Summary
Background. Peroxisome proliferator-activated receptor γ (PPARγ) is expressed in certain human cancers; ligand-induced PPARγ activation can result in growth inhibition and differentiation in these cells. However, the precise mechanism for the antiproliferative effect of PPARγ ligands is not entirely known.
Aim of Study. The purpose of this study was to examine the effect of PPARγ ligands on pancreatic cancer cell growth and invasiveness.
Methods. The effect of two PPARγligands, 15 deoxy-Δ12,14prostaglandin J2 (15d-PGJ2) and ciglitazone, on the growth of four human pancreatic cancer cell lines (BxPC-3, MIA PaCa-2, Panc-1, and L3.6) was assessed. Expression of cell-cycle and apoptotic-related proteins was measured. Finally, the effect of 15d-PGJ2 on pancreatic cancer cell invasiveness and matrix metalloproteinase expression was determined.
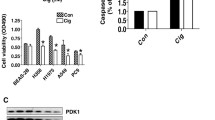
Results. Both 15d-PGJ2 and ciglitazone inhibited the growth of all four pancreatic cancer cell lines in a dose- and time-dependent fashion. Treatment of BxPC-3 cells with 15d-PGJ2 resulted in a time-dependent decrease in cyclin D1 expression associated with a concomitant induction of p21waf1 and p27kip1. In addition, 15d-PGJ2 treatment induced apoptosis through activation of caspase-8, -9, and -3. Moreover, pancreatic cancer cell invasiveness was significantly suppressed after treatment with a nontoxic dose of 15d-PGJ2, which was associated with a reduction of MMP-2 and MMP-9 protein levels and activity.
Conclusion. These results demonstrate that PPARγ ligands have the dual advantage of inhibiting pancreatic cancer cell growth while reducing the invasiveness of the tumor cells, suggesting a potential role for these agents in the adjuvant treatment of pancreatic cancer.
Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA. Cancer statistics, 1999. CA Cancer J Clin 1999;49:8–31.
Büchler M, Friess H, Schultheiss KH, Gebhardt C, Kubel R, Muhrer KH, et al. A randomized controlled trial of adjuvant immunotherapy (murine monoclonal antibody 494/32) in resectable pancreatic cancer. Cancer 1991;68:1507–1512.
Link KH, Gansauge F, Pillasch J, Beger HG. Multimodal therapies in ductal pancreatic cancer. The future. Int J Pancreatol 1997;21:71–83.
Heinemann V. Gemcitabine: progress in the treatment of pancreatic cancer. Oncology 2001;60:8–18.
Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K, et al. The nuclear receptor superfamily: the second decade. Cell 1995;83:835–839.
Chawla A, Schwarz EJ, Dimaculangan DD, Lazar MA. Peroxisome proliferator-activated receptor (PPAR) γ: adipose-predominant expression and induction early in adipocyte differentiation. Endocrinology 1994;135:798–800.
Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone. N J Med 1994;331:1188–1193.
Ricote M, Li AC, Willson TM, Kelly CJ, Glass CK. The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 1998;391:79–82.
Jiang C, Ting AT, Seed B. PPAR-γ agonists inhibit production of monocyte inflammatory cytokines. Nature 1998;391:82–86.
Kliewer SA, Lehmann JM, Willson TM. Orphan nuclear receptors: shifting endocrinology into reverse. Science 1999;284:757–760.
Forman BM, Tontonoz P, Chen J, Brun RP, Spiegelman BM, Evans RM. 15-Deoxy-Δ12,14-prostaglandin J2 is a ligand for the adipocyte determination factor PPARγ. Cell 1995;83:803–812.
Kliewer SA, Lenhard JM, Willson TM, Patel I, Morris DC, Lehmann JM. A prostaglandin J2 metabolite binds peroxisome proliferator-activated receptor γ and promotes adipocyte differentiation. Cell 1995;83:813–819.
Tontonoz P, Singer S, Forman BM, Sarraf P, Fletcher JA, Fletcher CD, et al. Terminal differentiation of human liposarcoma cells induced by ligands for peroxisome proliferator-activated receptor γ and the retinoid X receptor. Proc Natl Acad Sci USA 1997;94:237–241.
