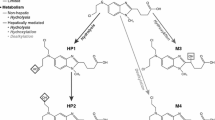
Abstract
This review discusses several issues in the clinical pharmacology of the antitumour agent ifosfamide and its metabolites. Ifosfamide is effective in a large number of malignant diseases. Its use, however, can be accompanied by haematological toxicity, neurotoxicity and nephrotoxicity. Since its development in the middle of the 1960s, most of the extensive metabolism of ifosfamide has been elucidated. Identification of specific isoenzymes responsible for ifosfamide metabolism may lead to an improved efficacy/toxicity ratio by modulation of the metabolic pathways.
Whether ifosfamide is specifically transported by erythrocytes and which activated ifosfamide metabolites play a key role in this transport is currently being debated. In most clinical pharmacokinetic studies, the phenomenon of autoinduction has been observed, but the mechanism is not completely understood. Assessment of the pharmacokinetics of ifosfamide and metabolites has long been impaired by the lack of reliable bioanalytical assays. The recent development of improved bioanalytical assays has changed this dramatically, allowing extensive pharmacokinetic assessment, identifying key issues such as population differences in pharmacokinetic parameters, differences in elimination dependent upon route and schedule of administration, implications of the chirality of the drug and interpatient pharmacokinetic variability. The mechanisms of action of cytotoxicity, neurotoxicity, urotoxicity and nephrotoxicity have been pivotal issues in the assessment of the pharmacodynamics of ifosfamide. Correlations between the new insights into ifosfamide metabolism, pharmacokinetics and pharmacodynamics will rationalise the further development of therapeutic drug monitoring and dose individualisation of ifosfamide treatment.





Similar content being viewed by others
References
Dechant KL, Brogden RN, Pilkington T, et al. Ifosfamide/ mesna: a review of its antineoplastic activity, phannacokinetic properties and therapeutic efficacy in cancer. Drugs 1991; 42: 428–67
Brock N, Hilgard P, Peukert M, et al. Basis and new development in the field of oxazaphosphorines. Cancer Invest 1988; 6:513–32
Seker H, Bertram B, Biirkle A, et al. Mechanistic aspects of the cytotoxic activity of glufosfamide, a new tumour therapeutic agent. Br J Cancer 2000; 82: 629–34
Calabresi P, Parks RE. Alkylating agents, antimetabolites, hormones and other antiproliferative agents. In: Goodman LS, Gilman A, editors. The pharmacological basis of therapeutics 5th ed. New York: Macmillan, 1975: 1254–5
Gilman A. The initial clinical trial of nitrogen mustard. Am J Surg 1963; 105: 574–8
Loehrer PJ. The history of ifosfamide. Semin Oncol 1992; 19: 2–7
Kamen BA, Frenkel E, Colvin OM. Ifosfamide: should the honeymoon be over? J Clin Oncol 1995; 13: 307–9
Brock N. Oxazaphosphorine cytostatics: past-present-future. Cancer Res 1989; 49: 1–7
Bryant BM, Jarman M, Ford HT, et al. Prevention of isophosphamide-induced urothelial toxicity with 2-mercaptoethane sulphonate sodium (mesnum) in patients with advanced carcinoma Lancet 1980; II: 657–9
Zalupski M, Baker LH. Ifosfamide [review]. J Natl Cancer Inst 1988; 80: 556–66
Sladek NE. Metabolism of oxazaphosphorine. Pharmacol Ther 1988; 37: 301–55
Highley MS, Momerency G, Van Cauwenberghe K, et al. Formation of chloroethylamine and 1,3-oxazolidine-2-one following ifosfamide administration in humans. Drug Metab Dispos 1995; 23: 433–7
Norpoth K. Studies on the metabolism of isophosphamide (NSC-109724) in man. Cancer Treat Rep 1976; 60: 437–43
Springate J, Zamlauski-Tucker MJ, Lu H, et al. Renal clearance of ifosfamide. Drug Metab Dispos 1997; 29: 1081–2
Yu L, Waxman DJ. Role of CYP in oxazaphosphorine metabolism: deactivation via N-dechloroethylation and activation via 4-hydroxylation catalyzed by distinct subsets of rat liver cytochromes P450. Drug Metab Dispos 1996; 24: 1254–62
Weber GF, Waxman DJ. Activation of the anticancer drug ifosphamide by rat liver microsomal P450 enzymes. Biochem Pharmacol 1993; 45: 1685–94
Brain EGC, Gustafsson K, Drewes P, et al. Modulation of P450-dependent ifosfamide pharmacokinetics: a better understanding of drug activation in vivo. Br J Cancer 1998; 77: 1768–76
Crommentuyn KML, Schellens JHM, van den Berg JD, et al. In-vitro metabolism of anticancer drugs, methods and applications: paclitaxel, docetaxel, tamoxifen and ifosfamide. Cancer Treat Rev 1998; 24: 345–66
Wainer IW, Granvil CP, Wang T, et al. Efficacy and toxicity of ifosfamide stereoisomers in an in vivo rat mammary carcinoma model. Cancer Res 1994; 54: 4393–7
