Abstract
Vacuolar-type H+-adenosine triphosphatase (V-ATPase) is one of the most fundamental enzymes in nature. V-ATPases are responsible for the regulation of proton concentration in the intracellular acidic compartments. It has similar structure with the mitochondrial F0F1-ATP synthase (F-ATPase).† The V-ATPases are composed of multiple subunits and have various physiological functions, including membrane and organelle protein sorting, neurotransmitter uptake, cellular degradative processes, and cytosolic pH regulation. The V-ATPases have been involved in multidrug resistance. Recently, plasma membrane V-ATPases have been involved in regulation of extracellular acidity, essential for cellular invasiveness and proliferation in tumor metastasis. The current knowledge regarding the structure and function of V-ATPase and its role in cancer biology is discussed.
Similar content being viewed by others
References
Mellman, I., Fuchs, R., and Helenius, A. (1986) Acidification of the endocytic and exocytic pathway. Ann. Rev. Biochem. 55, 663–700.
Nelson, N. (2003) A journey from mammals to yeast with vacuolar H+-ATPase (V-ATPase). J. Bioenerg. Biomembr. 35, 281–289.
Wieczorek, H., Brown, D., Grinstein, S., Ehrenfeld, J., and Harvey, W. R. (1999) Energization of animal plasma membranes by proton-motive V-ATPase. Bioassays 21, 637–648.
Anraku, Y., Umemoto, N., Hirata, R., and Wada, Y. (1989) Structure and function of the yeast vacuolar membrane proton ATPase. J. Bioenerg. Biomembr. 21, 589–603.
Futai, M., Oka, T., Moriyama, Y., and Wada, Y. (1998) Diverse roles of single membrane organelles: factors establishing the acid luminal pH. J. Biochem. 124, 259–267.
Nelson, N. (1992) Structural conservation and functional diversity of V-ATPases. J. Bioenerg. Biomembr. 24, 407–414.
Nelson, N. and Harvey, W. R. (1999) Vacuolar and plasma membrane proton-adenosinetriphosphatases. Physiol. Rev. 7, 361–385.
Stevens, T. H. and Forgac, M. (1997) Structure, function and regulation of the vacuolar H+-ATPase. Annu. Rev. Cell Dev. Biol. 13, 779–808.
Cross, R. L. (2000) The rotary binding change mechanism of ATP synthases. Biochim. Biophys. Acta 1458, 270–275.
Weber, J. and Senior, A. E. (2000) ATP synthase: what we know about ATP hydrolysis and what we do not know about ATP synthesis. Biochim. Biophys. Acta 1458, 300–309.
Fillingame, R. H., Jiang, W., and Dmitriev, O. Y. (2000) Coupling H+ transport to rotary catalysis in F-type ATP synthases. J. Exp. Biol. 203, 9–17.
Forgac, M. (1999) Structure and properties of the vacuolar (H+)-ATPases. J. Biol. Chem. 274, 12951–12954.
Finbow, M. E. and Harrison, M. A. (1997) The vacuolar H+-ATPase: a universal proton pump of eukaryotes. Biochem. J. 324, 697–712.
Bowman, E. J. and Bowman, B. J. (2000) Cellular role of the V-ATPases in Neurospora crassa. J. Exp. Biol. 203, 97–106.
Nishi, T. and Forgac M. (2002) The vacuolar (H+)-ATPase—nature's most versatile proton pumps. Nat. Rev. Mol. Cell Biol. 3, 94–103.
Xu, T., Vasilyeva, E., and Forgac, M. (1999) Subunit interactions in the clathrin-coated vesicle V-ATPase complex. J. Biol. Chem. 274, 28909–28915.
Powell, B., Graham, L. A., and Stevens, T. H. (2000) Molecular characterization of the yeast vacuolar H+-ATPase proton pore. J. Biol. Chem. 275, 23654–23660.
Bowman, B. J., Vazquez-Laslop, N., and Bowman, E. J. (1992) The vacuolar ATPase of Neurospora crassa. J. Bioenerg. Biomembr. 24, 361–370.
Graham, L. A., Hill, K. J., and Stevens, T. H. (1995) VMA8 encodes a 32 kDa V1 subunit of Saccharomyces cerevisiae vacuolar H+-ATPase required for functions and assembly of the enzyme complex. J. Biol. Chem. 270, 15037–15044.
Nelson, H., Mandian, S., and Nelson, N. (1995) A bovine cDNA and yeast gene (VMA8) encoding the subunit D of vacuolar H+-ATPase. Proc. Natl. Acad. Sci. USA 92, 497–501.
Perin, M. S., Fried, V. A., Stone, D. K., Xie, X. S., and Sudhof, T. C. (1991) Structure of the 116-kDa polypeptide of the clathrin-coated vesicle/synaptic vesicle proton pump. J. Biol. Chem. 266, 3877–3881.
Leng, X. H., Manolson, M., Liu, Q., and Forgac, M. (1996) Site-directed mutagenesis of the 100 kDa subunit (Vph1p) of the yeast vacuolar (H+)-ATPase. J. Biol. Chem. 271, 22487–22493.
Leng, X. H., Manolson, M., and Forgac, M. (1998) Function of the COOH-terminal domain of Vph1p in activity and assembly of the yeast V-ATPase. J. Biol. Chem. 273, 6717–6723.
