Abstract
The availability of the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors has revolutionised the treatment of lipid abnormalities in patients at risk for the development of coronary atherosclerosis. The relatively widespread experience with HMG-CoA therapy has allowed a clear picture to emerge concerning the relative tolerability of these agents. While HMG-CoA reductase inhibitors have been shown to decrease complications from atherosclerosis and to improve total mortality, concern has been raised as to the long term safety of these agents. They came under close scrutiny in early trials because ocular complications had been seen with older inhibitors of cholesterol synthesis. However, extensive evaluation demonstrated no significant adverse alteration of ophthalmological function by the HMG-CoA reductase inhibitors.
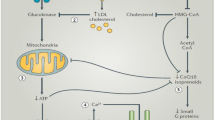
Extensive experience with the potential adverse effect of the HMG-CoA reductase inhibitors on hepatic function has accumulated. The effect on hepatic function for the various HMG-CoA reductase inhibitors is roughly dose-related and 1 to 3% of patients experience an increase in hepatic enzyme levels. The majority of liver abnormalities occur within the first 3 months of therapy and require monitoring. Rhabdomyolysis is an uncommon syndrome and occurs in approximately 0.1% of patients who receive HMG-CoA reductase inhibitor monotherapy. However, the incidence is increased when HMG-CoA reductase inhibitors are used in combination with agents that share a common metabolic path.
The role of the cytochrome P450 (CYP) enzyme system in drug-drug interactions involving HMG-CoA reductase inhibitors has been extensively studied. Atorvastatin, cerivastatin, lovastatin and simvastatin are predominantly metabolised by the CYP3A4 isozyme. Fluvastatin has several metabolic pathways which involve the CYP enzyme system. Pravastatin is not significantly metabolised by this enzyme and thus has theoretical advantage in combination therapy. The major interactions with HMG-CoA reductase inhibitors in combination therapy involving rhabdomyolysis include fibric acid derivatives, erythromycin, cyclosporin and fluconazole.
Additional concern has been raised relative to overzealous lowering of cholesterol which could occur due to the potency of therapy with these agents. Currently, there is no evidence from clinical trials of an increase in cardiovascular or total mortality associated with potent low density lipoprotein reduction. However, a threshold effect had been inferred by retrospective analysis of the Cholesterol and Recurrent Events study utilising pravastatin and the role of aggressive lipid therapy is currently being addressed in several large scale trials.






Similar content being viewed by others
References
Frick MH, Elo O, Haapa K, et al. Helsinki Heart Study: Primary prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N Engl J Med 1987; 317: 1237–45
Lipid Research Clinics Program. The Lipid Research Clinics Program Trial results I. The relationship of reduction in incidence of coronary heart disease to cholesterol lowering. JAMA 1984; 251: 365–74
Scandinavian Simvastatin Survival Study Group. Randomized trial of cholesterol lowering in 444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 1994; 344: 1383–9
The Longterm Intervention with Pravastatin in Ischemic Disease (LIPID) Study Group. Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease in a broad range of initial cholesterol levels. N Engl JMed 1998; 339: 1349–57
Gerson RJ, MacDonald JS, Alberts AW, et al. On the etiology of subcapsular lenticular opacities produced in dogs receiving HMG CoA reductase inhibitors. Exp Eye Res 1990; 50(1): 65–78
Laties AM, Shear CL, Leppa EA, et al. Expanded clinical evaluation of lovastatin (EXCEL) study results. II. Assessment of the human lens after 48 weeks of treatment with lovastatin therapy. Am J Cardiol 1991; 67(6): 447–53
Lundh BL, Nilsson SE. Lens changes in matched normals and hyperlipidemic patients treated with simvastatin for two years. Acta Ophthalmol 1990; 68(6): 658–60
Schmid J, Schmidt C, Hockwi O. No lens changes caused by simvastatin results from a prospective drug safety study. Lens Eye Toxic Res 1990; 7(3-4): 643–50