Chang TKH, Weber GF, Crepi CL, et al. Differential activation of cyclophosphamide and ifosfamide by cytochrome P-450 2B and 3A in human liver microsomes. Cancer Res 1993; 53: 5629–37
Walker D, Flinois JP, Monkman SC, et al. Identification of the major human hepatic CYP involved in the activation and N-dechloroethylation of ifosfamide. Biochem Pharmacol 1994; 47: 1157–63
Roy P, Yu LJ, Crespi CL, et al. Development of a substrate-activity based approach to identify the major human liver P-450 catalysts of cyclophosphamide and ifosfamide activation based on cDNA-expressed activities and liver microsomal P-450 profiles. Drug Metab Dispos 1999; 27: 655–66
Granvil CP, Madan A, Sharkawi M, et al. Role of CYP2B6 and CYP3A4 in the in vitro N-dechloroethylation of (R)- and (S)ifosfamide in human liver microsomes. Drug Metab Dispos 1999; 27: 533–41
Roy P, Tretyakov O, Wright J, et al. Stereoselective metabolism of ifosfamide by human P-450s 3A4 and 2B6. Favorable metabolic properties of R-enantiomer. Drug Metab Dispos 1999 27: 1309–18
Huang Z, Roy P, Waxman DJ. Role of human liver microsomal CYP3A4 and CYP2B6 in catalyzing N-dechloroethylation of cyclophosphamide and ifosfamide. Biochem Pharmacol 2000; 59: 961–72
Chen L, Waxman DJ, Chen D, et al. Sensitization of human breast cancer cells to cyclophosphamide and ifosfamide by transfer of a liver cytochrome P450 gene. Cancer Res 1996; 56: 1331–40
Lokiec F, Santoni J, Weill S, et al. Phenobarbital administration does not affect high-dose ifosfamide pharmacokinetics in humans. Anticancer Drugs 1996; 7: 893–6
Chen L, Waxman DJ. Intratumoral activation and enhanced chemotherapeutic effect of oxazaphosphorines following cytochrome P-450 gene transfer: development of a combined chemotherapy/cancer gene therapy strategy. Cancer Res 1995;55:581–9
Waxman DJ, Chen L, Hecht JED, et al. Cytochrome P450-based cancer gene therapy. Recent advances and future prospects. Drug Metab Rev 1999; 31: 503–22
Chang TK, Yu L, Maurel P, Waxman DJ. Enhanced cyclophosphamide and ifosfamide activation in primary human hepatocyte cultures: response to cytochrome P-450 inducers and autoinduction by oxazaphosphorines. Cancer Res 1997; 57: 1946–54
Ducharme MP, Bernstein ML, Granvil CP, et al. Phenytoin-in-duced alteration in the N-dechloroethylation of ifosfamide stereoisomers. Cancer Chemother Pharmacol 1997;40: 531–3
Kivistö KT, Kroemer HK, Eichelbaum M. The role of human cytochrome P450 enzymes in the metabolism of anticancer agents: implications for drug interactions. Br J Clin Pharmacol 1995; 50: 523–30
Bertz RJ, Granneman GR. Use of in vitro and in vivo data to estimate the likelihood of metabolic pharmacokinetic interactions. Clin Pharmacokinet 1997; 32: 210–58
Lind MJ, Margison JM, Cerny T, et al. Comparative pharmacokinetics and alkylating activity of fractionated intravenous and oral ifosfamide in patients with brochogenic carcinoma. Cancer Res 1989; 49: 753–7
von Bahr C, Steiner E, Koike Y, et al. Time course of enzyme induction in humans: effect of pentobarbital on nortriptyline metabolism. Clin Pharmacol Ther 1998; 64: 18–26
Pitlick WH, Levy RH, Troupin AS, et al. Pharmacokinetic model to describe self-induced decreases in steady-state concentrations of carbamazepine. J Pharm Sci 1975; 65: 462–3
Lind MJ, Cerny T, Margison J, et al. Decreased half-life of ifosfamide (I) after daily oral administration of ifosfamide [abstract no. 10]. First Annual Meeting of the Association of Cancer Physicians; 1986 Mar 24–26; Bristol, 15
Watkins PB, Wrighton SA, Schuetz EG, et al. Macrolide antibiotics inhibit the degradation of the glucocorticoid-responsive cytochrome P450P in rat hepatocytes in vivo and in primary monolayer culture. J Biol Chem 1986; 261: 6264–71
Gibson GG, Tamburini P. Cytochrome P-450 spin state, inorganic biochemistry of haem iron ligation and functional significance. Xenobiotica 1984; 14: 27–47
Lewis LD, Fitzgerald DL, Harper PG, et al. Fractionated ifosfamide therapy produces a time-dependent increase in ifosfamide metabolism. Br J Clin Pharmacol 1990; 30:725–32
Highley MS, Harper PG, Slee PHTJ, et al. Preferential location of circulating activated cyclophosphamide within the erythrocyte. Int J Cancer 1996; 65: 711–2
Highley MS, De Bruijn EA. Erythrocytes and the transport of drugs and endogenous compounds. Pharm Res 1996; 13:186–95
Highley MS, Schrijvers D, Van Oosterom AT, et al. Activated oxazaphosphorines are transported predominantly by erythrocytes. Ann Oncol 1997; 8: 1139–44