Vik, S. B. and Antonio, B. J. (1994) A mechanism of proton translocation by F1F0-ATP synthases suggested by double mutants of the a subunit. J. Biol. Chem. 269, 30364–30369.
Junge, W., Lill, H., and Engelbrecht, S. (1997) ATP synthase: an electrochemical transducer with rotatory mechanics. Trends Biol. Sci. 22, 420–423.
Zhang, V. J., Feng, Y., and Forgac, M. (1994) Proton conduction and bafilomycin binding by the domain of the coated vesicle V-ATPase. J. Biol. Chem. 269, 23518–23523.
Bowman, B. J. and Bowman, E. J. (2002) Mutations in subunit c of the vacuolar ATPase confer resistance to bafilomycin and identify a conserved antibiotic binding site. J. Biol. Chem. 277, 3965–3972.
Huss, M., Ingenhorst, G., Konig, S., Gassel, M., Drose, S., Zeeck, A., et al. (2002) Concanamycin A, the specific inhibitor of V-ATPases, binds to the V(o) subunit c. J. Biol. Chem. 277, 40544–40548.
Fillingame, R. H. (1997) Coupling H+ transport and ATP synthesis in F1F0-ATP synthases: glimpses of interacting parts in a dynamic molecular machine. J. Exp. Biol. 200, 217–224.
Adachi, I., Puopolo, K., Marquez-Sterling, N., Arai, H., and Forgac, M. (1990) Dissociation, cross-linking, and glycosylation of the coated vesicle proton pump. J. Biol. Chem. 265, 967–973.
Forgac, M. (1998) Structure, function and regulation of the vacuolar H+-ATPase. FEBS Letters 440, 258–263.
Nelson, N. (1989) Structure, molecular genetics and evolution of vacuolar H+-ATPases. J. Bioenerg. Biomembr. 21, 553–571.
Nelson, N. (1992) The vacuolar H+-ATPase—One of the most fundamental ion pumps in nature. J. Exp. Biol. 172, 19–27.
MacLeod, K. J., Vasilyeva, E., Merdek, K., Vogel, P. D., and Forgac, M. (1999) Photoaffinity labeling of wild-type and mutant forms of the yeast V-ATPase A subunit by 2-azido-[32P]-ADP. J. Biol. Chem. 274, 32869–32874.
Arata, Y., Nishi, T., Kawasaki-Nishi, S., Shao, E., Wilkens, S., and Forgac, M. (2002) Structure, subunit function and regulation of the coated vesicle and yeast vacuolar H+-ATPases. Biochim Biophys Acta. 1555, 71–4.
Bowman, E. J., Tenney, K., and Bowman, B. J. (1988) Isolation of genes encoding the Neurospora vacuolar ATPase. Analysis of vma-1 encoding the 67-kDa subunit reveals homology to other ATPases. J. Biol. Chem. 263, 13994–14001.
Hirata, R., Ohsumk, Y., Nakano, A., Kawasaki, H., Suzuki, K., and Anraku, Y. (1990) Molecular structure of a gene, VMA1, encoding the catalytic subunit of H(+)-translocating adenosine triphosphatase from vacuolar membranes of Saccharomyces cerevisiae. J. Biol Chem. 265, 6726–6733.
Puopolo, K., Kumamoto, C., Adachi, I., and Forgac, M. (1991) A single gene encodes the catalytic “A” subunit of the bovine vacuolar H(+)-ATPase. J. Biol. Chem. 266, 24564–24572.
Shao, E., Nishi, T., Kawasaki-Nishi, S., and Forgac, M. (2003) Mutational analysis of the non-homologous region of subunit A of the yeast V-ATPase. J Biol Chem. 278, 12985–12991.
Zimniak, L., Dittrich, P., Gogarten, J. P., Kibak, H., and Taiz, L. (1988) The cDNA sequence of the 69-kDa subunit of the carrot vacuolar H+-ATPase. Homology to the beta-chain of 0F1-ATPases. J. Biol. Chem. 263, 9102–9112.
Xu, T. and Forgac, M. (2000) Subunit D (Vma8p) of the yeast vacuolar (H+)-ATPase plays a role in coupling of proton transport and ATP hydrolysis. J. Biol. Chem. 275, 22075–22081.
Doherty, R. D. and Kane, P. M. (1993) Partial assembly of the yeast vacuolar H+-ATPase in mutants laking one subunit of the enzyme. J. Biol. Chem. 268, 16845–16851.
Ho, M., Hill, K. J., Lindorfer, M. A., and Stevens, T. H. (1993) Isolation of vacuolar membrane H+-ATPase-deficient yeast mutants: the VMA5 and VMA4 genes are essential for assembly and activity of the vacuolar H+-ATPase. J. Biol. Chem. 268, 221–227.
Parra, K. J., Keenan, K. L., and Kane, P. M. (2000) The H subunit of the yeast V-ATPase inhibits the ATPase activity of cytosolic V1 complexes. J. Biol. Chem. 275, 21761–21767.
Arata, Y., Baleja, J. D., and Forgac, M. (2002) Cysteine-directed crosslinking to subunit B suggests that subunit E forms part of the peripheral stalk of the V-ATPase. J. Biol. Chem. 277, 3357–3363.
Tomashek, J. J., Graham, L. A., Hutchins, M. U., Stevens, T. H., and Klionsky, D. J. (1997) V1-situated stalk subunits of the yeast V-ATPase. J. Biol. Chem. 272, 26787–26793.