Harris ML, Bron AJ, Brown NA, et al., Oxford Cholesterol Study Group. Absence of effect of simvastatin on the progression of lens opacities in a randomized, placebo-controlled study. Br J Ophthalmol 1995; 79(11): 996–1002
Schaefer EJ. HMG CoA reductase inhibitors for hypercholesterolemia [letter]. N Engl J Med 1998; 319: 122
Guillot F, Misslin P, Lemaire M. Comparison of fluvastatin and lovastatin blood-brain barrier transfer utilizing in vitro and in vivo methods. J Cardiovasc Pharmacol 1993; 21(2): 339–46
Harrison RW, Ashton CH. Do cholesterol lowering agents affect brain activity? A comparison of simvastatin, pravastatin and placebo in healthy volunteers. Br J Clin Pharmacol 1994; 37(3): 231–6
Partinen M, Pihl S, Strandberg TE. Comparison of effects on sleep of lovastatin, pravastatin and placebo in patients with hypercholesterolemia. Am J Cardiol 1994; 73: 876–80
Eckernas SA, Roos BE, Kvidal P, et al. The effects of simvastatin and pravastatin on objective and subjective measures of nocturnal sleep: a comparison of two structurally different HMG CoA reductase inhibitors in patients with primary moderate hypercholesterolemia. Br J Clin Pharmacol 1993; 35(3): 284–9
Bradford RH, Schear CL, Chremos AN, et al. Expanded clinical evaluation of lovastatin (EXCEL) study results: two-year efficacy and safety follow-up. Am J Cardiol 1994; 74(7): 667–73
Kostis JB, Rosen RC, Wilson AC. Central nervous system effects of HMG CoA reductase inhibitors: lovastatin and pravastatin on sleep and cognitive performance in patients with hypercholesterolemia. J Clin Pharmacol 1994; 34(10): 989–96
Gengo F, Cwudzinski D, Kinkel P, et al. Effects of treatment of lovastatin and pravastatin on daytime cognitive performance. Clin Cardiol 1995; 18(4): 209–14
Lovastatin Study Groups I-IV. Lovastatin 5-year safety and efficacy study. Arch Intern Med 1993; 153 (9): 1079–87
Tobert J. Efficacy and long term adverse effect pattern of lovastatin. Am J Cardiol 1988; 62(15): 28J–34J
Boccuzzi SJ, Bocanegra TS, Walker JF, et al. Longterm safety and efficacy profile of simvastatin. Am J Cardiol 1991; 68(11): 1127–31
Molgaard J, Lundh EL, von Schenck H, et al. Longterm efficacy and safety of simvastatin alone and in combination therapy and treatment of hypercholesterolemia. Atherosclerosis 1991; 91Suppl.: S21–8
Black DM, Bakker-Arkema RG, Nawrocki JW. An overview of the clinical safety profile of atorvastatin, a new HMG CoA reductase inhibitor. Arch Intern Med 1998; 158(6): 577–84
Dart A, Jerums G, Nicholson G, et al. A multi-center, double-blind, one-year study comparing safety and efficacy of atorvastatin versus simvastatin in hypercholesterolemia. Am J Cardiol 1997; 80(1): 39–44
Davidson M, McKenney J, Stein E, et al. Comparison of one-year efficacy and safety of atorvastatin versus lovastatin in primary hyper-cholesterolemia. Am J Cardiol 1997; 79(11): 1475–81
Stein E. Cerivastatin in primary hyperlipidemia: a multicenter analysis of efficacy and safety. Atherosclerosis 1998; 139Suppl. 1: S15–22
Davignon J, Hanefeld M, Nakaya N, et al. Clinical efficacy and safety of cerivastatin: summary of pivotal phase IIB/III studies. Am J Cardiol 1998; 82(4B): 32J–9J
Jokubaitis LA. Updated clinical safety experience with fluvastatin. Am J Cardiol 1994; 73(14): 18D–24D
Peters TK, Muratti EN, Mehra M. Fluvastatin and primary hypercholesterolemia: efficacy and safety in patients at high risk. An analysis of a clinical trial database. Am J Med 1994; 96(6A): 79S–83S
Banga JD, Jacotot B, Phister P, et al., French-Dutch Fluvastatin Study Group. Longterm treatment of hyper-cholesterolemia with fluvastatin: a 52-week multicenter safety and efficacy study. Am J Med 1994; 96(6A): 87S–93S
Knipscheer HC, Boelen CC, Kastelein JJ, et al. Short-term efficacy and safety of pravastatin in 72 children with familial hyper-cholesterolemia. Pediatr Res 1996; 39(5): 867–71
Simons LA, Nestel PJ, Clifton P, et al. Treatment of primary hypercholesterolemia with pravastatin: efficacy and safety over three years. Med J Aust 1992; 157(9): 584–9
Santinga JT, Rosman HS, Rubenfire M, et al. Efficacy and safety of pravastatin in the longterm treatment of elderly patients with hyper-cholesterolemia. Am J Med 1994; 96(6): 509–15
Behounnek BD, McGovern ME, Kassler-Taub KB, et al., Pravastatin Multi-national Study Group for Diabetes. A multinational study of the effects of low dose pravastatin in patients with noninsulin dependent diabetes mellitus and hypercholesterolemia. Clin Cardiol 1994; 17(10): 558–62