Momerency G, Van Cauwenberghe K, Highley MS, et al Partitioning of ifosfamide and its metabolites between red blood cells and plasma. J Pharm Sci 1996; 85: 262–5
Domeyer BE, Sladek NE. Kinetics of cyclophosphamide biotransformation in vivo. Cancer Res 1980; 40: 174–80
Wagner T, Peter G, Voelcker G, et al. Characterisation and quantitative estimation of activated cyclophosphamide in blood and urine. Cancer Res 1977; 37: 2592–6
Struck RF, Dykes DJ, Corbett TH, et al. Isophosphoramide mustard, a metabolite of ifosfamide with activity against murine tumours comparable to cyclophosphamide. Br J Cancer 1983; 47: 15–26
Tjaden UR, De Bruijn EA. Chromatographic analysis of anticancer drugs. J Chromatogr 1990; 531: 235–94
Kaijser GP, Korst A, Beijnen JH, et al. The analysis of ifosfamide and its metabolites [review]. Anticancer Res 1993; 13: 1311–24
Lind MJ, Roberts HL, Thatcher N, et al. The effect of route of administration and fractionation of dose on the metabolism of ifosfamide. Cancer Chemother Pharmacol 1990; 26: 105–11
Boddy AV, Idle JR. Combined thin-layer chromatography-photography-densitometry for the quantification of ifosfamide and its principal metabolites in urine, cerebrospinal fluid and plasma. J Chromatogr 1992; 575: 137–42
Payne GS, Pinkerton CR, Leach MO. Magnetic resonance spectroscopy studies of ifosfamide in vivo [abstract no. 487]. Clin Cancer Res 1999; 5 Suppl.: 3827
Gillard F, Malet-Martino MC, de Forni M, et al. Determination of the urinary excretion of ifosfamide and its phosphorated metabolites by phosphorus-31 nuclear magnetic resonance spectroscopy. Cancer Chemother Pharmacol 1993; 31: 387–94
Martino R, Crasnier F, Chouini-Lalanne N, et al. A new approach to the study of ifosfamide metabolism by the analysis of human body fluids with 31P nuclear magnetic resonance spectroscopy. J Pharmacol Exp Ther 1992; 260: 1133–44
Burton LC, James CA. Rapid method for the determination of ifosfamide and cyclophosphamide in plasma by high-performance liquid chromatography with solid-phase extraction. J Chromatogr 1988; 431: 450–4
Margison JM, Wilkinson PM, Cerny T, et al. A simple quantitative HPLC assay for ifosfamide in biological fluids. Biomed Chromatogr 1986; 1: 101–3
Zolezzi C, Ferrari S, Bacci G, et al. Determination of ifosfamide by HPLC using on-line sample pretreatment. J Chemother 1999; 11: 69–73
Goren MP. Determination of urinary 2- and 3-dechloroethylated metabolites of ifosfamide by high-performance liquid chromatography. J Chromatogr 1991; 570: 351–9
Corlett SA, Chrystyn H. Enantiomeric separation of R- and Sifosfamide and their determination in serum from clinical subjects. J Chromatogr 1994; 654: 152–8
Masurel D, Wainer IW. Analytical and preparative high-performance liquid Chromatographic separation of the enantiomers of ifosfamide, cyclophosphamide and trofosfamide and their determination in plasma. J Chromatogr 1989; 490: 133–43
Kaijser GP, Beijnen JH, Jeunink EL, et al. Determination of chloroacetaldehyde, a metabolite of oxazaphosphorine cytostatic drugs, in plasma. J Chromatogr 1993; 614: 253–9
Kaijser GP, Ter Riet PGJH, de Kraker J, et al. Determination of 4-hydroxyifosfamide in biological matrices by high-performance liquid chromatography. J Pharmaceut Biomed Anal 1997; 15: 773–81
Kurowski V, Wagner T. Comparative pharmacokinetics of ifosfamide, 4-hydroxyifosfamide, chloroacetaldehyde, and 2- and 3-dechloroemylifosfamide in patients on fractionated intravenous ifosfamide therapy. Cancer Chemother Pharmacol 1993; 33: 36–42
Kerbusch T, Huitema ADR, Kettenes-van den Bosch JJ, et al. High-performance liquid Chromatographic determination of stabilized 4-hydroxyifosfamide in human plasma and erythrocytes. J Chromatogr B 1998; 716: 275–84
Kaijser GP, Beijnen JH, Rozendom E, et al. Analysis of ifosforamide mustard, the active metabolite of ifosfamide, in plasma. J Chromatogr B 1996; 686: 249–55
Kerbusch T, Huitema ADR, Jeuken M-JJ, et al. Simultaneous determination of ifosfamide and its metabolite ifosforamide mustard in human plasma by high-performance liquid chromatography. J Liq Chromatogr Relat Tech 2000; 23: 2991–3010
Mehta AC, Calvert RT. Determination of ifosfamide in plasma by gas chromatography with nitrogen detector. J Chromatogr 1987;421:377–80
Talha MRZ, Rogers HJ. Rapid gas Chromatographie determination of ifosfamide in biological fluids. J Chromatogr 1984; 311: 194–8
Kaijser GP, Beijnen JH, Bult A, et al. The determination of 2-and 3-dechloroethylifosfamide in plasma and urine. J Chromatogr 1992; 583: 175–82