Sarraf P, Mueller E, Jones D, King FJ, DeAngelo DJ, Partridge JB, et al. Differentiation and reversal of malignant changes in colon cancer through PPARγ. Nat Med 1998;4:1046–1052.
Mueller E, Sarraf P, Tontonoz P, Evans RM, Martin KJ, Zhang M, et al. Terminal differentiation of human breast cancer through PPAR γ. Mol Cell 1998;1:465–470.
Chang TH, Szabo E. Induction of differentiation and apoptosis by ligands of peroxisome proliferator-activated receptor γ in non-small cell lung cancer. Cancer Res 2000;60:1129–1138.
Sano H, Kawahito Y, Wilder RL, Hashiramoto A, Mukai S, Asai K, et al. Expression of cyclooxygenase-1 and -2 in human colorectal cancer. Cancer Res 1995;55:3785–3789.
Kawamori T, Rao CV, Seibert K, Reddy BS. Chemopreventive activity of celecoxib, a specific cyclooxygenase-2 inhibitor, against colon carcinogenesis. Cancer Res 1998;58:409–412.
Oshima M, Dinchuk JE, Kargman SL, Oshima H, Hancock B, Kwong E, et al. Suppression of intestinal polyposis in Apc delta716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 1996;87:803–809.
Inoue H, Tanabe T, Umesono K. Feedback control of cyclooxygenase-2 expression through PPARγ. J Biol Chem 2000;275:28028–28032.
Meade EA, McIntyre TM, Zimmerman GA, Prescott SM. Peroxisome proliferators enhance cyclooxygenase-2 expression in epithelial cells. J Biol Chem 1999;274:8328–8334.
Bruns CJ, Harbison MT, Kuniyasu H, Eue I, Fidler IJ. In vivo selection and characterization of metastatic variants from human pancreatic adenocarcinoma by using orthotopic implantation in nude mice. Neoplasia 1999;1:50–62.
Evers BM, Zhou Z, Celano P, Li J. The neurotensin gene is a downstream target for Ras activation. J Clin Invest 1995;95:2822–2830.
Litvak DA, Evers BM, Hwang KO, Hellmich MR, Ko TC, Townsend CM, Jr. Butyrate-induced differentiation of Caco-2 cells is associated with apoptosis and early induction of p21 Waf1/Cip1 and p27Kip1. Surgery 1998;124:161–169; discussion 169–170.
Albini A, Iwamoto Y, Kleinman HK, Martin GR, Aronson SA, Kozlowski JM, McEwan RN. A rapid in vitro assay for quantitating the invasive potential of tumor cells. Cancer Res 1987;47:3239–3245.
Vamecq J, Latruffe N. Medical significance of peroxisome proliferator-activated receptors. Lancer 1999;354:141–148.
Kubota T, Koshizuka K, Williamson EA, Asou H, Said JW, Holden S, et al. Ligand for peroxisome proliferatoractivated receptor γ (troglitazone) has potent antitumor effect against human prostate cancer both in vitro and in vivo. Cancer Res 1998;58:3344–3352.
Chinery R, Coffey RJ, Graves-Deal R, Kirkland SC, Sanchez SC, Zackert WE, et al. Prostaglandin J2 and 15-deoxy-Δ12,14-prostaglandin J2 induce proliferation of cyclooxygenase-depleted colorectal cancer cells. Cancer Res 1999;59:2739–2746.
Tsujii M, DuBois RN. Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell 1995;83:493–501.
Tsujii M, Kawano S, DuBois RN. Cyclooxygenase-2 expression in human colon cancer cells increases metastatic potential. Proc Natl Acad Sci USA 1997;94:3336–3340.
Molina MA, Sitja-Arnau M, Lemoine MG, Frazier ML, Sinicrope FA. Increased cyclooxygenase-2 expression in human pancreatic carcinomas and cell lines: growth inhibition by nonsteroidal anti-inflammatory drugs. Cancer Res 1999;59:4356–4362.