Crider, B. P., Andersen, P., White, A. E., Zhou, Z., Li, X., Mattsson, J. P., et al. (1997) Subunit G of the vacuolar proton pump. Molecular characterization, and functional expression. J. Biol. Chem. 272, 10721–10728.
Lepier, A., Graf, R., Azuma, M., Merzendorfer, H., Harvey, W. R., and Wieczorek, H. (1996) The peripheral complex of the tobacco hornworm V-ATPase contains a novel 13-kDa subunit G. J. Biol. Chem. 271, 8502–8508.
Supekova, L., Supek, F., and Nelson, N. (1995) The Saccharomyces cerevisiae VMA10 is an intron-containing gene encoding a novel 13-kDa subunit of vacuolar H+-ATPase. J. Biol. Chem. 270, 13726–13732.
Hunt, I. E. and Bowman, B. J. (1997) The intriguing evolution of the b and G subunits in F-type and V-type ATPases. J. Bioenerg. Biomembr. 29, 533–540.
Sorgen, P. L., Caviston, T. L., Perry, R. C., and Cain, B. D. (1998) Deletions in the second stalk of F1F0-ATP synthase in E. coli. J. Biol. Chem. 273, 27873–27878.
Charsky, C. M., Schumann, N. J., and Kane, P. M. (2000) Mutational analysis of subunit G (VmA10p) of the yeast V-ATPase. J. Biol. Chem. 275, 37232–37239.
Arata, Y., Baleja, J. D., and Forgac, M. (2002) Localization of subunits D, E, and G in the yeast V-ATPase complex using cysteine-mediated cross-linking to subunit B. Biochemistry 41: 11301–7.
Curtis, K. K. and Kane, P. M. (2002) Novel V-ATPase complexes resulting from overproduction of Vma5p and VmA13p. J. Biol. Chem. 277, 8979–8988.
Hirata, R., Graham, L. A., Takatsuki, A., Stevens, T. H., and Anraku, Y. (1997) VMA11 and VMA16 encode second and third proteolipid subunits of the Saccharomyces cerevisiae vacuolar membrane H+-ATPase. J. Biol. Chem. 272, 4795–4803.
Sun, S. Z., Xie, Z. S., and Stone, D. K. (1987) Isolation and reconstitution of the DCCDsensitive proton pore of the clathrincoated vesicle proton transclocating complex. J. Biol. Chem. 262, 14790–14794.
Vik, S. B., Long, J. C., Wada, T., and Zhang, D. (2000) A model for the structure of subunit a of the E. coli ATP synthase and its role in proton translocation. Biochim. Biophys. Acta 1458, 457–466.
Cain, B. D. (2000) Mutagenic analysis of F0 stator subunits. J. Bioenerg. Bioemembr. 32, 365–371.
Landolt-Marticorena, C., Williams, K. M., Correa, J., Chen, W., and Manolson, M. F. (2000). Evidence that the NH2-terminus of Vph1p, an integral subunit of the V0 sector of the yeast V-ATPase, interacts directly with the VmA1p and VmA13p subunits of the V1 sector. J. Biol. Chem. 275, 15449–15457.
Schneider, E. and Altendorf, K. (1985) All three subunits are required for the reconstitution of an active proton channel F0 of E. coli ATP synthase F1F0. EMBO 4, 515–518.
Kawasaki-Nishi, S., Nishi, T., and Forgac, M. (2001) Arg735 of the 100 kDa a subunit of the yeast V-ATPase is essential for proton translocation. Proc. Natl Acad. Sci. 98, 12397–12402.
Vaananen, H. K., Karhukorpi, E. K., Sundquist, K., Wallmark, B., Roininen, I., Hentunen, T., t al. (1990) Evidence for the presence of a proton pump of the vacuolar (H(+)-ATPase type in the ruffled borders of osteoclasts. J. Cell Biol. 111, 1305–1311.
Gluck, S. L., Underhill, D. M., Iyori, M., Holliday, L. S., Kostrominova, T. Y., and Lee, B. S. (1996) Physiology and biochemistry of the kidney vacuolar H+-ATPase. Annu. Rev. Physiol. 58, 427–445.
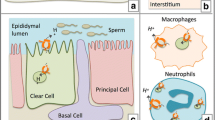
Breton, S., Smith, P. J., Lui, B., and Brown, D. (1996) Acidification of the male reproductive tract by a proton pumping H(+)-ATPase. Nat. Med. 2, 470–472.
Martinez-Zaguilan, R. (1999) Angiostatin's partners. Science 284, 433–434.
Forgac, M. (1989) Structure and function of vacuolar class of ATP-driven proton pumps. Physiol. Rev. 69, 765–796.
Davoust, J., Gruenberg, J., and Howell, K. E. (1987) Two threshold values of low pH block endocytosis at different stages. EMBO 6, 3601–3609.
Van Weert, A. W., Dunn, K. W., Geuze, H. J., Maxfield, F. R., and Stoorvogel, W. (1995) Transport from late endosomes to lysosomes, but sorting of integral membrane proteins in endosomes, depends on the vacuolar proton pump. J. Cell Biol. 130, 821–834.
Maxfield, F. R. and Yamashiro, D. J. (1987) Endosome acidification and the pathways of receptor-mediated endocytosis. Adv. Exp. Med. Biol. 225, 189–198.
