Summary
Abstract
Piperacillin/tazobactam is a β-lactam/β-lactamase inhibitor combination with a broad spectrum of antibacterial activity encompassing most Gram-positive and Gram-negative aerobic bacteria and anaerobic bacteria, including many pathogens producing β-lactamases.
Evidence from clinical trials in adults has shown that piperacillin/tazobactam, administered in an 8: 1 ratio, is an effective treatment for patients with lower respiratory tract, intra-abdominal, urinary tract, gynaecological and skin/soft tissue infections, and for fever in patients with neutropenia. Combination regimens of piperacillin/tazobactam plus an aminoglycoside are used to treat patients with severe nosocomial (hospital-acquired) infections.
In clinical trials, piperacillin/tazobactam was significantly more effective than ticarcillin/clavulanic acid in terms of clinical and microbiological outcome in patients with community-acquired pneumonia. In patients with intra-abdominal infections, clinical and bacteriological response rates were significantly higher with piperacillin/tazobactam than with imipenem/cilastatin (administered at a dosage lower than is recommended in countries outside Scandinavia).
Piperacillin/tazobactam in combination with amikacin was at least as effective as ceftazidime plus amikacin in the treatment of ventilator-associated pneumonia and was significantly more effective than ceftazidime plus amikacin in the empirical treatment of febrile episodes in patients with neutropenia or granulocytopenia. In other trials, the efficacy of piperacillin/tazobactam was similar to that of standard aminoglycoside-containing and other treatment regimens in patients with intra-abdominal, skin/soft tissue or gynaecological infections.
Piperacillin/tazobactam is generally well tolerated. The most frequent adverse events are gastrointestinal symptoms (most commonly diarrhoea) and skin reactions. The incidence of adverse events with piperacillin/tazobactam is higher when the combination is given in combination with an aminoglycoside than when given as monotherapy.
Conclusion: Because of the broad spectrum of antibacterial activity provided by piperacillin/tazobactam, it is useful for the treatment of patients with polymicrobial infections caused by aerobic or anaerobic β-lactamase-producing bacteria. Piperacillin/tazobactam appears to have a particularly useful role in the treatment of patients with intra-abdominal infections and, in combination with amikacin, in the treatment of patients with febrile neutropenia, especially given the current prevalence of Gram-positive infections in this group.
Rationale for the Use Of Piperacillin in Combination with Tazobactam
Although piperacillin has a broad spectrum of antibacterial activity, the increased prevalence of β-lactamase-producing bacteria over recent years has led to an increase in resistance to this agent, and has compromised its activity in the clinical setting. When coadministered with piperacillin, tazobactam, a β-lactamase inhibitor, restores and extends the antibacterial cover provided by piperacillin and thus enhances its clinical potential.
Pharmacodynamic Properties
Tazobactam shows good inhibitory activity against plasmid-mediated β-lactamases, staphylococcal penicillinase and chromosomal 2e β-lactamases. However, it is less inhibitory against group 1 β-lactamase subtypes and against group 3 metallo-β-lactamases. Tazobactam has a minimal ability to induce chromosomally mediated class I β-lactamases, whereas clavulanic acid is a moderate to strong inducer of these enzymes and thus has the potential to compromise the antibacterial activity of coadministered β-lactam agents.
Piperacillin/tazobactam has good in vitro activity against methicillin-sensitive Staphylococcus aureus and coagulase-negative staphylococci. Piperacillin/tazobactam is also active against Streptococcus pyogenes and penicillin-sensitive strains of S. pneumoniae. Most strains of Enterococcus faecalis are also susceptible to the combination. Methicillin-resistant strains of S. aureus and many methicillin-resistant coagulase-negative staphylococci are resistant to piperacillin/tazobactam. Strains of E. faecium were resistant to piperacillin/tazobactam in 2 in vitro studies.
Many Enterobacteriaceae, including Escherichia coli, Klebsiella spp. and Enterobacter spp. are susceptible to piperacillin/tazobactam. The combination exhibits excellent activity against Haemophilus influenzae and Moraxella catarrhalis and is active against Pseudomonas aeruginosa. Although there has been a change in susceptibility patterns of P. aeruginosa over recent years, with a general trend towards an increase in resistance to various antibacterial agents, piperacillin/tazobactam has maintained its activity against this pathogen: results of several recent surveys showed that 91 to 95% of isolates of P. aeruginosa were susceptible to piperacillin/tazobactam. Stenotrophomonas (Xanthomonas) maltophilia strains are resistant to the combination.
Piperacillin/tazobactam is highly active against anaerobes, including Bacteroides spp., notably B. fragilis. It is also highly active against Clostridium spp. The combination demonstrated good efficacy in murine models of intra-abdominal infection or pneumonia caused by extended spectrum β-lactamase-producing strains of K. pneumoniae.
Pharmacokinetic Properties
After single intravenous 4/0.5g doses of piperacillin/tazobactam, mean maximum plasma concentrations (Cmax) of piperacillin and tazobactam were 264.4 to 368 and 29.1 to 39 mg/L, respectively, in healthy adult volunteers. There was no evidence of accumulation of either agent after multiple doses in healthy adult volunteers or in adult patients with infection. The ratio of piperacillin Cmax to tazobactam Cmax was about 8: 1 in healthy volunteers or in patients with intra-abdominal infections after multiple doses of 4/0.5g 6- or 8-hourly. However, mean piperacillin area under the plasma concentration-time curve (AUC) values were >2-fold higher in patients with burns than in healthy volunteers or in patients with intra-abdominal infections.
