Abstract
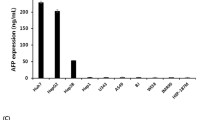
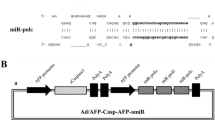
Hepatocellular carcinoma (HCC) is one of the most common malignancies with poor prognosis and is highly amenable to the development of novel therapeutic strategy. The human alpha-fetoprotein (AFP) gene is normally expressed in fetal liver and is transcriptionally silent in adult liver but overexpressed in HCC. In order to destroy AFP-producing HCC specifically, replication defective adenoviral vectors containing the transcriptional control elements of the AFP gene were designed. Expression of suicide genes by the AFP promoter/enhancer induced prodrug sensitivity in AFP (+) cells but not AFP (−) cells. The expression of suicide genes by ubiquitous promoter, however, showed no selectivity after prodrug treatment. Adenoviral vector transduced genes efficiently not only in vitro but also in vivo, and AFP-producing HCC xenografts regressed by transduction with transcriptionally targeted vectors and subsequent systemic administration of prodrug in animal model. Utilization of the transcriptional regulatory element to drive drug sensitive genes can be a promising strategy for cancer specific therapy.
Similar content being viewed by others
References
Moolten, F. L. (1994) Drug sensitivity (“suicide”) genes for selective cancer chemotherapy. Cancer Gene Ther. 1, 279–287.
Roth, J. A. and Cristiano, R. J. (1997). Gene therapy for cancer: what have we done and where are we going? J. Natl. Cancer Inst. 89, 21–39.
Calabresi, P. and Chabner, B. A. (1995) Chemotherapy of neoplastic diseases. in Goodman and Gilman’s The Pharmacological Basis of Therapeutics (9th ed.) (Hardman, J. G., Limbird, L. E., Molinoff, P. B., Ruddon, R. W., and Gilman, A. G. eds.) Pergamon, New York, pp. 1225–1287.
Kanai, F., Kawakami, T., Hamada, H., Sadata, A., Yoshida, Y., Tanaka, T., et al. (1998) Adenovirus-mediated transduction of Escherichia coli uracil phosphoribosyl-transferase gene sensitizes cancer cells to low concentration of 5-fluorouracil, Cancer Res. 58, 1946–1951.
Miller, N. and Whelan, J. (1997). Progress in transcriptionally targeted and regulatable vectors for genetic therapy. Hum. Gene Ther. 8, 803–815.
Niwa, H., Yamamura, K. I., and Miyazaki, J. I. (1991). Efficient selection for high expression transfectants with a novel eukaryotic vector. Gene. 108, 193–200.
Kanegae, Y., Lee, G., Sato, Y., Tanaka, M., Nakai, M., Sakaki, T., et al. (1995) Efficient gene activation in mammalian cells by using recombinant adenovirus expressing site-specific Cre recombinase. Nucleic Acids Res. 23(19), 3816–3821.
Miyake, S., Makimura, M., Kanegae, Y., Harada, S., Sato, Y., Takamori, K., et al. (1996) Efficient generation of recombinant adenoviruses using adenovirus DNA-terminal protein complex and a cosmid bearing full-length virus genome. Proc. Natl. Acad. Sci. USA 93(3), 1320–1324.
Kanai, F., Shiratori, Y., Yoshida, Y., Wakimoto, H., Hamada, H., Kanegae, Y., et al. (1996) Gene therapy for α-fetoprotein producing human hepatoma cells by adenovirus-mediated transfer of herpes simplex virus thymidine kinase gene. Hepatology. 23, 1359–1368.
Kanai, F., Lan, K.-H., Shiratori, Y., Tanaka, T., Ohashi, M., Yoshida, Y., et al. (1997). In vivo gene therapy for α-fetoprotein producing hepatocellular carcinoma by adenovirus-mediated transfer of cytosine deaminase gene. Cancer Res. 57, 461–465.
Lan, K. H., Kanai, F., Shiratori, Y., Ohashi, M., Tanaka, T., Yoshida, Y., et al. (1997). In vivo selective gene expression and therapy mediated by adenoviral vector for human carcinoembryonic antigen-producing gastric carcinoma. Cancer Res. 57, 4279–4284.
Kaneko, S., Hallenbeck, P., Kotani, T., Nakabayashi, H., McGarrity, G., Tamaoki, T., et al. (1995). Adenovirus-mediated gene therapy of hepatocellular carcinoma using cancer-specific gene expression. Cancer Res. 55, 5283–5287.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kanai, F. Transcriptional targeted gene therapy for hepatocellular carcinoma by adenovirus vector. Mol Biotechnol 18, 243–250 (2001). https://doi.org/10.1385/MB:18:3:243
Issue Date:
DOI: https://doi.org/10.1385/MB:18:3:243