Mellman, I. (1992) The importance of being acid: the role of acidification in intracellular membrane traffic. J. Exp. Biol. 172, 39–45.
Demaurex, N. (2002) pH homeostasis of cellular organelles. News Physiol Sci 17, 1–5.
Mellman, I. (1996) Endocytosis and molecular sorting. Annu. Rev. Cell Dev. Biol. 12, 575–625.
Trowbridge, J. S., Collawn, J. F., and Hopkins, C. R. (1993) Signal-dependent membrane protein trafficking in the endocytic pathway. Annu. Rev. Cell Biol. 9, 129–161.
Clague, M. J., Urbe, S., Aniento, F., and Gruenberg, J. (1994) Vacuolar ATPase activity is required for endosomal carrier vesicle formation. J. Biol. Chem. 269, 21–24.
Han, X., Bushweller, J. H., Cafiso, D. S., and Tamm, L. K. (2001) Membrane structure and fusion-triggering conformational change of the fusion domain from influenza hemagglutinin. Nat. Struct. Biol. 8, 715–720.
White, J. M. (1992) Membrane fusion. Science 258, 917–924.
Kornfeld, S. (1992) Structure and function of the mannose 6-phosphate/insulinlike growth factor II receptors. Annu. Rev. Biochem. 61, 307–330.
Schapiro, F. B. and Grinstein, S. (2000) Determinants of the pH of the Golgi complex. J. Biol. Chem. 275, 21025–21032.
Brown, D. and Breton, S. (1996) Mitochondriarich, proton-secreting epithelial cels. J. Exp. Biol. 199, 2345–2358.
Al-Awquati, Q. (1996) Plasticity in epithelial polarity of renal intercalated cells: targeting of the H(+)-ATPase and band 3. Am. J. Physiol. 270, C1571-C1580.
McKinney, T. D. and Burg, M. B. (1977) Bicarbonate transport by rabbit cortical collecting tubules. Effect of acid and alkali loads in vivo on transport in vitro. J. Clin. Invest. 60, 766–768.
Lambard, W. E., Kokko, J. P., and Jacobson, H. R. (1983) Bicarbonate transport in cortical and outer medullary collecting tubules. Am. J. Physiol. 244, F289-F296.
Brown, D. and Breton, S. (2000) H(+)V-ATPasedependent luminal acidification in the kidney collecting duct and the epididymis/vas deferens: vesicle recycling and transcytotic pathways. J. Exp. Biol. 1, 137–145.
Brown, D., Gluck, S., and Hartwig, J. (1987) Structure of the novel membrane-coating material in proton-secreting epithelial cells and identification as an H+ATPase. J. Cell Biol. 105, 1637–1648.
Frattini, A., Orchard, P. J., Sobacchi, C., Giliani, S., Abinun, M., Mattsson, J. P., et al. (2000) Defects in TCIRG1 subunit of the vacuolar proton pump are responsible for a subset of human autosomal recessive osteopetrosis. Nat. Genet. 25, 343–346.
Karet, F. E., Finberg, K. E., Nelson, R. D., Nayir, A., Moan, H., Sanjad, S. A., et al. (1999) Mutations in the gene encoding B1 subunit of H+-ATPase cause renal tubular acidosis with sensorineural deafness. Nature Genet. 21, 84–90.
Smith, A. N., Skaug, J., Choate, K. A., Nayir, A., Bakkaloglu, A., Ozen, S., et al. (2000) Mutations in ATP6N1B, encoding a new kidney vacuolar proton pump 116-kDa subunit, cause recessive distal renal tubular acidosis with preserved hearing. Nat. Genet. 26, 71–75.
Gluck, S., Cannon, C., and Al-Awqati, Q. (1982) Exocytosis regulates urinary acidification in turtle bladder by rapid insertion of H+ pumps into the luminal, membrane. Proc. Natl. Acad. Sci. USA 79, 4327–31.
Swallow, C. J., Grinstein, S., and Rotstein, O. D. (1990) A vacuolar type (H+)-ATPase regulates cytoplasmic pH in murine macrophages. J. Biol. Chem. 265, 7645–7654.
Swallow, C. J., Grinstein, S., Sudsbury, R. A., and Rotstein, O. D. (1993) Relative roles of Na+/H+ exchange and vacuolar-type H+ ATPases in regulating cytoplasmic pH and function in murine peritoneal macrophages. J. Cell Physiol. 157, 453–460.
Nanda, A., Gukovskaya, A., Tseng, J., and Grinstein, S. (1992) Activation of vacuolar-type proton pumps by protein kinase C. Role in neutrophil pH regulation. J. Biol. Chem. 267, 22740–22746.
Gottlieb, R. A., Giesing, H. A., Zhu, J. Y., Engler, R. L., and Babior, B. M. (1995) Cell acidification in apoptosis: granulocyte colonystimulating factor delays programmed cell death in neutrophils by up-regulating the vacuolar H(+)-ATPase. Proc. Natl. Acad. Sci. USA 92, 5965–8.
Niessen, H., Meisenholder, G. W., Li, H. L., Guck, S. L., Lee, B. S., Bowman, B., et al. (1997) Granulocyt colony-stimulating factor upregulates the vacuolar proton ATPase in human neutrophils. Blood 90, 4598–4601.