At steady-state, the volume of distribution of piperacillin ranged from 15 to 21L in healthy volunteers and patients with infection; the corresponding range for tazobactam was 18 to 34.6L. Both agents are rapidly and widely distributed in various body tissues and fluids.
About 50 to 60% of an administered dose of piperacillin/tazobactam is excreted via the renal route; biliary excretion accounts for the elimination of <2% of the dose. In healthy volunteers or patients with intra-abdominal infection, piperacillin and tazobactam each have a plasma elimination half-life of 0.8 to 1 hour. Dosage reduction is required for patients with moderate renal impairment [creatinine clearance <1.2 L/h (<20 ml/min)]. As haemodialysis removes up to 50% of piperacillin/tazobactam over 4 hours, an additional 2/0.25g dose should be given after each dialysis session. The pharmacokinetics of piperacillin/ tazobactam are not markedly affected in patients with hepatic impairment. In children with infection, mean Cmax and AUC values of piperacillin and tazobactam increase in a dose-dependent manner.
The pharmacokinetics of piperacillin and tazobactam are not markedly altered when it is coadministered with vancomycin, tobramycin, ondansetron or ranitidine.
Therapeutic Efficacy
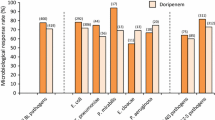
Clinical cure or improvement was achieved in 85 to 94% of patients with community-acquired lower respiratory tract infections treated with various dosages of piperacillin/tazobactam. At a dosage of 3/0.375g 6-hourly, piperacillin/ tazobactam was significantly more effective than ticarcillin/clavulanic acid 3/0. 1g 4 times daily in patients with community-acquired pneumonia. Evaluations at the trial end-point (generally 10 to 14 days after discontinuation of treatment) showed favourable clinical responses in 84 and 64% of piperacillin/ tazobactam and ticarcillin/clavulanic acid recipients, respectively (p < 0.01). Piperacillin/tazobactam also achieved a significantly higher rate of bacterial eradication than ticarcillin/clavulanic acid at the end of treatment (91 vs 68%; p < 0.01) and 10 to 14 days later (91 vs 83%; p = 0.02).
In patients with nosocomial pneumonia associated with mechanical ventilation in the intensive care unit, piperacillin/tazobactam 4/0.5g 4 times daily plus amikacin 7.5 mg/kg twice daily was at least as effective as ceftazidime 1g 4 times daily plus amikacin 7.5 mg/kg twice daily, with successful clinical and bacteriological outcomes documented in 51 and 36% of piperacillin/tazobactam- and ceftazidime-treated patients 6 to 8 days after the end of treatment. The efficacy of piperacillin/tazobactam was similar to that of imipenem/cilastatin in patients with nosocomial pneumonia. In patients with hospital-acquired acute purulent bronchitis or acute bacterial pneumonia, piperacillin/tazobactam 3/0.375g every 4 hours (plus tobramycin or amikacin) was significantly more effective than ceftazidime 2g every 8 hours (plus tobramycin or amikacin); clinical responses at the study end-point were achieved in 75 and 50% of patients (p < 0.01).
Rates of bacterial eradication ranged from 76 to 100% in patients with intra-abdominal infections treated with piperacillin/tazobactam. The clinical efficacy of piperacillin/tazobactam was similar to that of clindamycin plus gentamicin and in 1 study was significantly better than that of imipenem/cilastatin 0.5g 8-hourly (a dosage lower than is recommended in countries outside Scandinavia). Piperacillin/tazobactam (80/10 mg/kg 8-hourly) was also beneficial in the treatment of children with appendicitis or peritonitis, with 91% of patients experiencing cure or improvement.
Clinical success rates of 41 to 83% were reported in patients with febrile neutropenia or granulocytopenia who received empirical treatment with piperacillin/tazobactam 12–16/1.5–2 g/day (in divided doses) in combination with an aminoglycoside. 72 hours after the initiation of treatment, clinical response rates were significantly higher in patients treated with piperacillin/tazobactam plus amikacin than in ceftazidime plus amikacin-treated patients (61% vs 45 or 54%; p ≤ 0.05). In similar patients, piperacillin/tazobactam in combination with gentamicin was significantly more effective than piperacillin/gentamicin; clinical response rates of 83 and 48% (p < 0.001) were reported at 72 hours.
The efficacy of piperacillin/tazobactam monotherapy was similar to that of ceftazidime plus amikacin in patients with febrile neutropenia with 81 and 83% of febrile episodes resolved in patients treated with piperacillin/tazobactam and ceftazidime plus amikacin; median times to fever defervescence were also similar in the 2 treatment groups (3.3 vs 2.9 days).
The piperacillin/tazobactam combination also showed good clinical and bacteriological efficacy in patients with bacteraemia and in patients with skin and soft tissue, gynaecological or bone and joint infections. Piperacillin/tazobactam is also an effective treatment for patients with complicated urinary tract infections, achieving cure or improvement in 88 and 90.4% of patients 5 to 9 days after the end of treatment and in ≥80% of patients after 4 to 6 weeks of follow-up. Bacterial eradication rates after the same period of follow-up were 79.6 and 73%; E. coli, K. pneumoniae and P. aeruginosa were identified as common persistent pathogens.