Raskin P, Ganda OP, Schwartz S, et al. Efficacy and safety of pravastatin in the treatment of patients with type I or type II diabetes mellitus and hypercholesterolemia. Am J Med 1995; 99(4): 362–9
Davignon J, Roederer G, Montighy M, et al. Comparative efficacy and safety of pravastatin, nicotinic acid and the two combined in patients with hypercholesterolemia. Am J Cardiol 1994; 73(5): 339–45
Stalenhoef AF, Lansberg PJ, Kroon AA, et al. Treatment of primary hypercholesterolemia. Short-term efficacy and safety of increasing doses of simvastatin and pravastatin: a double-blind comparative study. J Intern Med 1993; 234(1): 77–82
Jackobson TA, Chin MM, Curry CL, et al. Efficacy and safety of pravastatin in African-Americans with primary hypercholesterolemia. Arch Intern Med 1995; 155(17): 1900–6
Wiklund O, Angelin B, Bergman M, et al. Pravastatin and gemfibrozil alone and in combination for the treatment of hypercholesterolemia. Am J Med 1993; 94(1): 13–20
Downs JR, Clearfield M, Weis S, et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels. JAMA 1998; 279: 1615–22
Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. N Engl J Med 1996; 335: 1001–9
Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med 1995; 333: 1301–7
Hodis HN. Acute hepatic failure associated with the use of low dose sustained release niacin. JAMA 1990; 264(2): 181
Grob D. Rhabdomyolysis and drug relatedmyopathies. Curr Opin Rheumatol 1990; 2(6): 908–15
Lofberg M, Jankala H, Paetau A, et al. Metabolic causes of recurrent rhabdomyolysis. ActaNeurol Scand 1998; 98(4): 268–75
Flint OP, Masters BA, Gregg RE, et al. HMG CoA reductase inhibitor induced myotoxicity: pravastatin and lovastatin inhibit the geranylgeranylation of low molecular weight proteins in neonatal rat muscle cell culture. Toxicol Appl Pharmacol 1997; 145(1): 99–110
Gadbut AP, Caruso AP, Galper JB. Differential sensitivity of C3-C12 striated muscle to lovastatin and pravastatin. J Mol Cell Cardiol 1995, 10: 2397–402
Nakagawa H, Mutoh T, Kumano T, et al. HMG CoA reductase inhibitor induced L6 myoblast cell death: involvement of the phosphatidylinositol 3-kinase pathway. FEBS Lett 1998; 438(3): 289–92
Watts GF, Castelluccio C, Rice-Evans C, et al. Plasma coenzyme Q concentrations in patients treated with simvastatin. J Clin Pathol 1993; 46(11): 1055–7
De Pinieux G, Chariot P, Ammi-Said M, et al. Lipid lowering drugs and mitochondrial function: effects of HMG CoA reductase inhibitors and serum ubiquinone and blood lactate/pyruvate ratio. Br J Clin Pharmacol 1996 Sep; 43(3): 333–7
Laaksonen R, Jokelainen K, Saudi-Shi T, et al. Decreases in serum ubiquinone concentrations do not result in reduced levels in muscle tissue during short-term simvastatin treatment in humans. Clin Pharmacol Ther 1995; 57(1): 62–6
Stemmermann GN, Chyou PH, Kaga A, et al. Serum cholesterol and mortality among Japanese American men in the Honolulu Heart Program. Arch Intern Med 1991; 151(5): 69–72
Iribarren C, Reed M, Chin R, et al. Low serum cholesterol and mortality: which is the cause and which is the effect? Circulation 1995; 92(9): 2396–403
Kannel WB. Range of serum cholesterol values in the population developing coronary artery disease. Am J Cardiol 1995; 76(9): 69C–77C
The Post Coronary Artery BypassGraft Trial Investigators. The effect of aggressive lowering of low density lipoprotein cholesterol levels and low dose anticoagulation on obstructive changes in saphenous vein coronary artery bypass grafts. N Engl J Med 1997; 336 (3): 153–62
McCormick LS, Black DM, Waters D, et al. Rationale, design and baseline characteristics of a trial comparing aggressive lipid lowering with atorvastatin versus revascularization treatments (AVERT). Am J Cardiol 1997; 80(9): 1130–3
Gruer PJ, Vega JM, Mercuri MF, Dobrinska MR, Tobert JA. Concomitant use of cytochrome P450 3A4 inhibitors and simvastatin. Am J Cardiol 1999; 84(7): 811–5
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Farmer, J.A., Torre-Amione, G. Comparative Tolerability of the HMG-CoA Reductase Inhibitors. Drug-Safety 23, 197–213 (2000). https://doi.org/10.2165/00002018-200023030-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-200023030-00003