Kerbusch T, Jeuken MJJ, Derraz J, et al. Determination of ifosfamide and its 2- and 3-dechloroethylated metabolites using gas chromatography, comparing nitrogen-phosphorous selective and ion-trap mass spectrometry detection. Ther Drug Monit 2000; 22: 613–20
Kaijser GP, Beijnen JH, Bult A, et al. Chromatographie analysis of the enantiomers of ifosfamide and some of its metabolites in plasma and urine. J Chromatogr B 1997; 690: 131–8
Courmel B, Granvil CP, Denis SL, et al. Determination of 4-hydroxyifosfamide concomitantly with ifosfamide and its dechloroethylated metabolites using gas chromatography and a nitrogen phosphorus-selective detector. J Chromatogr B 1999; 732:3–15
Bryant BM, Jarman M, Baker MH, et al. Quantification by gas chromatography of N, N′-di-(2-chloroethyl)-phosphordiamidic acid in the plasma of patients receiving isophosphamide. Cancer Res 1980; 40: 4734–8
Allen LM, Creaven PJ. Gas Chromatographie method for the determination of plasma isophosphamide (NSC-109724). Cancer Chemother Rep 1972; 56: 721–3
Manz I, Dietrich I, Przybylski M, et al. Identification and quantification of metabolite conjugates of activated cyclophosph-amide and ifosfamide with mesna in urine by ion-pair extraction and fast atom bombardment mass spectrometry. Biomed Mass Spectrom 1985; 12: 545–53
Lambrechts H, Gheuens EOO, Van Cauwenberghe KA, et al. Determination of ifosfamide by gas chromatography-mass spectrometry. Anal Chim Acta 1991; 247: 229–33
Momerency G, Van Cauwenberghe K, De Bruijn EA, et al. Determination of iphosphamide and seven metabolites in blood plasma, as stable trifluoroacetyl derivatives by electron capture chemical ionization GC-MS. J High Res Chromatogr 1994; 17: 655–61
Young CL, Frank H, Stewart CR, et al. The determination of (−)-(S)- and (+)-(R)ifosfamide in plasma using enantioselective gas chromatography: a validated assay for pharmacokinetic and clinical studies. Chirality 1989; 1: 235–8
Granville CP, Gehrcke B, König WA, et al. Determination of the enantiomers of ifosfamide and its 2- and 3-N-dechloroethylated metabolites in plasma and urine using enantioselective gas chromatography with mass spectrometric detection. J Chromatogr 1993; 622: 21–31
Wang JJ-H, Chan KK. Analysis of ifosfamide, 4-hydroxyifosfamide, N2-dechloroethylifosfamide, N3-dechloroethylifosfamide and iphosphoramide mustard in plasma by gas chromatography-mass spectrometry. J Chromatogr B 1995; 674: 205–17
De Bruijn EA, Tjaden UR, Leclercq PA. Capillary chromatography in cancer research. Eur Chromatogr News 1988; 2: 16–9
Kaijser GP, Beijnen JH, Bult A, et al. Capillary gas Chromatographie determination of ifosfamide in microvolumes of urine and plasma. J Chromatogr 1991; 571: 121–31
Kaijser GP, Beijnen JH, Bult A, et al. Ifosfamide metabolism and pharmacokinetics [review]. Anticancer Res 1994; 14: 517–32
Nelson RL, Allen LM, Creaven PJ. Pharmacokinetics of divideddose ifosfamide. Clin Pharmacol Ther 1976; 19: 365–70
Allen LM, Creaven PJ. Pharmacokinetics of ifosfamide. Clin Pharmacol Ther 1975; 17: 492–8
McNiel NO, Morgan LR. The bioavailability of oral and intravenous ifosfamide in the treatment of bronchogenic carcinoma. Int J Clin Pharmacol Ther Toxicol 1981; 19: 490–3
Piazza E, Cattaneo MT, Varini M. Pharmacokinetic studies in lung cancer patients. Cancer 1984; 54: 1187–92
Cerny T, Margison JM, Thatcher N, et al. Bioavailability of ifosfamide in patients with bronchial carcinoma. Cancer Chemother Pharmacol 1986; 18: 261–4
Cerny T, Leyvraz S, von Briel T, et al. Saturable metabolism of continuous high-dose ifosfamide with mesna and GM-CSF: a pharmacokinetic study in advanced sarcoma patients. Ann Oncol 1999; 10: 1087–94
Pearcey R, Calvert R, Mehta A. Disposition of ifosfamide in patients receiving ifosfamide infusion therapy for the treatment of cervical carcinoma. Cancer Chemother Pharmacol 1988; 22: 353–5
Lewis LD, Mohan P, Thatcher N, et al. Time dependent ifosfamide half-life and metabolism when given as a fractionated regime over 5 days to treat thoracic neoplasms [abstract no. 10.24]. 5th NCI-EORTC Symposium on New Drugs in Cancer Therapy; 1986 Oct 22–24; Amsterdam
Lewis LD, Fitzgerald DL, Mohan P, et al. The pharmacokinetics of ifosfamide given as short and long intravenous infusions in cancer patients. Br J Clin Pharmacol 1991; 31: 77–82
Lewis LD. A study of 5 day fractionated ifosfamide pharmacokinetics in consecutive treatment cycles. Br J Clin Pharmacol 1996; 42: 179–86
Wagner T, Drings P. Pharmacokinetics and bioavailability of oral ifosfamide. Contr Oncol 1987; 26: 53–9