Gluck, S. (1992) The osteoclast as a unicellular proton-transporting epithelium. Am. J. Med. Sci. 303, 134–139.
Li, Y. P., Chen, W., Liang, Y., Li, E., and Stashenko, P. (1999) Atp6i-deficient mice exhibit severe osteopetrosis due to loss of osteoclast-mediated extracellular acidification. Nat. Genet. 23, 447–451.
Blair, H. C., Teitelbaum, S. L., Ghiselli, R., and Gluck, S. (1989) Osteoclastic bone resorption by a polarized vacuolar proton pump. Science 245, 855–857.
Baron, R. (1989) Molecular mechanisms of bone resorption by the osteoclast. Anat. Rec. 224, 317–324.
Chatterjee, D., Chakraborty, M., Leit, M., Neff, L., Jamsa-Kellokumpu, S., Fuchs, R., et al. (1992) Sensitivity to vanadate and isoforms of subunits A and B distinguish the osteoclast proton pump from other vacuolar H+ATPases. Proc. Natl. Acad. Sci. USA 89, 6257–6261.
Chatterjee, D., Chakraborty, M., Leit, M., Neff, L., Jamsa-Kellokumpu, S., Fuchs, R., et al. (1992) The osteoclast proton pump differs in its pharmacology and catalytic subunits from other vacuolar H(+)-ATPases. J. Exp. Biol. 172, 193–204.
Mattsson, J. P., Schlesinger, P. H., Keeling, D. J., Teitelbaum, S. L., Stone, D. K., and Xie, X. S. (1994) Isolation and reconstitution of a vacuolar-type proton pump of osteoclast membranes. J. Biol. Chem. 269, 24979–24988.
Bedford, J. M. (1970) Sperm capacitation and fertilization in mammals. Biol. Reprod. 2, 128–158.
Meizel, S. and Deamer, D. W. (1978) The pH of the hamster sperm acrosome. J. Histochem. Cytochem. 26, 98–105.
Huang, T. T. Jr., Hardy, D., Yanagimachi, H., Teuscher, C., Tung, K., Wild, G., et al. (1985) pH and protease control of acrosomal content stasis and release during the guinea pig sperm acrosome reaction. Biol. Reprod. 32, 451–462.
Kawa, G., Yamamoto, A., Yoshimori, T., Muguruma, K., Matsuda, T., and Moriyama, Y. (2000) Immunohistochemical localization of V-ATPases in rat spermatids. Int. J. Androl. 23, 278–283.
Martínez-Zaguilán, R., Lynch, R., Martinez, G., and Gillies, R. (1993) Vacuolar-type H+ATPase are functionally expressed in plasma membrane of human tumor cells. Am. J. Physiol. 265, C1015-C1029.
Griffiths, J. R. (1991) Are cancer cells acidic? Br. J. Cancer 64, 425–427.
Stubbs, M., Veech, R. L., and Griffiths, J. R. (1995) Tumor metabolism: the lessons of magnetic resonance spectroscopy. Adv. Enzyme Regul. 35, 101–115.
Putna, R. W. (2001) Intracellular pH regulation, in Cell Physiology Source (Speralakis, N., ed.), 3rd ed., Academic Press, pp. 357–376.
Gillies, R. J. and Martínez-Zaguilán, R. (1991) Regulation of intracellular pH in BALB/c 3T3 cells. Bicarbonate raises pH via NaHCO3/HCl exchange and attenuates the activation of Na+/H+ exchange by serum. J. Biol. Chem. 266, 1551–1556.
Perona, R. and Serrano, R. (1988) Increased pH and tumorigenicity of fibroblasts expressing a yeast proton pump. Nature 334, 438–400.
Perona, R., Portillo, F., Giraldez, F., and Serrano, R. (1990) Transformation and pH hoemostasis of fibroblasts expressing yeast H(+)-ATPase containing site-directed mutations. Mol. Cell Biol 10, 4110–4115.
Peterson, E. P., Martinez, G. M., Martínez-Zaguilán, R., Perona, R., and Gillies, R. J. (1994) NIH 3T3 cells transfected with a yeast H (+)-ATPase have altered sensitivity to insulin, insulin growth, factor-I, and platelet-derived growth factor-AA. J. Cell Physiol. 159, 551–560.
Gillies, R. J., Martínez-Zaguilán, R., Martinez, G. M., Serrano, R., and Perona, R. (1990) Tumorigenic 3T3 cells maintain an alkaline intracellular pH under physiological conditions. Proc. Natl. Acad. Sci. USA 8, 7414–7418.
Heinz, A., Sachs, G., and Schafer, J. A. (1981) Evidence for activation of an active electrogenic proton pump in Ehrlich ascites tumor cells during glycolysis. J. Membr. Biol. 61, 143–153.
Ober, S. S., and Pardee, A. B. (1987) Intracellular pH is increased after trasformation of Chinese hamster embryo fibroblasts. Proc. Natl, Acad. Sci. USA 84, 2766–2770.
Cassel, D., Katz, M., and Rotman, M. (1986) Depletion of cellular ATP inhibits Na+/H+ antiport in cultured human cells. Modulation of the regulatory effect of intracellular protons on the antiporter activity. J. Biol. Chem. 261, 5460–5466.
Gillies, R. J., Ogino, T., Shulman, R. G., and Ward, D. C. (1982) 31P nuclear magnetic resonance evidence for the regulation of intracellular pH by Ehrlich ascites tumor cells. J. Cell Biol. 95, 24–28.
