarcinoma: Results from a Single Institution over Three Decades. Ann Surg Oncol 19, 169–175 (2012). https://doi.org/10.1245/s10434-011-1900-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-1900-3