Pharmacoeconomic Considerations
In a US economic evaluation, piperacillin/tazobactam was estimated to be less costly than comparator therapies in patients with community-acquired lower respiratory tract or intra-abdominal infection. Direct costs of therapy with piperacillin/tazobactam 3/0.375g 6-hourly were $US2981 per patient lower than with ticarcillin/clavulanic acid 3/0.1g 6-hourly in patients with community-acquired pneumonia. In patients with intra-abdominal infections, direct costs of piperacillin/tazobactam treatment were $US284 per patient lower than costs of treatment with clindamycin plus gentamicin.
A UK study showed that direct costs of treatment of patients with intra-abdominal infections were lower for piperacillin/tazobactam than for ceftazidime plus metronidazole alone or in combination with either gentamicin or netilmicin. Costs of acquisition, preparation, administration and waste disposal were included in this analysis.
However, although the acquisition cost of piperacillin/tazobactam is lower than that of imipenem/cilastatin, a comparative cost analysis showed a cost advantage for the latter combination when the number of days spent in hospital was included in the economic model.
Results of a Canadian study that included patients with various serious infections showed that direct per patient costs of piperacillin/tazobactam and imipenem/cilastatin were broadly similar ($Can696 vs $Can762).
A pharmacoeconomic study, based on data from a trial conducted in patients with febrile neutropenia, showed that piperacillin/tazobactam plus amikacin was more cost effective than ceftazidime plus amikacin: estimated costs per successfully treated patient were DM16 616 and DM20 828 for piperacillin/tazobactam plus amikacin and ceftazidime plus amikacin, respectively. Both direct and indirect costs of treatment were incorporated in this model.
Tolerability
Pooled data from numerous clinical trials indicate that piperacillin/tazobactam, given in dosages of up to 4/0.5g 6-hourly, is generally well tolerated in patients with mild, moderate or severe infections. The most frequently reported adverse events are gastrointestinal symptoms (most commonly diarrhoea) and skin reactions. Adverse events are typically mild or moderate in severity and rarely necessitate the discontinuation of treatment. Incidences of these types of events increase markedly in patients receiving piperacillin/tazobactam in combination with an aminoglycoside.
Other adverse events reported in patients receiving piperacillin/tazobactam include minor changes in laboratory test values (e.g. increases in alanine amino-transferase and in total bilirubin).
Pseudomembranous colitis, bleeding manifestations and anaphylactic reactions have been reported in patients receiving penicillins, including piperacillin.
Dosage and Administration
Piperacillin/tazobactam is given intravenously as a bolus injection over 3 to 5 minutes or by infusion over 20 to 30 minutes. Recommended dosages range from 2/0.25g every 6 to 12 hours (for the treatment of patients with relatively mild infections) to 4/0.5g every 6 or 8 hours for the treatment of more severe infections.
Article PDF
Similar content being viewed by others
References
Bryson HM, Brogden RE. Piperacillin/tazobactam: a review of its antibacterial activity, pharmacokinetic properties and therapeutic potential. Drugs 1994 Mar; 47: 506–35
Marra F, Jewesson P. A comparison of piperacillin/tazobactam versus imipenem: formulary considerations. J Infect Dibs Pharmacother 1997; 2(3): 55–80
Daniel KP, Krop LC. Piperacillin-tazobactam: a new β-lactam-β-lactamase inhibitor combination. Pharmacotherapy 1996 Mar–Apr; 16: 149–62
Schoonover LL, Occhipinti DJ, Rodvold KA, et al. Piperacillin/tazobactam: a new beta-lactam/beta-lactamase inhibitor combination. Ann Pharmacother 1995 May; 29: 501–14
Walmsley S, Committee on Antimicribial Agents. Piperacillin/ tazobactam. Can J Infect Dis 1997 Mar–Apr; 8: 79–84
Bush K, Jacoby GA, Medeiros AA. A functional classification scheme for β-lactamases and its correlation with molecular structure. Antimicrob Agents Chemother 1995: 39(6): 1211–33
Coleman K, Griffin DRJ, Page JWJ, et al. In vitro evaluation of BRL42715, a novel β-lactamase inhibitor. Antimicrob Agents Chemother 1989; 33: 1580–7
Hart SM, Bailey EM. A practical look at the clinical usefulness of the beta-lactam/beta-lactamase inhibitor combinations. Ann Pharmacother 1996 Oct; 30: 1130–40
Pankuch GA, Jacobs MR, Rittenhouse SF, et al. Susceptibilities of 123 strains of Xanthomonas maltophilia to eight β-lactams (including β-lactam-β-lactamase inhibitor combinations) and ciprofloxacin tested by five methods. Antimicrob Agents Chemother 1994 Oct; 38: 2317–22