Yip-Schneider MT, Barnard DS, Billings SD, Cheng L, Heilman DK, Lin A, et al. Cyclooxygenase-2 expression in human pancreatic adenocarcinomas. Carcinogenesis 2000;21:139–146.
Buolamwini JK. Cell cycle molecular targets in novel anticancer drug discovery. Curr Pharm Des 2000;6:379–392.
Hunter T, Pines J. Cyclins and cancer. II: Cyclin D and CDK inhibitors come of age. Cell 1994;79:573–582.
Elstner E, Muller C, Koshizuka K, Williamson EA, Park D, Asou H, et al. Ligands for peroxisome proliferator-activated receptor γ and retinoic acid receptor inhibit growth and induce apoptosis of human breast cancer cells in vitro and in BNX mice. Proc Natl Acad Sci USA 1998;95:8806–8811.
Tsubouchi Y, Sano H, Kawahito Y, Mukai S, Yamada R, Kohno M, et al. Inhibition of human lung cancer cell growth by the peroxisome proliferator-activated receptor-γ agonists through induction of apoptosis. Biochem Biophys Res Commun 2000;270:400–405.
Foglieni C, Meoni C, Davalli AM. Fluorescent dyes for cell viability: an application on prefixed conditions. Histochem Cell Biol 2001;115:223–229.
Warshaw AL, Fernandez-del Castillo C. Pancreatic carcinoma. N Engl J Med 1992;326:455–465.
Jimenez RE, Hartwig W, Antoniu BA, Compton CC, Warshaw AL, Fernandez-del Castillo C. Effect of matrix metalloproteinase inhibition on pancreatic cancer invasion and metastasis: an additive strategy for cancer control. Ann Surg 2000;231:644–654.
Birchmeier W, Hulsken J, Behrens J. Adherens junction proteins in tumour progression. Cancer Surv 1995;24:129–140.
Elnemr A, Ohta T, Iwata K, Ninomia I, Fushida S, Nishimura G, et al. PPAR γ ligand (thiazolidinedione) induces growth arrest and differentiation markers of human pancreatic cancer cells. Int J Oncol 2000;17:1157–1164.
Motomura W, Okumura T, Takahashi N, Obara T, Kohgo Y. Activation of peroxisome proliferator-activated receptor γ by troglitazone inhibits cell growth through the increase of p27Kip1 in human. Pancreatic carcinoma cells. Cancer Res 2000;60:5558–5564.
Sato H, Ishihara S, Kawashima K, Moriyama N, Suetsugu H, Kazumori H, et al. Expression of peroxisome proliferator-activated receptor (PPAR)γ in gastric cancer and inhibitory effects of PPARγ agonists. Br J Cancer 2000;83:1394–1400.
Mueller E, Smith M, Sarraf P, Kroll T, Aiyer A, Kaufman DS, et al. Effects of ligand activation of peroxisome proliferator-activated receptor γ in human prostate cancer. Proc Natl Acad Sci USA 2000;97:10990–10995.
Halachmi S, Marden E, Martin G, MacKay H, Abbondanza C, Brown M. Estrogen receptor-associated proteins: possible mediators of hormone-induced transcription. Science 1994;264:1455–1458.
Horlein AJ, Naar AM, Heinzel T, Torchia J, Gloss B, Kurokawa R, et al. Ligand-independent repression by the thyroid hormone receptor mediated by a nuclear receptor co-repressor. Nature 1995;377:397–404.
DuBois RN, Awad J, Morrow J, Roberts LJ, 2nd, Bishop PR. Regulation of eicosanoid production and mitogenesis in rat intestinal epithelial cells by transforming growth factor-α and phorbol ester. J Clin Invest 1994;93:493–498.
Subbaramaiah K, Telang N, Ramonetti JT, Araki R, DeVito B, Weksler BB, Dannenberg AJ. Transcription of cyclooxygenase-2 is enhanced in transformed mammary epithelial cells. Cancer Res 1996;56:4424–4429.
Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994; 107: 1183–1188.
