Abstract
-
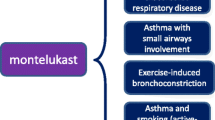
▴ Montelukast is a selective antagonist of the leukotriene D4 (LTD4) receptor. In patients with asthma, montelukast 5 to 250 mg/day attenuated LTD4-induced bronchoconstriction and, at a dosage of 10mg, significantly reduced early and late airway response to allergen (dust mite extract) relative to placebo.
-
▴ In studies evaluating the effects of various dosages of montelukast on exercise-induced bronchoconstriction the optimal dose of the drug was found to be 10mg.
-
▴ Montelukast 10 mg/day controlled asthma significantly more effectively than placebo in a 3-month randomised double-blind study. In a 9-month open extension of this trial, during which patients were randomised to treatment with montelukast 10 mg/day or beclomethasone (≈400 μg/day), daytime symptom score and β-agonist use decreased to a similar extent in each group.
-
▴ In a further study, treatment with montelukast 10 mg/day permitted clinically significant tapering of corticosteroid dosage in patients with stable asthma.
-
▴ Montelukast (5 mg/day) has also demonstrated efficacy in childhood asthma.
-
▴ The tolerability profile of montelukast was similar to that of placebo in placebo-controlled clinical trials in adults and children; the most common adverse event was headache.
Similar content being viewed by others
References
Jones TR, Champion E, Charette L, et al. Pharmacology of MK-0476, a potent and selective leukotriene D-4-receptor antagonist [abstract]. Am J Respir Crit Care Med 1994 Apr; 149 (2 Pt 1): 463
Grootendorst DC, Diamant Z, Veselic M, et al. Effect of oral montelukast, a cysLT1-receptor antagonist, on eosinophil counts and ECP in induced sputum before and after allergen challenge in asthmatics [abstract]. Am J Respir Crit Care Med 1997; 155(4): A976
Leff JA, Pizzichini E, Efthimiadis A, et al. Effect of montelukast (MK-0476) on airway eosinophilic inflammation in mildly uncontrolled asthma: a randomized placebo-controlled trial [abstract]. Am J Respir Crit Care Med 1997; 155(4): A977
De-Lepeleire I, Reiss TF, Rochette F, et al. Montelukast causes prolonged, potent leukotriene D4-receptor antagonism in the airways of patients with asthma. Clin Pharmacol Ther 1997 Jan; 61: 83–92
Diamant Z, Timmers MC, van der Veen H, et al. Effect of oral montelukast (MK-0476), a potent leukotriene receptor antagonist, on allergen-induced airway responses in asthmatic subjects [abstract]. Am J Respir Crit Care Med 1996; 153: A346
Reiss TF, Bronsky E, Hendeles L, et al. MK-0476, a potent leukotriene (LT)D4 receptor antagonist inhibits exercise induced bronchoconstriction in asthmatics at the end of a once daily dosing interval [abstract]. Am J Respir Crit Care Med 1995; 151(4): A377
Reiss TF, Bronsky E, Kemp J. MK-0476, a potent and specific leukotriene D4 receptor antagonist exhibits a dose response in the inhibition of exercise induced bronchoconstriction at the end of a once daily dosing interval [abstract]. J Invest Med 1995 Apr; 43 Suppl. 2: 275A
Bronsky EA, Kemp JP, Zhang J, et al. Dose-related protection of exercise bronchoconstriction by montelukast, a cysteinyl leukotriene-receptor antagonist, at the end of a once-daily dosing interval. Clin Pharmacol Ther 1997 Nov; 62: 556–61
Leff JA, Bronsky EA, Kemp J, et al. Montelukast (MK-0476) inhibits exercise-induced bronchoconstriction (EIB) over 12-weeks without causing tolerance [abstract]. Am J Respir Crit Care Med 1997; 155(4): A977