Sanders Jr WE, Sanders CC. Piperacillin/tazobactam: a critical review of the evolving clinical literature. Clin Infect Dis 1996 Jan; 22: 107–23
Bonomo RA, Rudin SA, Shlaes DM. Tazobactam is a potent inactivator of selected inhibitor-resistant class A β-lactamases. FEMS Microbiol Lett 1997 Mar 1; 148: 59–62
Forward KR, Franks PA, Low DE, et al. Cross-Canada survey of resistance of 2747 aerobic blood culture isolates to piperacillin/tazobactam and other antibiotics. Can J Infect Dis 1998 Jan–Feb; 9: 33–44
Murray PR, Cantrell HF, Lankford RB, et al. Multicenter evaluation of the in vitro activity of piperacillin-tazobactam compared with eleven selected β-lactam antibiotics and ciprofloxacin against more than 42,000 aerobic Gram-positive and Gram-negative bacteria. Diagn Microbiol Infect Dis 1994 Jun; 19: 111–20
Marshall SA, Wilke WW, Chen Y-S, et al. In vitro activity of cefepime and other broad-spectrum β-lactams tested against 129 mec A-negative Staphylococcus spp. isolates: a multicenter sample. Diagn Microbiol Infect Dis 1998 Jan; 30: 65–9
Endtz HP, van Dijk WC, Verbrugh HA, et al. Comparative in-vitro activity of meropenem against selected pathogens from hospitalized patients in The Netherlands. J Antimicrob Chemother 1997 Feb; 39: 149–56
Pfaller MA, Jones RN, Doern GV. Multicenter evaluation of the antimicrobial activity for six broad-spectrum β-lactams in Venezuela using the Etest method. Diagn Microbiol Infect Dis 1998 Jan; 30: 45–52
Gould IM, Milne K. In-vitro pharmacodynamic studies of piperacillin/tazobactam with gentamicin and ciprofloxacin. J Antimicrob Chemother 1997 Jan; 39: 53–61
Dembry LM, Roberts JC, Schock KD, et al. Comparison of n vitro activity of trovafloxacin against Gram-positive and Gram-negative organisms with quinolones and β-lactam antimicrobial agents. Diagn Microbiol Infect Dis 1998; 31: 301–11
Doern GV, Jones RN, Pfaller MA, et al. Multicenter evaluation of the in vitro activity of six broad-spectrum β-lactam antimicrobial agents in Puerto Rico. Diagn Microbiol Infect Dis 1998 Feb; 30: 113–9
Stobberingh EE, Maclaren DM, Schmitz PIM. Comparative in-vitro activity of piperacillin-tazobactam against recent clinical isolates, a Dutch national multicentre study. J Antimicrob Chemother 1994 Nov; 34: 777–83
Marshall SA, Aldridge KE, Allen SD, et al. Comparative antimicrobial activity of piperacillin-tazobactam tested against more than 5000 recent clinical isolates from five medical centers: a reevaluation after five years. Diagn Microbiol Infect Dis 1995 Mar; 21: 153–68
Fung-Tomc J, Bush K, Minassian B, et al. Antibacterial activity of BMS-180680, a new catechol-containing monobactam. Antimicrob Agents Chemother 1997 May; 41: 1010–6
Kuck NA, Jacobus NV, Spengler MD, et al. In vitro activity of piperacillin/tazobactam against isolates from patients enrolled in clinical trials. Int J Antimicrob Agents 1996; 7(1): 15–21
Qadri SM, Ueno Y, Cunha BA. Susceptibility of clinical isolates to expanded-spectrum β-lactams alone and in the presence of β-lactamase inhibitors. Chemotherapy 1996 Sep–Oct; 42: 334–42
Verbist L, Verhaegen J, Wouters C, et al. Comparative activity of piperacillin/tazobactam against 5625 isolates from hospitalised patients. J Antimicrob Chemother 1996 Feb; 37: 285–93
Pankuch GA, Jacobs MR, Appelbaum PC. Susceptibilities of 200 penicillin-susceptible and -resistant pneumococci to piperacillin, piperacillin-tazobactam, ticarcillin, ticarcillin-clavulanate, ampicillin, ampicillin-sulbactam, ceftazidime, and ceftriaxone. Antimicrob Agents Chemother 1994 Dec; 38: 2905–7
Klepser ME, Marangos MN, Zhu Z, et al. Comparison of the bactericidal activities of piperacillin-tazobactam, ticarcillin-clavulanate, and ampicillin-sulbactam against clinical isolates of Bacteroides fragilis, Enterococcus faecalis, Escherichia coli, and Pseudomonas aeruginosa. Antimicrob Agents Chemother 1997 Feb; 41: 435–9
Nomura S, Hanaki H, Furukawa T, et al. In vitro and in vivo antibacterial activities of TOC-50, a new parenteral cephalosporin, against Enterococcus faecalis. Chemotherapy 1996 Nov–Dec; 42: 432–8
Hoellman DB, Visalli MA, Jacobs MR, et al. Activities and time-kill studies of selected penicillins, β-lactamase inhibitor combinations and glycopeptides against Enterococcus faecalis. Antimicrob Agents Chemother 1998; 42(4): 857–61
Traub WH, Leonhard B. Piperacillin tazobactam compared with co-amoxiclav, ampicillin plus sulbactam and timentin against beta-lactamase-producing clinical isolates of Escherichia coli, Klebsiella pneumoniae and Klebsiella oxytoca. Chemotherapy 1995 Sep–Oct; 41: 345–52