Ristimaki A, Honkanen N, Jankala H, Sipponen P, Harkonen M. Expression of cyclooxygenase-2 in human gastric carcinoma. Cancer Res 1997;57:1276–1280.
Zimmermann KC, Sarbia M, Weber AA, Borchard F, Gabbert HE, Schrör K. Cyclooxygenase-2 expression in human esophageal carcinoma. Cancer Res 1999;59:198–204.
Tsujii M, Kawano S, Tsuji S, Sawaoka H, Hori M, DuBois RN. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell 1998;93:705–716.
Subbaramaiah K, Lin DT, Hart JC, Dannenberg AJ. Peroxisome proliferator-activated receptor γ ligands suppress the transcriptional activation of cyclooxygenase-2. Evidence for involvement of activator protein-1 and CREB-binding protein/p300. J Biol Chem 2001;276:12440–12448.
Sherr CJ. G1 phase progression: cycling on cue. Cell 1994;79:551–555.
Gansauge S, Gansauge F, Ramadani M, Stobbe H, Rau B, Harada N, Beger HG. Overexpression of cyclin D1 in human pancreatic carcinoma is associated with poor prognosis. Cancer Res 1997;57:1634–1637.
Lu CD, Morita S, Ishibashi T, Hara H, Isozaki H, Tanigawa N. Loss of p27Kip1 expression independently predicts poor prognosis for patients with resectable pancreatic adenocarcinoma. Cancer 1999;85:1250–1260.
Nio Y, Dong M, Uegaki K, Hirahara N, Minari Y, Sasaki S, et al. Comparative significance of p53 and WAF/1-p21 expression on the efficacy of adjuvant chemotherapy for resectable invasive ductal carcinoma of the pancreas. Pancreas 1999;18:117–126.
Kornmann M, Arber N, Korc M. Inhibition of basal and mitogen-stimulated pancreatic cancer cell growth by cyclin D1 antisense is associated with loss of tumorigenicity and potentiation of cytotoxicity to cisplatinum. J Clin Invest 1998;101:344–352.
Joshi US, Dergham ST, Chen YQ, Dugan MC, Crissman JD, Vaitkevicius VK, Sarkar FH. Inhibition of pancreatic tumor cell growth in culture by p21WAF1 recombinant adenovirus. Pancreas 1998;16:107–113.
Takahashi N, Okumura T, Motomura W, Fujimoto Y, Kawabata I, Kohgo Y. Activation of PPARγ inhibits cell growth and induces apoptosis in human gastric cancer cells. FEBS Lett 1999;455:135–139.
Butler R, Mitchell SH, Tindall DJ, Young CY. Nonapoptotic cell death associated with S-phase arrest of prostate cancer cells via the peroxisome proliferator-activated receptor γ ligand, 15-deoxy-Δ12,14-prostaglandin J2. Cell Growth Differ 2000;11:49–61.
Rossi A, Kapahi P, Natoli G, Takahashi T, Chen Y, Karin M, Santoro MG. Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IkappaB kinase. Nature 2000;403:103–108.
Israels LG, Israels ED. Apoptosis. Oncologist 1999;4:332–339.
Thornberry NA, Lazebnik Y. Caspases: enemies within. Science 1998;281:1312–1316.
Gress TM, Muller-Pillasch F, Lerch MM, Friess H, Buchler M, Adler G. Expression and in-situ localization of genes coding for extracellular matrix proteins and extracellular matrix degrading proteases in pancreatic cancer. Int J Cancer 1995;62:407–413.
Marx N, Schonbeck U, Lazar MA, Libby P, Plutzky J. Peroxisome proliferator-activated receptor gamma activators inhibit gene expression and migration in human vascular smooth muscle cells. Circ Res 1998;83:1097–1103.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hashimoto, K., Ethridge, R.T. & Evers, B.M. Peroxisome proliferator-activated receptor γ ligand inhibits cell growth and invasion of human pancreatic cancer cells. Int J Gastrointest Canc 32, 7–22 (2002). https://doi.org/10.1385/IJGC:32:1:7
Issue Date:
DOI: https://doi.org/10.1385/IJGC:32:1:7