Benvenuto JA, Ayele W, Legha SS, et al. Clinical pharmacokinetics of ifosfamide in combination with N-acetylcysteine. Anticancer Drugs 1992; 3: 19–23
Kurowski V, Wagner T. Urinary excretion of ifosfamide, 4-hydroxyifosfamide, chloroacetaldehyde, and 3- and 2-dechloroethylifosfamide, mesna, and dimesna in patients on fractionated intravenous ifosfamide and concomitant mesna therapy. Cancer Chemother Pharmacol 1997; 39: 431–9
Wiedermann GJ, d’Oleire F, Knop E, et al. Ifosfamide and carboplatin combined with 41.8°C whole-body hyperthermia in patients with refractory sarcoma and malignant teratoma. Cancer Res 1994; 54: 5346–50
Hartley JM, Hansen L, Harland SJ, et al. Metabolism of ifosfamide during a 3 day infusion. Br J Cancer 1994; 69: 931–6
Corlett SA, Parker D, Chrystyn H. Pharmacokinetics of ifosfamide and its enantiomers following a single 1h intravenous infusion of the racemate in patients with small cell lung carcinoma. Br J Clin Pharmacol 1995; 39: 452–5
Boddy AV, Proctor M, Simmonds D, et al. Pharmacokinetics, metabolism and clinical effects of ifosfamide in breast cancer patients. Eur J Cancer 1995; 31: 69–76
Kaijser GP, Keizer HJ, Beijnen JH, et al. Pharmacokinetics of ifosfamide, 2- and 3-dechloroethylifosfamide in plasma and urine of cancer patients treated with a 10-day continuous infusion of ifosfamide. Anticancer Res 1996; 16: 3247–58
Kaijser GP, Beijnen JH, de Kraker J, et al. Pharmacokinetics of enantiomers of ifosfamide, 2- and 3-dechloroethylifosfamide in plasma, urine and cerebrospinal fluid. J Drug Dev Clin Pract 1996; 7:319–26
Granvil CP, Ducharme J, Leyland-Jones B, et al. Stereoselective pharmacokinetics of ifosfamide and its 2- and 3-dechloroethylated metabolites in female cancer patients. Cancer Chemother Pharmacol 1996; 37: 451–6
Wainer TW, Ducharme J, Granvil CR The N-dechloroethylation of ifosfamide: using stereochemistry to obtain an accurate picture of a clinically relevant metabolic pathway. Cancer Chemother Pharmacol 1996; 37: 332–6
Comandone A, Leone L, Oliva C, et al. Pharmacokinetics of ifosfamide administered according to three different schedules in metastatic soft tissue and bone sarcomas. J Chemother 1998; 10:385–93
Singer JM, Hartley JM, Brennan C, et al. The pharmacokinetics and metabolism of ifosfamide during bolus and infusional administration: a randomized cross-over study. Br J Cancer 1998; 77:978–84
Lind MJ, Margison JM, Cerny TL, et al. The effect of age on the pharmacokinetics of ifosfamide. Br J Clin Pharmacol 1990; 30: 140–3
Lind MJ, Margison JM, Cerny T, et al. Prolongation of ifosfamide elimination half-life in obese patients due to altered drug distribution. Cancer Chemother Pharmacol 1989; 25: 139–42
Passe P, Delepine N, Arnaud P, et al. Pharmacokinetic modelling of ifosfamide administered by continuous infusion on 5 days at the dose of 6 g/m2. Anticancer Res 1999; 19: 837–42
Boddy AV, Yule SM, Wyllie R, et al. Pharmacokinetics and metabolism of ifosfamide administered as a continuous infusion in children Cancer Res 1993; 53: 3758–64
Boddy AV, Yule SM, Wyllie R, et al. Comparison of continuous infusion and bolus administration of ifosfamide in children. Eur J Cancer 1995; 31A: 785–90
Boddy AV, Yule SM, Wyllie R, et al. Intrasubject variation in children of ifosfamide pharmacokinetics and metabolism during repeated administration. Cancer Chemother Pharmacol 1996; 38: 147–54
Boddy AV, Cole M, Pearson ADJ, et al. The pharmacokinetics of the auto-induction of ifosfamide metabolism during continuous infusion. Cancer Chemother Pharmacol 1995; 36: 53–60
Silies H, Blaschke G, Hohenlochter B, et al. Excretion kinetics of ifosfamide side-chain metabolites in children on continuous and short-term infusion. Int J Clin Pharmacol Ther 1998; 36: 246–52
Boos J, Welslau U, Ritter J, et al. Urinary excretion of the enantiomers of ifosfamide and its inactive metabolites in children. Cancer Chemother Pharmacol 1991; 28: 455–60
Prasad VK, Corlett SA, Abaasi K, et al. Ifosfamide enantiomers: pharmacokinetics in children. Cancer Chemother Pharmacol 1994; 34: 447–9
Ducharme MP, Bernstein ML, Granvil CP, et al. Population PK of IFF and its DCE metabolites in children using an enantiospecific PK model [abstract]. Clin Pharmacol Ther 1997; 61:OIII–B–3
Yule SM, Price L, Pearson AD, Boddy AV. Cyclophosphamide and ifosfamide metabolites in the cerebrospinal fluid of children. Clin Cancer Res 1997; 3: 1985–92
Kaijser GP, de Kraker J, Bult A, et al. Pharmacokinetics of ifosfamide and some metabolites in children. Anticancer Res 1998; 18: 1941–9