Manabe, T., Yoshimori, T., Henomatsu, N., and Tashiro, Y. (1993) Inhibitors of vacuolar type H +-ATPase suppresses proliferation of cultured cells. J. Cell Physiol. 157, 445–452.
Liotta, L. A., Rao, C. N., and Wewer, U. M. (1986) Biochemical interactions of tumor cells with the basement membrane. Annu. Rev. Biochem. 55, 1037–1057.
Yagel, S., Khokha, R., Denhardt, D. T., Kerbel, R. S., Parhar, R. S., and Lala, P. K. (1989) Mechanisms of cellular invasiveness: a comparison of amnion invasion in vitro and metastatic behavior in vivo. J. Natl. Cancer Inst. 81, 768–775.
Hendrix, M. J., Seftor, E. A., Seftor, R. E., and Fidler, I. J. (1987) A simple quantitative assay for studying the invasive potential of high and low human metastatic variants. Cancer Lett. 38, 137–147.
Marks, P. W. and Maxfield, F. R. (1990) Transient increases in cytosolic free calcium appear to be required for the migration of adherent human neutrophils. J. Cell Biol. 110, 43–52.
Milne, J. L. and Coukel, M. B. (1991) A Ca2+ transport system associated with the plasma membrane of Dictyostelium discoideum is activated by different, chemoattractant receptors. J. Cell Biol. 112, 103–110.
Savarese, D. M., Russell, J. T., Fatatis, A., and Liotta, L. A. (1992) Type IV collagen stimulates an increase in intracellular calcium. Potential role in tumor cell motility. J. Biol. Chem. 267, 21928–21935.
Korczak, B., Whale, C., and Kerbel, R. S. (1989) Possible involvement of Ca2+ mobilization and protein kinase C activation in the induction of spontaneous metastasis by mouse mammary adenocarcinoma cells. Cancer Res. 49, 2597–2602.
Miller, F. R. and Heppner, G. H. (1990) Cellular interactions in metastasis. Cancer Metastasis Rev. 9, 21–34.
Mareel, M. M., Van Roy, F. M., and De Baetselier, P. (1990) The invasive phenotypes. Cancer Metastasis Rev. 9, 45–62.
Stetler-Stevenson, W. G. (1990) Type IV collagenases in tumor invasion and metastasis. Cancer Metastasis Rev. 9, 289–303.
Lah, T. T., Kokalj-Kunovar, M., Strukelj, B., Pungercar, J., Barlic-Maganja, D., Drobnic-Kosorok, M., et al. (1992) Stefins and lysosomal cathepsins B, L and D in human breast carcinoma. Int. J. Cancer 50, 36–44.
Rozhin, J., Sameni, M., Ziegler, G., and Sloane, B. F. (1994) Pericellular pH affects distribution and secretion of cathepsin B in malignant cells. Cancer Res. 54, 6517–25.
Matrisian, L. M. (1992) The matrix-degrading metalloproteinases. Bioassays 14, 455–463.
Fidler, I. J. (1991) Cancer metastasis. Br. Med. Bull. 47, 157–177.
Vassalli, J. D., Sappino, A. P., and Belin, D. (1991) The plasminogen activator/plasmin system. J. Clin. Invest. 88, 1067–1072.
Morisset, M., Capony, F., and Rochefort, H. (1986) the 52-kDa estrogen-induced protein secreted by MCF-7 cells is a lysosomal acidic protease. Biochim. Biophys. Res. Commun. 138, 102–109.
Webb, S. D., Sherratt, J. A., and Fish, R. G. (1999) Alterations in proteolytic activity at low pH and its association with invasion: a theoretical model. Clin. Exp. Metastasis 17, 397–407.
Martínez-Zaguilán, R., Seftor, E. A., Seftor, R. E. B., Chu, Y. W., Gillies, R. J., and Hendrix, M. J. C. (1996) Acidic pH enhance the invasive behavior of human melanoma cells. Clin. Exp. Metastasis 14, 176–186.
Stubbs, M., McSheehy, P. M., Griffiths, J. R., and Bashford, C. L. (2000) Causes and consequences of tumour acidity and implications for treatment. Mol. Med. Today 6, 15–19.
Gatenby, R. A. and Gawlinski, E. T. (1996) A reaction-diffusion model of cancer invasion. Cancer Res. 56, 5745–5753.
Cuvier, C., Jang, A., and Hill, R. P. (1997) Exposure to hypoxia, glucose starvation and acidosis: effect on invasive capacity of murine tumor cells and correlation with cathepsin (L+B) secretion. Clin. Exp. Metastasis 15, 19–25.
Thomsen, P., Rudenko, O., Berezin, V., and Norrild, B. (1999) The HPV-16 E5 oncogene and bafilomycin A1 influence cell motility. Biochim. Biophys. Acta 1452, 285–295.
Hwang, E. S., Nottoli, T., and Dimaio, D. (1995) The HPV16 E5 protein: expression, detection, and stable complex formation with transmembrane proteins in COS cells. Virology 211, 227–233.
Yamshchikov, V. F. and Compans, R. W. (1995) Formation of the flavivirus envelope: role of the viral NS2B-NS3 protease. J. Virol. 69, 1995–2003.