Jett BD, Ritchie DJ, Reichley R, et al. In vitro activities of various β-lactam antimicrobial agents against clinical isolates of Escherichia coli and Klebsiella spp. resistant to oxyimino cephalosporins. Antimicrob Agents Chemother 1995 May; 39: 1187–90
Liebowitz LD, Klugman KP. Comparative in vitro activity of piperacillin/tazobactam against Gram-negative bacilli. S Afr Med J 1996 Oct; 86: 1276–80
Platsouka E, Zissis NP, Constantoulaki S, et al. Comparative in vitro evaluation of piperacillin/tazobactam in a tertiary care hospital. J Chemother 1997 Oct; 9: 336–40
Visalli MA, Jacobs MR, Moore TD, et al. Activities of β-lactams against Acinetobacter genospecies as determined by agar dilution and E-test MIC methods. Antimicrob Agents Chemother 1997 Apr; 41: 767–70
Spangler SK, Visalli MA, Jacobs MR, et al. Susceptibilities of non-Pseudomonas aeruginosa Gram-negative nonfermentative rods to ciprofloxacin, ofloxacin, levofloxacin, D-ofloxacin, sparfloxacin, cetrazidime, piperacillin, piperacillin-tazobactam, trimethoprim-sulfamethoxazole, and imipenem. Antimicrob Agents Chemother 1996 Mar; 40: 772–5
Fass RJ, Barnishan J, Solomon MC, et al. In vitro activities of quinolones, β-lactams, tobramycin, and trimethoprim-sulfamethoxazole against nonfermentative Gram-negative bacilli. Antimicrob Agents Chemother 1996 Jun; 40: 1412–8
Pina P, Lorin de Reure V, Godard T, et al. Stenotrophomonas maltophilia: betalactam susceptibility in 12 French hospitals [in French]. Med Mal Infect 1997 Mar; 27: 271–3
Moody J, Olson D. Synergistic potential of ticarcillin clavulanate (TCA) or piperacillin tazobactam (PTZ) combined with ciprofloxacin (CIP) or trimethoprim sulfamethoxazole (SXT) against Xanthomonas maltophilia [abstract]. 34th Interscience Conference on Antimicrobial Agents and Chemotherapy 1994: 223
Nishida K, Higashitani F, Hyodo A. Superior effect of tazobactam/piperacillin compared to piperacillin on beta-lactamase-producing Pseudomonas aeruginosa. Chemotherapy Basel 1997 May–Jun; 43: 171–8
Jones RN, Pfaller MA, Marshall SA, et al. Antimicrobial activity of 12 broad-spectrum agents tested against 270 nosocomial blood stream infection isolates caused by non-enteric Gram-negative bacilli: occurrence of resistance, molecular epidemiology, and screening for metallo-enzymes. Diagn Microbiol Infect Dis 1997; 29: 187–92
National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial susceptibility testing. NCCLS Document M100-S7 1997; 17 (2)
Baron EJ, Jones RN. National survey of the in vitro spectrum of piperacillin-tazobactam tested against more than 40,000 aerobic clinical isolates from 236 medical centers. Diagn Microbiol Infect Dis 1995 Mar; 21: 141–51
Pankuch GA, Visalli MA, Jacobs MR, et al. Activities of oral and parenteral agents against penicillin-susceptible and -resistant pneumococci. Antimicrob Agents Chemother 1995 Jul; 39: 1499–504
Stefani S, Bonfiglio G, Bianchi C, et al. Susceptibility survey of piperacillin/tazobactam and some β-lactam comparators in Italy. Drugs Exp Clin Res 1998; XXIV(2): 105–13
Hoogkamp-Korstanje JAA, Verduyn Lunel F, Meis JFGM, et al. Ceftpirome: epidemiological survey in intensive care units and hematological units in the Netherlands. Diagn Microbiol Infect Dis 1998; 31: 489–91
Gobernado M, Bouza E, Perea E, et al. National multicenter study of the in vitro activity of piperacillin-tazobactam. Rev Esp Quimioter 1998; 11(2): 139–46
Lederle Laboratories. Piperacillin sodium and tazobactam sodium prescribing information. Physicians Desk Reference Generics 1998 (Fourth Edition)
Hanberger H, Garcia-Rodriguez J-A, Gobernado M, et al. Antibiotic susceptibility among aerobic Gram-negative bacilli in intensive care units in 5 European countries. JAMA 1999; 2281(1): 67–71
Gorbach SL. Treatment of intra-abdominal infections. J Antimicrob Chemother 1993; 31 Suppl. A: 67–78
Pierard D, DeMeyer A, Rosseel P, et al. In vitro activity of amoxycillin/clavulanate and ticarcillin/clavulanate compared with that of other antibiotics against anaerobic bacteria: comparison with the results of the 1987 survey. Acta Clin Belg 1996; 51: 70–9
Hecht DW, Lederer L. Effect of choice of medium on the results of in vitro susceptibility testing of eight antibiotics against the Bacteroides fragilis group. Clin Infect Dis 1995 Jun; 20 Suppl. 2: 346–9
Spangler SK, Jacobs MR, Appelbaum PC. Activity of CP 99,219 compared with those of ciprofloxacin, grepafloxacin, metronidazole, cefoxitin, piperacillin, and piperacillin-tazobactam against 489 anaerobes. Antimicrob Agents Chemother 1994 Oct; 38: 2471–6