Kemp JP, Dockhorn RJ, Shapiro GG, et al. Montelukast, a leukotriene receptor antagonist, inhibits exercise-induced bronchoconstriction in 6- to- 14-year old children [abstract]. J Allergy Clin Immunol 1997 Jan; 99 (Pt 2): 321
Cheng H, Leff JA, Amin R, et al. Pharmacokinetics, bioavailability, and safety of montelukast sodium (MK-0476) in healthy males and females. Pharm Res 1996 Mar; 13: 445–8
Merck & Co., Inc. Montelukast prescribing information. White-house Station, New Jersey, USA, 1997
Zhao JJ, Rogers JD, Holland SD, et al. Pharmacokinetics and bioavailability of montelukast sodium (MK-0476) in healthy young and elderly volunteers. Biopharm Drug Dispos 1997 Dec; 18: 769–77
Balani SK, Xu X, Pratha V, et al. Metabolic profiles of montelukast sodium (Singulair), a potent cysteinyl leukotriene, receptor antagonist, in human plasma and bile. Drug Metab Dispos 1997 Nov; 25: 1282–7
Reiss TF, Sorkness CA, Stricker W, et al. Effects of montelukast (MK-0476), a potent cysteinyl leukotriene receptor antagonist, on bronchodilation in asthmatic subjects treated with and without inhaled corticosteroids. Thorax 1997 Jan; 52: 45–8
Reiss TF, Altman LC, Chervinsky P, et al. Effects of montelukast (MK-0476), a new potent cysteinyl leukotriene (LTD4) receptor antagonist, in patients with chronic asthma. J Allergy Clin Immunol 1996 Sep; 98: 528–34
Sorkness CA, Reiss TF, Zhang J, et al. Bronchodilation with a selective and potent leukotriene D-4 antagonist (MK-0476) in patients with asthma [abstract]. Am J Respir Crit Care Med 1994 Apr; 149 (Pt 2: 216
Reiss TF, Altaian LC, Munk ZM, et al. MK-0476 an LTD4 receptor antagonist improves the signs and symptoms of asthma with a dose as low as 10 mg once daily [abstract]. Am J Respir Crit Care Med 1995; 151(4): A378
Noonan MJ, Chervinsky P, Brandon M, et al. Montelukast, a potent leukotriene receptor antagonist, causes dose-related improvements in chronic asthma. Eur Respir J 1998 (11): 1232–1239
Reiss TF, Chervinsky P, Edwards T, et al. Montelukast (MK-0476), a CysLT1 receptor antagonist, improves the signs and symptoms of asthma over a 3 month treatment period [abstract]. Eur Respir J 1996 Sep; 9 Suppl. 23: 273
Reiss TF, White R, Noonan G, et al. Montelukast (MK-0476), A CysLT1 receptor antagonist, improves the signs and symptoms of asthma over one year of treatment [abstract]. Eur Respir J 1997 Sep; 10 (25 Suppl.): 437s
Leff JA, Israel E, Noonan MJ, et al. Montelukast (MK-0476) allows tapering of inhaled corticosteroids (ICS) in asthmatic patients while maintaining clinical stability [abstract]. Am J Respir Crit Care Med 1997; 155(4): A976
Kuna P, Malmstrom K, Dahlen SE, et al. Montelukast(MK-0476) a CysLTi receptor antagonist, improves asthma control in aspirin-intolerant asthmatic patients [abstract]. Am J Respir Crit Care Med 1997; 155 (4 Suppl.): A975
Knorr B, Matz J, Bernstein JA, et al. Montelukast for chronic asthma in 6- to 14-year-old children. JAMA 1998 Apr 15; 279(15): 1181–6
Knorr BA, Matz J, Sveum RJ, et al. Montelukast (MK-0476) improves asthma over 6 months of treatment in 6- to 14-year old patients [abstract]. Eur Respir J 1997 Sep; 10 Suppl. 25: 219s
Schoors DF, De Smet M, Reiss T, et al. Single dose pharmacokinetics, safety and tolerability of MK-0476, a new leukotriene D4-receptor antagonist, in healthy volunteers. Br J Clin Pharmacol 1995 Sep; 40: 277–80
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Markham, A., Faulds, D. Montelukast. Drugs 56, 251–256 (1998). https://doi.org/10.2165/00003495-199856020-00010
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199856020-00010