Kurowski V, Cerny T, Kupfer A, et al. Metabolism and pharmacokinetics of oral and intravenous ifosfamide. J Cancer Res Clin Oncol 1991; 117 Suppl. IV: 148–53
Cerny T, Küpfer A, Zeugin T, et al. Bioavailability of subcutaneous ifosfamide and feasibility of continuous outpatient application in cancer patients. Ann Oncol 1990; 1: 365–8
Lokich J, Anderson N, Bern M, et al. Ifosfamide continuous infusion without mesna. Cancer 1991; 67: 883–5
Frei III E, Cucchi CA, Rosowsky A, et al. Alkylating agent resistance. In vitro studies with human cell lines. Proc Natl Acad Sci USA 1985; 82: 2158–62
Verweij J. High-dose ifosfamide forsofttissue sarcomas: set the scene, or senescence? [editorial]. Ann Oncol 1998; 9: 807–9
Crom WR. Effect of chirality on pharmacokinetics and pharmacodynamics. Am J Hosp Pharm 1992; 49 Suppl. 1: S9–S14
Wainer IW, Cranvil CP. Stereoselective separations of chiral anticancer drugs and their application to pharmacodynamic and pharmacokinetic studies. Ther Drug Monit 1993; 15: 570–5
Breimer DD. Interindividual variations in drug disposition: clinical implications and methods of investigation. Clin Pharmacokinet 1983; 8: 371–7
Kivistö KT, Fritz P, Linder A, et al. Immunohistochemical localization of cytochrome P450 3A in human pulmonary carcinomas and normal bronchial tissue. Histochem J 1995; 71: 36–43
Carlsson L, Goren MP, Bush JC, et al. Toxicity, pharmacokinetics, and in vitro hemodialysis clearance of ifosfamide and metabolites in an anephric pediatric patient with Wilms’ tumor. Cancer Chemother Pharmacol 1998; 41: 140–6
Wagner T. Ifosfamide clinical pharmacokinetics. Clin Pharmacokinet 1994; 26: 439–56
Nowrousian MR, Burkert H, Hendrich K, et al. Ifosfamide in cancer therapy. Jena: ASTAMedica AG, 1993: 79–81
Gourmel B, Lefebre P, Extra JM, et al. Etude pharmacoinetique plasmique et de l’elimination de l’ifosfamide en situation de drainage biliarie complet [abstract]. Therapie 1988; 43: 519
Hassan M, Svensson USH, Ljungman P, et al. A mechanismbased pharmacokinetic-enzyme model for cyclophosphamideautoinduction in breast cancer patients. Br J Clin Pharmacol 1999; 48: 669–77
Kerbusch T, Huitema ADR, Ouwerkerk J, et al. Evaluation of the autoinduction of ifosfamide metabolism by a population pharmacokinetic approach using NONMEM. Br J Clin Pharmacol 2000; 49: 555–61
Dong Q, Barsky D, Colvin ME, et al. A structural basis for a phosphoramide mustard-induced DNA interstrand cross-link at 5′-d(GAC). Proc Natl Acad Sci USA 1995; 92: 12170–4
Brock N. The oxazaphosphorines. Cancer Treat Rev 1983; 10 Suppl. A: 3–15
Hartley JM, Spanswick VJ, Gander M, et al. Measurement of DNA cross-linking in patients on ifosfamide therapy using single cell gel electrophoresis (Comet) assay. Clin Cancer Res 1999; 5:507–12
Colvin OM. Drug resistance in the treatment of sarcomas. Semin Oncol 1997; 24: 580–91
Issels RD, Meier TH, Müller E, et al. Ifosfamide induced stress response in human lymphocytes. Mol Aspects Med 1993; 14: 281–6
Lind MJ, McGown AT, Hadfield JA, et al. The effect of ifosfamide and its metabolites on intracellular gluthatione levels in vitro and in vivo. Biochem Pharmacol 1989; 38: 1835–40
Wagner T. Mode of action of ifosfamide: new aspects. Onkologie 1998; 21 Suppl. 2: 1–4
Brüggemann SK, Kisro J, Wagner T. Ifosfamide cytotoxicity on human tumor and renal cells: role of chloroacetaldehyde in comparison to 4-hydroxyifosfamide. Cancer Res 1997; 57: 2676–80
Crook TR, Souhami RL, McLean AEM. Cytotoxicity, DNA cross-linking and single strand breaks induced by activated cyclophosphamide and acrolein in human leukemia cells. Cancer Res 1986; 46: 5029–34
Colvin M. The comparative pharmacology of cyclophosphamide and ifosfamide. Semin Oncol 1982; 9 Suppl. 1: 1–7
van der Wall E, Beijnen JH, Rodenhuis S. High-dose chemotherapy regimens for solid tumors. Cancer Treat Rev 1995; 21: 105–32
Boal JH, Williamson M, Boyd VL, et al. 31P NMR studies of the kinetics of bisalkylation by isophosphoramide mustard: comparison with phosphoramide mustard. J Med Chem 1989; 32: 1768–74
Brugger W, Bross K, Frish J, et al. The mobilization of peripheral blood progenitor cells by sequential administration of interleukin-3 and granulocyte-macrophage colony-stimulating factor following polychemotherapy with etoposide, ifosfamide, and cisplatin. Blood 1992; 79: 1193–200
Brade WP, Hendrich K, Varini M. Ifosfamide — pharmacology, safety and therapeutic potential. Cancer Treat Rev 1985; 12: 1–47
Morgan LR, Harrison EF, Hawke JE, et al. Toxicity of single-vs fractionated-dose ifosfamide in non-small cell lung cancer: a multi-center study. Semin Oncol 1982; 9 Suppl. 1: 66–70