Aldridge KE, Gelfand M, Relier LB, et al. A five-year multicenter study of the susceptibility of the Bacteroides fragilis group isolates to cephalosporins, cephamins, penicillins, clindamycin, and metronidazole in the United States. Diagn Microbiol Infect Dis 1994 Apr; 18: 235–41
Namavar F, Severin WPJ, Stobberingh E, et al. The sensitivity of clinical isolates of anaerobic species to piperacillin-tazobactam and other antimicrobial agents. J Antimicrob Chemother 1994 Sep; 34: 415–9
Betriu C, Gómez M, Sánchez A, et al. Piperacillin-tazobactam: comparative in vitro activity against Bacteroides fragilis group [in Spanish]. Rev Esp Quimioter 1997 Jun; 10: 161–6
Borobio MV, Suárez AI, Ramirez E, et al. Evaluation of in vitro activity of a new fluoroquinolone trovafloxacin (cp-99,219) compared with other anti-anaerobic antimicrobials against members of the Bacteroides fragilis group. Int J Antimicrob Agents 1997 May; 8: 249–52
Snydman DR, McDermott L, Cuchural GJ Jr, et al. Analysis of trends in antimicrobial resistance patterns among clinical isolates of Bacteroides fragilis group species from 1990 to 1994. Clin Infect Dis 1996 Dec; 23 Suppl 1: S54–65
Rice LB, Carias LL, Shlaes DM. In vivo efficacies of β-lactam-β-lactamase inhibitor combinations against a TEM-26-producing strain of Klebsiella pneumoniae. Antimicrob Agents Chemother 1994 Nov; 38: 2663–4
Thauvin-Eliopoulos C, Tripodi M-F, Moellering RCJr, et al. Efficacies of piperacillin-tazobactam and cefepime in rats with experimental intra-abdominal abscesses due to an extended-spectrum β-lactamase-producing strain of Klebsiella pneumoniae. Antimicrob Agents Chemother 1997 May; 41: 1053–7
Fournier J-L, Ramisse F, Jacolot AC, et al. Assessment of two penicillins plus β-lactamase inhibitors versus cefotaxime in treatment of murine Klebsiella pneumoniae infections. Antimicrob Agents Chemother 1996 Feb; 40: 325–30
Livermore DM, Yuan M. Antibiotic resistance and production of extended-spectrum β-lactamases amongst Klebsiella spp. from intensive care units in Europe. J Antimicrob Chemother 1996 Sep; 38: 409–24
Urban C, Rahal JJ. Klebsiella and extended spectrum β-lactamases. Int J Antimicrob Agents 1997; 8(1): 37–43
Quinn JP. Clinical significance of extended-spectrum beta-lactamases. Eur J Clin Microbiol Infect Dis 1994; 13 Suppl. 1: S39–42
Reed MD, Goldfarb J, Yamashita TS. Single-dose pharmacokinetics of piperacillin and tazobactam in infants and children. Antimicrob Agents Chemother 1994 Dec; 38: 2817–26
Bourget P, Lesne-Hulin A, Le Reveille RR. Clinical pharmacokinetics of piperacillin-tazobactam combination in patients with major burns and signs of infection. Antimicrob Agents Chemother 1996 Jan; 40: 139–45
Jhee SS, Kern JW, Burm J-P, et al. Piperacillin-tazobactam pharmacokinetics in patients with intraabdominal infections. Pharmacotherapy 1995 Jul–Aug; 15: 472–8
Occhipinti DJ, Pendland SL, Schoonover LL, et al. Pharmacokinetics and pharmacodynamics of two multiple-dose piperacillin-tazobactam regimens. Antimicrob Agents Chemother 1997 Nov; 41: 2511–7
Jehl F, Muller-Serieys C, deLV, et al. Penetration of piperacillin-tazobactam into bronchial secretions after multiple doses to intensive care patients. Antimicrob Agents Chemother 1994 Dec; 38: 2780–4
Sunder-Plasmann L, Halter G, Kinzig M, et al. Penetration of piperacillin/tazobactam into the respiratory tract [abstract]. Pharm Res 1996 Sep; 13 Suppl.: S119
Westphal J-F, Brogard J-M, Caro-Sampara F, et al. Assessment of biliary excretion of piperacillin-tazobactam in humans. Antimicrob Agents Chemother 1997 Aug; 41: 1636–40
Incavo SJ, Ronchetti PJ, Choi JH, et al. Penetration of piperacillin-tazobactam into cancellous and cortical bone tissues. Antimicrob Agents Chemother 1994 Apr; 38: 905–7
Kinzig M, Sörgel F, Brismar B, et al. Pharmacokinetics and tissue penetration of tazobactam and piperacillin in patients undergoing colorectal surgery. Antimicrob Agents Chemother 1992; 36: 1997–2004
Von Rosenstiel N-A, Grimm H. Antibiotic profile: piperacillin/ tazobactam. Antiinfective Drugs Chemother 1996; 14(3): 187–99
Sörgel F, Kinzig M. The chemistry, pharmacokinetics and tissue distribution of piperacillin/tazobactam. J Antimicrob Chemother 1993; 31 Suppl. A: 39–60
Wyeth Laboratories. Piperacillin/tazobactam prescribing information. 1997 (ABPI Compendium of Data Sheets and Summaries of Product Characteristics)
Fujii R, Okuno A, Fujita K, et al. Comprehensive evaluation of pharmacokinetic and clinical studies on tazobactam/piperacillin in pediatric field [in Japanese]. Jpn J Antibiot 1995 Mar; 48: 311–45