Montcourrier, P., Mangeat, P. H., Valembois, C., Salazar, G., Sahuquet, A., Duperray, C., et al. (1994) Characterization of very acidic phagosomes in breast cancer cells and their association with invasion. J. Cell Sci. 107, 2381–91.
Gluck, S. L. (1992) The structure and biochemistry of the vacuolar H+-ATPase in proximal and distal urinary acidification. J. Bioenerg. Biomembr. 24, 351–360.
Brown, D., Lui, B., Gluck, S., and Sabolic, I. (1992) A plasma membrane proton ATPase in specialized cells of rat epididymis. Am. J. Physiol. 263, C913-C916.
Harvey, W. R. and Wieczorek, H. (1997) Animal plasma membrane energization by chemiosmotic H+ V-ATPases. J. Exp. Biol. 200, 203–216.
Martínez-Zaguilán, R., Martinez, G. M., Gomez, A., Hendrix, M. J. C., and Gillies, R. J. (1998) Distinct regulation of pHin and [Ca2+]in in human melanoma cells with different metastatic potential. J. Cell Physiol. 176, 196–205.
Vitavska, O., Wieczorek, H., and Merzendorfer, H. (2003) A novel role for subunit C in mediating binding of the H+-V-ATPase to the actin cytoskeleton. J. Biol. Chem. 278, 18499–18505.
Holliday, L. S., Lu, M., Lee, B. S., Nelson, R. D., Solivan, S., Zhang, L., et al. (2000) The aminoterminal domain of the B subunit of vacuolar H+-ATPase contains a filamentous actin binding site. J. Biol. Chem. 275, 32331–32337.
Lee, B. S., Gluck, S. L., and Holliday, L. S. (1999) Interaction between vacuolar H(+)-ATPase and microfilaments during osteoclast activation. J. Biol. Chem. 274, 29164–29171.
Toyomura, T., Murata, Y., Yamamoto, A., Oka, T., Sun-Wada, G-H., Wada, Y., and Futaie, M. (2003) From lysosomes to plasma membrane: localization of vacuolar type H+-ATPase with the a3 isoform during osteoclast differentiation. J. Biol. Chem. 278, 22023–22030.
Bernstein, B. W., Painter, W. B., Chen, H., Minamide, L. S., Abe, H., and Bamburg, J. R. (2000) Intracellular pH modulation of ADF/cofilin proteins. Cell Motil. Cytoskeleton 47, 319–336.
Denker, S. P., Huang, D. C., Orlowski, J., Furthmayr, H., and Barber, D. L. (2000) Direct binding of the Na−H exchanger NHE1 to ERM proteins regulates the cortical cytoskeleton and cell shape independently of H(+) translocation. Mol. Cell 6, 1425–1436.
Hawkins, M., Pope, B., Maciver, S. K., and Weeds, A. G. (1993) Human actin depolymerizing factor mediates a pH-sensitive destruction of actin filaments. Biochemistry 32, 9985–9993.
Hayden, S. M., Miller, P. S., Brauweiler, A., and Bamburg, J. R. (1993) Analysis of the interactions of actin depolymerizing factor with G- and F-actin. Biochemistry 32, 9994–10004.
Chan, A. Y., Bailly, M., Zebda, N., Segall, J. E., and Condeelis, J. S. (2000) Role of cofilin in epidermal growth factor-stimulated actin polymerization and lamellipod protrusion. J Cell Biol 148, 531–542.
Zebda, N., Bernard, O., Bailly, M., Welti, S., Lawrence, D. S., and Condeelis, J. S. (2000) Phosphorylation of ADF/cofilin abolishes EGF-induced actin nucleation at the leading edge and subsequent lamellipod extension. J. Cell Biol. 151, 1119–1128.
Ichetovkin, I., Grant, W., and Condeelis, J. (2002) Cofilin produces newly polymerized actin filaments that are preferred for dendritic nucleation by the Arp2/3 complex. Curr. Biol. 12, 79–84.
Sumi, T., Matsumoto, K., Takai, Y., and Nakamura, T. (1999) Cofilin phosphorylation and actin cytoskeletal dynamics regulated by rho- and Cdc42-activated LIM-kinase 2. J. Cell Biol. 147, 1519–1532.
Lawler, S. (1999) Regulation of actin dynamics: the LIM kinase connection. Curr Biol. 9, R800-R802.
Juranka, P. F., Zastawny, R. L., and Ling, V. (1989) P-glycoprotein: multidrug-resistance and a superfamily of membrane-associated transport proteins. FASEB J. 3, 2583–2592.
Roninson, I. B. (1987) Molecular mechanism of multidrug resistance in tumor cells. Clin. Physiol. Biochem. 5, 140–151.
Gottesman, M. M. and Pastan, I. (1993) Biochemistry of multidrug resistance mediated by the multidrug transporter. Annu. Rev. Biochem. 62, 385–427.
Altenberg, G. A., Young, G., Horton, J. K., Glass, D., Belli, J. A., and Reuss, L. (1993) Changes in intra- or extracellular pH do not mediate P-glycoprotein-dependent multidrug resistance. Proc. Natl. Acad. Sci. USA 90, 9735–9738.
Keizer, H. G. and Joenje, H. (1989) Increased cytosolic pH in multidrug-resistant human lung tumor cells: effect of verapamil. J. Natl. Cancer Inst. 81, 706–709.