Anderson NR, Tandon DS. Ifosfamide extrapyramidal neurotoxicity. Cancer 1991; 68: 72–5
Pallotta MG, Velazco A, Sadler A, et al. Ifosfamide extrapyramidal neurotoxicity [correspondence]. Cancer 1992; 70: 2743–5
Cerny T, Küpfer A. The enigma of ifosfamide encephalopathy [editorial]. Ann Oncol 1992; 3: 679–81
Danesh MM, De Giorgio CM, Beydoun SR, et al. Ifosfamide encephalopathy. Clin Toxicol 1989; 27: 293–8
Simonium NA, Gilliam FG, Chiappa KH. Ifosfamide causes a diazepam-sensitive encephalopathy. Neurology 1993; 43: 2700–2
DiMaggio JR, Brown R, Baile WF, et al. Hallucinations and ifosfamide-induced neurotoxicity. Cancer 1994; 73: 1509–14
Salloum E, Flamant F, Ghosn M, et al. Irreversible encephalopathy with ifosfamide-mesna. J Clin Oncol 1987; 5: 1303–4
Verdeguer A, Castel V, Esquembre C, et al. Fatal encephalopathy with ifosfamide-mesna. Pediatr Hematol Oncol 1989; 6: 383–5
Pratt CB, Green AA, Horowitz ME, et al. Central nervous system toxicity following treatment of pediatric patients with ifosfamide/mesna. J Clin Oncol 1986; 4: 1253–61
Miller LJ, Eaton VE. Ifosfamide-induced neurotoxicity: a case report and review of the literature. Ann Pharmacother 1992; 26: 183–7
Watkin SW, Husband DJ, Green JA, et al. Ifosfamide encephalopathy: a reappraisal. Eur J Cancer Clin Oncol 1989; 25: 1303–10
Meanwell CA, Blake AE, Kelly KA, et al. Prediction of ifosfamide/mesna associated encephalopathy. Eur J Cancer Clin Oncol 1986; 22:815–9
Alonso JL, Nieto Y, López JA, et al. Ifosfamide encephalopathy and methylene-blue: a case report. Ann Oncol 1996; 7: 643–4
Goren MP, Wright RK, Pratt CB, et al. Potentiation of ifosfamide neurotoxicity, hematotoxicity, and tubular nephrotoxicity by prior cis-diamminedichloroplatinum(II) therapy. Cancer Res 1987; 47: 1457–60
Cantwell BMJ, Harris AL. Ifosfamide/mesna and encephalopathy [letter]. Lancet 1985; I: 752
Lewis LD, Meanwell CA. Ifosfamide pharmacokinetics and neurotoxicity. Lancet 1990; 335: 175–6
Ninane J, Baurain R, de Kraker J, et al. Alkylating activity in serum, urine, and CSF following high-dose ifosfamide in children. Cancer Chemother Pharmacol 1989; 24 Suppl.: S2–6
Wagner T. Alkylating activity in serum, urine, and CSF following high-dose ifosfamide in children: a comment. Cancer Chemother Pharmacol 1989; 24 Suppl.: S7
Goren MP, Wright RK, Pratt CB, et al. Dechloroethylation of ifosfamide and neurotoxicity. Lancet 1986; II: 1219–20
Wainer IW, Ducharme J, Granvil CP, et al. Ifosfamide stereoselective dechloroethylation and neurotoxicity. Lancet 1994; 343: 982–3
Cerny T, Castiglione M, Brunner K, et al. Ifosfamide by continuous infusion to prevent encephalopathy [letter]. Lancet 1990; 335: 175
Kupfer A, Aeschlimann C, Cerny T. Methylene blue and the neurotoxic mechanisms of ifosfamide encephalopathy. Eur J Clin Pharmacol 1996; 50: 249–52
Sood C, O’Brien PJ. 2-Chloroacetaldehyde-induced cerebral gluthatione depletion and neurotoxicity. Br J Cancer 1994; 74 Suppl. XXVII: S287–93
Küpfer A, Aeschlimann C, Wermuth B, et al. Prophylaxis and reversal of ifosfamide encephalopathy with methylene-blue. Lancet 1994; 343: 763–4
Visarius TM, Stucki JW, Lauterberg BH. Inhibition and stimulation of long-chain fatty acid oxidation by chloroacetaldehyde and methylene blue in rats. J Pharmacol Exp Ther 1999; 289: 820–4
Aeschlimann C, Cerny T, Küpfer A. Inhibition of (mono)amine oxidase activity and prevention of ifosfamide encephalopathy by methylene blue. Drug Metab Dispos 1996; 24: 1336–9
Serve H, Hilgenfeld, Maurer J, et al. Use of methylene blue and bicarbonate in ifosfamide-related CNS toxicity: case report [abstract no. 1248]. Eur J Cancer 1995; 31A: S260–1
Zulian GB, Tullen E, Maton B. Methylene blue for ifosfamideassociated encephalopathy. N Engl J Med 1995; 332: 1239–40
Aeschlimann C, Küpfer A, Schefer H, et al. Comparative pharmacokinetics of oral and intravenous ifosfamide/mesna/methylene blue therapy. Drug Metab Dispos 1998; 26: 883–90
Shaw IC, Graham MI. Mesna: a short review. Cancer Treat Rev 1987; 14: 67–86
Goren MR Oral mesna: a review. Semin Oncol 1992; 19: 65–72
Schoenike SE, Dana WJ. Ifosfamide and mesna. Clin Pharm 1990; 9: 179–91
Hilgard P, Pohl J. Oxazaphosphorine toxicity reduction of mesna. Cancer Treat Rev 1990; 17: 217–20
Hoekman K, van der Vijgh WJF, Vermorken JB. Clinical and preclinical modulation of chemotherapy-induced toxicity in patients with cancer. Drugs 1999; 57(2): 133–55
Siu LL, Moore MJ. Use of mesna to prevent ifosfamide-induced urotoxicity. Support Care Cancer 1998; 6: 144–54