Choi J-S, Burm J-P, Jhee SS, et al. Stability of piperacillin sodium-tazobactam sodium and ranitidine hydrochloride in 0.9% sodium chloride injection during simulated Y-site administration. Am J Hosp Pharm 1994 Sep 15; 51: 2273–6
Chung KC, Moon YSK, Chin A, et al. Compatibility of ondansetron hydrochloride and piperacillin sodium-tazobactam sodium during simulated Y-site administration. Am J Health System Pharm 1995 Jul 15; 52: 1554–6
Hitt CM, Patel KB, Nicolau DP, et al. Influence of piperacillin-tazobactam on pharmacokinetics of gentamicin given once daily. Am J Health System Pharm 1997 Dec 1; 54: 2704–8
Shlaes DM, Baughman R, Boylen CT, et al. Piperacillin/ tazobactam compared with ticarcillin/clavulanate in community-acquired bacterial lower respiratory tract infection. J Antimicrob Chemother 1994 Oct; 34: 565–77
Jaccard C, Cometta A, Harbhardt S, et al. Prospective randomized comparison of imipenem (IM) and piperacillin/ tazobactam (PT) for the treatment of nosocomial pneumonia or peritonitis [abstract]. 36th Interscience Conference on Antimicrobial Agents and Chemotherapy. 1996 Sep 15: 285
Jaccard C, Troillet N, Harbarth S, et al. Prospective randomized comparison of imipenem-cilastatin and piperacillin-tazobactam in nosocomial pneumonia or peritonitis. Antimicrob Agents Chemother 1998; 42(11): 2966–72
Joshi M, Solomkin JS, Bernstein JM, et al. Open, randomized, multicenter comparison of piperacillin/tazobactam (PIP/TAZ) vs ceftazidime (CAZ), both plus tobramycin (TM), in 300 patients with hospital-acquired lower respiratory tract infections (LRTI) [poster]. 6th International Congress for Infectious Diseases in Prague. 1994.
Joshi M, Bernstein J, Solomkin J, et al. Piperacillin/tazobactam plus tobramycin versus ceftazidime plus tobramycin for the treatment of patients with nosocomial lower respiratory tract infection. Philadelphia: Wyeth Ayerst Laboratories, 1998. (Data on file)
Oizumi K, Ohno T, Kawahara M, et al. A comparative study of tazobactam/piperacillin and piperacillin in bacterial pneumonia [in Japanese]. Jpn J Antibiot 1995 Apr; 48: 449–81
Oizumi K, Kawahara M, Kawaguchi S, et al. A comparative study of tazobactam/piperacillin and piperacillin in chronic respiratory tract infections [in Japanese]. Jpn J Antibiot 1995 Apr; 48: 482–513
Speich R, Imhof E, Vogt M, et al. Efficacy, safety, tolerance of piperacillin/tazobactam compared to co-amoxiclav plus an aminoglycoside in the treatment of severe pneumonia. Eur J Clin Microbiol Infect Dis 1998; 17: 313–7
Sifuentes-Osornio J, Ruíz-Palacios GM, Jakob E, et al. Piperacillin/tazobactam in the treatment of lower respiratory tract infections: an open non-comparative and multicentered trial. J Chemother 1994 Jun; 6: 197–203
Smith DL. Parenteral piperacillin/tazobactam combined with amikacin for the treatment of severe pulmonary infections in intensive care units. Infect Dis Clin Pract 1995 Mar–Apr; 4 Suppl. 1: 33–6
Mouton Y, Leroy O, Beuscart C, et al. Efficacy, safety and tolerance of parenteral piperacillin/tazobactam in the treatment of patients with lower respiratory tract infections. J Antimicrob Chemother 1993; 31 Suppl. A: 87–95
Brun-Buisson C, Sollet JP, Schweich H, et al. Treatment of ventilator-associated pneumonia with piperacillin-tazobactam/ amikacin versus ceftazidime/amikacin: a multicenter, randomized controlled trial. Clin Infect Dis 1998 Feb; 26: 346–54
Davis R, Bryson HM. Ceftriaxone: a pharmacoeconomic evaluation of its use in the treatment of serious infections. Pharmacoeconomics 1994 Sep; 6: 249–69
Smith JMB, Payne JE. Antimicrobial therapy in selected surgical patients. Aust N Z J Surg 1994; 64(10): 658–66
Wilson SE, Nord CE. Clinical trials of extended spectrum penicillin/beta-lactamase inhibitors in the treatment of intra-abdominal infections: European and North American experience. Am J Surg 1995 May; 169 Suppl.: 21–6
Gorbach SL. Piperacillin/tazobactam in the treatment of polymicrobial infections. Intensive Care Med 1994 Jul; 20 Suppl. 3: S27–34
Barie PS, Bennion RS, Cheadle WG. Results of the North American trial of piperacillin/tazobactam compared with clindamycin and gentamicin in the treatment of severe intra-abdominal infections. Eur J Surg 1994; Suppl. 573: 61–6
Eklund A-E, Nord CE, Brismar B, et al. A randomized multicenter trial of piperacillin/tazobactam versus imipenem/ cilastin in the treatment of severe intra-abdominal infections. J Antimicrob Chemother 1993; 31 Suppl. A: 79–85
Polk Jr HC, Fink MP, Laverdiere M, et al. Prospective randomized study of piperacillin/tazobactam therapy of surgically treated intra-abdominal infection. American Surgeon 1993; 59: 598–605