Boscoboinik, D., Gupta, R. S., and Epand, R. M. (1990) Investigation of the relationship between altered intracellular pH and multidrug resistance in mammalian cells. Br. J. Cancer 61, 568–572.
Andersson, G. N., Torndal, U. B., and Eriksson, L. C. (1989) Decreased vacuolar acidification capacity in drug-resistant rat liver preneoplastic nodules. Cancer Res. 49, 3765–3769.
Marsh, W., Sicheri, D., and Center, M. S. (1986) Isolation and characterization of adriamycin-resistant HL-60 cells which are not defective in the initial intracellular accumulation of drug. Cancer Res. 46, 4053–4057.
McGrath, T. and Center, M. S. (1987) Adriamycin resistance in HL60 cells in the absence of detectable P-glycoprotein. Biochem. Biophys. Res. Commun. 145, 1171–1176.
Marquardt, D. and Center, M. S. (1991) Involvement of vacuolar H+-adenosine triphosphatase activity in multidrug resistance in HL60 cells. J. Natl. Cancer Inst. 83, 1098–1102.
Martínez-Zaguilán, R., Raghunand, N., Lynch, R. M. Bellamy, W., Martinez, G. M., Rojas, B., et al. (1999) pH and drug resistance. I. Functional expression of plasmalemmal V-type H+-ATPase in drug-resistant human breast carcinoma cell lines. Biochem. Pharmacol. 57, 1037–1046.
Raghunand, N., Martínez-Zaguilán, R., Wright, S. H., and Gillies, R. J. (1999) pH and drug resistance. II. Turnover of acidic vesicles and resistance to weakly basic chemotherapeutic drugs. Biochem. Pharmacol. 57, 1047–1058.
Versantvoort, C. H., Broxterman, H. J., Pinedo, H. M., de Vries, E. G., Feller, N., Kuiper, C. M., et al. (1992) Energy-dependent processes involved in reduced drug accumulation in multidrug-resistant human lung cancer cell lines without P-glycoprotein expression. Cancer Res. 52, 17–23.
Halaban, R., Patton, R. S., Cheng, E., Svedine, S., Trombetta, E. S., Wahl, M. L., et al. (2002) Abnormal acidification of melanoma cells induces tyrosinase retention in the early secretory pathway. J. Biol. Chem. 277, 14821–14828.
Laurencot, C. M., Andrews, P. A., and Kennedy, K. A. (1995) Inhibitors of intracellular pH regulation induce cisplatin resistance in EMT6 mouse mammary tumor cells. Oncol Res. 7, 363–369.
Xu, J., Feng, H. T., Wang, C., Yip, K. H., Pavlos, N., Papadimitriou, J. M., et al. (2003) Effects of Bafilomycin A1: an inhibitor of vacuolar H (+)-ATPases on endocytosis and apoptosis in RAW cells and RAW cell-derived osteoclasts. J. Cell Biochem. 88, 1256–1264.
Tanigaki, K., Sasaki, S., and Ohkuma, S. (2003) In bafilomycin A1-resistant cells, bafilomycin A1 raised lysosomal pH and both prodigiosins and concanamycin A inhibited growth through apoptosis. FEBS Lett. 537, 79–84.
Nishihara, T., Akifusa, S., Koseki, T., Kato, S., Muro, M., and Hanada, N. (1995) Specific inhibitors of vacuolar type H(+)-ATPases induce apoptotic cell death. Biochem. Biophys. Res Commun. 212, 255–262.
Akifusa, S., Ohguchi, M., Koseki, T., Nara, K., Semba, I., Yamato, K., et al. (1998) Increase in Bcl-2 level promoted by CD40 ligation correlates with inhibition of B cell apoptosis induced by vacuolar type H(+)-ATPase inhibitor. Exp Cell Res. 238, 82–89.
Ishisaki, A., Hashimoto, S., Amagasa, T., and Nishihara, T. (1999) Caspase-3 activation during the process of apoptosis induced by a vacuolar type H(+)-ATPase inhibitor. Biol Cell. 91, 507–513.
Aiko, K., Tsujisawa, T., Koseki, T., Hashimoto, S., Morimoto, Y., Amagasa, T., et al. (2002) Involvement of cytochrome c and caspases in apoptotic cell death of human submandibular gland ductal cells induced by concanamycin A. Cell Signal 14, 717–722.
Torigoe, T., Izumi, H., Ishiguchi, H., Uramoto, H., Murakami, T., Ise, T., et al. (2002) Enhanced expression of the human vacuolar H+-ATPase c subunit gene (ATP6L) in response to anticancer agents. J Biol Chem. 277, 36534–36543.
Yoshimoto, Y. and Imoto, M. (2002) Induction of EGF-dependent apoptosis by vacuolartype H+-ATPase inhibitors in A431 cells overex-pressing the EGF receptor. Exp Cell Res. 279, 118–127.
Author information
Authors and Affiliations
Corresponding author
Additional information
F in F0F1 ATPase is the coupling energy factor.
Rights and permissions
About this article
Cite this article
Sennoune, S.R., Luo, D. & Martinez-Zaguilán, R. Plasmalemmal vacuolar-type H+-ATPase in cancer biology. Cell Biochem Biophys 40, 185–206 (2004). https://doi.org/10.1385/CBB:40:2:185
Issue Date:
DOI: https://doi.org/10.1385/CBB:40:2:185