Goren MP, McKenna LM, Goodman TL. Combined intravenous and oral mesna in outpatients treated with ifosfamide. Cancer Chemother Pharmacol 1997; 40: 371–5
Wolff JEA, Egeler RM, Anderson R, et al. Mesna inactivates platinum agents in vitro. Anticancer Res 1998; 18: 4077–82
Loebstein R, Koren G. Ifosfamide-induced nephrotoxicity in children: critical review of predictive risk factors [electronic version]. Pediatrics 1998; 101: E8
Skinner R, Sharkey IM, Pearson ADJ, et al. Ifosfamide, mesna, and nephrotoxicity in children. J Clin Oncol 1993; 11: 173–90
Rossi R. Nephrotoxicity of ifosfamide — moving towards understanding the molecular mechanism. Nephrol Dial Transplant 1997; 12: 1091–2
Tokuc G, Yalçiner A, Kebudi R, et al. Renal dysfunctions secondary to ifosfamide treatment in children. J Exp Clin Cancer Res 1997; 16: 227–30
Zamlauski-Tucker MJ, Morris ME, Springate JE. Ifosfamide metabolite chloroacetaldehyde causes Fanconi Syndrome in perfused rat kidney. Toxicol Appl Pharmacol 1994; 129: 170–5
Woodland C, Ito S, Klein J, et al. First evidence of renal biotransformation of ifosfamide to nephrotoxic metabolites [abstract]. Clin Pharmacol Ther 1998; 63: 236
Schlenzig JS, Charpentier C, Rabier D, et al. L-Carnitine: a way to decrease cellular toxicity of ifosfamide? Eur J Pediatr 1995; 154: 686–7
Marthaler NP, Visarius T, Küpfer A, et al. Increased urinary losses of carnitine during ifosfamide chemotherapy. Cancer Chemother Pharmacol 1999; 44: 170–2
Boddy AV, English M, Pearson AD, et al. Ifosfamide nephrotoxicity: limited influence of metabolism and mode of administration during repeated therapy in paediatrics. Eur J Cancer 1996; 32: 1179–84
Lauterburg B, Nguyen T, Hartmann B, et al. Depletion of total cysteine, gluthatione, and homocysteine in plasma by ifosfamide/mesna therapy. Cancer Chemother Pharmacol 1994; 35: 132–6
Stofer-Vogel B, Cerny T, Kupfer A, et al. Depletion of circulating cyst(e)ine by oral and intravenous mesna. Br J Cancer 1993; 68: 590–3
Holoye PY, Glisson BS, Lee JD, et al. Ifosfamide with mesna uroprotection in the management of lung cancer. Am J Clin Oncol 1990; 13: 148–55
Green JA, Watkin SW, Hammond P, et al. The efficacy and safety of GR38023F in the prophylaxis of ifosfamide-induced nausea and vomiting. Cancer Chemother Pharmacol 1989; 24: 137–9
Lakhi SA, Bais EM, Rutgers AJT, et al. Hypersensitivity after ifosfamide administration. J Oncol Pharm Practice 1996; 2: 113–6
Kandylis K, Vassilomanolakis M, Tsoussis S, et al. Ifosfamide cardiotoxicity in humans. Cancer Chemother Pharmacol 1989; 24: 395–6
Baker WJ, Fistel SJ, Jones RV, et al. Interstitial Pneumonitis associated with ifosfamide therapy. Cancer 1990; 65: 2217–21
Lieberman R, Crowell JA, Hawk ET, et al. Development of new cancer chemoprevention agents: role of pharmacokinetic/pharmacodynamic and intermediate endpoint biomarker monitoring. Clin Chem 1998; 44: 420–7
Ayash LJ, Wright JE, Tretyakov O, et al. Cyclophosphamide pharmacokinetics: correlation with cardiotoxicity and tumor response. J Clin Oncol 1992; 10: 995–1000
Petros WP, Broadwater G, Berry D, et al. Correlation of highdose cyclophosphamide, cisplatin, and carmustine pharmacokinetics to response and toxicity in patients with primary breast cancer [abstract no. 756]. Proc Am Soc Clin Oncol 1997; 16: 2169
Lind MJ, Ardiet C. Pharmacokinetics of alkylating agents. Cancer Surv 1993; 17: 157–88
Wright JE, Elias A, Tretyakov O, et al. High-dose ifosfamide, carboplatin, and etoposide pharmacokinetics: correlation of plasma drug levels with renal toxicity. Cancer Chemother Pharmacol 1995; 36: 345–51
Völcker G, Wagner T, Wientzek C, et al. Pharmacokinetics of ‘activated’ cyclophosphamide and therapeutic efficacies. Cancer 1984; 54: 1179–86
Kaijser GP, Beijnen JH. Oxazaphosphorines: cyclophosphamide and ifosfamide. In: Grochow LB, Ames MM, editors. A clinician’s guide to chemotherapy pharmacokinetics and pharmacodynamics. Baltimore: Williams & Wilkins, 1998: 229–58
Poikonen P, Saarto T, Lundin J, et al. Leukocyte nadir as a marker for chemotherapy efficacy in node-positive breast cancer treated with adjuvant CMF. Br J Cancer 1999; 80(11): 1763–6
Kvinnsland S. The leukocyte nadir, a predicator of chemotherapy efficacy [editorial]? Br J Cancer 1999; 11: 1681
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kerbusch, T., de Kraker, J., Keizer, H.J. et al. Clinical Pharmacokinetics and Pharmacodynamics of Ifosfamide and its Metabolites. Clin Pharmacokinet 40, 41–62 (2001). https://doi.org/10.2165/00003088-200140010-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200140010-00004