Brismar B, Malmborg AS, Tunevall G, et al. Piperacillin-tazobactam versus imipenem-cilastin for treatment of intra-abdominal infections. Antimicrob Agents Chemother 1992; 36: 2766–73
Dupont H, Carbon C, Carlet J, et al. Multicenter randomized clinical trial of piperacillin plus tazobactam (TAZ) vs piperacillin plus tazobactam and amikacin (AMK) in the treatment of severe generalized peritonitis (SGP) [abstract]. 38th Interscience Conference on Antimicrobial Agents and Chemotherapy. 1998 Sep 24–27; San Diego, 602
Cohn S, Haverstock D, Kowalsky S, et al. The safety and efficacy of sequential (IV to PO) ciprofloxacin (CIP) plus metronidazole (MET) vs. piperacillin/Tazobactam (PIP/TAZO) in the treatment of patients with complicated intra-abdominal (INTRAAB) infections [abstract]. 38th Interscience Conference on Antimicrobial Agents and Chemotherapy. 1998 Sep 24–27; San Diego, 602
Niinikoski J, Havia T, Alhava E, et al. Piperacillin/tazobactam versus imipenem/cilastin in the treatment of intra-abdominal infections. Surg Gynecol Obstet 1993; 176: 255–61
Shyr Y-M, Lui W-L, Su C-H, et al. Piperacillin/tazobactam in comparison with clindamycin plus gentamicin in the treatment of intra-abdominal infections. Chung Hua I Hsueh Tsa Chih Taipei 1995; 56: 102–8
Legrand JC, Bastin F, Belva P, et al. Piperacillin-tazobactam treatment for severe intra-abdominal infections. Acta Chir Belg 1995 May–Jun; 95: 162–5
Vestweber KH, Grundel E. Efficacy and safety of piperacillin/ tazobactam in intra-abdominal infections. Eur J Surg 1994; Suppl. 573: 57–60
Nord CE. Treatment of intraabdominal infections: worldwide clinical trials. Infect Dis Clin Pract 1995 Mar–Apr; 4 Suppl. 1: 17–25
Arguedas A, Sifuentes-Osornio J, Loaiza C, et al. An open, multicenter clinical trial of piperacillin/tazobactam in the treatment of pediatric patients with intra-abdominal infections. J Chemother 1996 Apr; 8: 130–6
Hazel DL, Graham J, Dickinson JP, et al. Piperacillin-tazobactam as empiric monotherapy in febrile neutropenic patients with haematological malignancies. J Chemother 1997 Aug; 9: 267–72
Donnelly JP. Is there a rationale for the use of antimicrobial prophylaxis in neutropenic patients? J Intern Med 1997; 242 Suppl. 740: 79–88
Fauser AA, Kontz B, Bischoff M, et al. Tazobactam/piperacillin for the treatment of febrile episodes in neutropenic cancer patients with peripheral blood stem cell support [abstract]. Can J Infect Dis 1995 Jul; 6 Suppl. C: 265
Klastersky J. Empiric therapy for febrile neutropenia [abstract]. 8th International Congress on Infectious Diseases. 1998 May; 15–18, Boston, 108
Cometta A, Zinner S, de Bock R. Piperacillin-tazobactam plus amikacin versus ceftazidime plus amikacin as empiric therapy for fever in granulocytopenic patients with cancer. Antimicrob Agents Chemother 1995 Feb; 39: 445–52
Marie JP, Vekhoff A, Pico JL, et al. Neutropenic infections: a review of the French Febrile Aplasia Study Group trials in 608 febrile neutropenic patients. J Antimicrob Chemother 1998; 41: 57–64
Hughes WT, Armstrong D, Bodey GP, et al. 1997 Guidelines for the use of antimicrobial agents in neutropenic patients with unexplained fever. Clin Infect Dis 1997; 25: 551–73
Balfour JA, Bryson HM, Brogden RN. Imipenem/cilastatin: an update of its antibacterial activity, pharmacokinetics and therapeutic efficacy in the treatment of serious infections. Drugs 1996 Jan; 51: 99–136
Kern WV, Maute B, Engel A, et al. Piperacillin-tazobactam plus netilmicin as empiric treatment for adult patients with acute leukemia, neutropenia, and fever: single-center experience and retrospective comparison with piperacillin plus netilmicin. Curr Ther Res Clin Exp 1997 Jan; 58: 56–69
Micozzi A, Nucci M, Venditti M, et al. Piperacillin/tazobactam/amikacin versus piperacillin/amikacin/teicoplanin in the empirical treatment of neutropenic patients. Eur J Clin Microbiol Infect Dis 1993; 12(1): 1–8
Glasmacher A, Hesselmann B, Molitor E, et al. Piperacillin-tazobactam plus gentamicin as empiric therapy for fever in granulocytopenic patients with leukemia [abstract]. 7th Eur Congr Clin Microbiol Infect Dis, 1995 26–30 Mar: 244
Glasmacher A, Reeder K, Risse E, et al. Piperacillin, tazobactam and gentamicin as empiric antibiotic treatment for febrile neutropenic patients with leukaemia [685]. Onkologie 18 (Suppl. 2). 196
Hazel DL, Gale R, Graham J, et al. Piperacillin-tazobactam (Tazocin) as single agent empiric therapy in neutropenic fever [abstract]. 9th Immunocompromised Host Society meeting; 1996 Jun 23: abstr.026
Sousa AB, Fernandes JP, Ferreira G, et al. Piperacillin-tazobactam and amikacin (PT-A) as empirical therapy for fever in granulocytopenic cancer patients (pts). Eur J Cancer 1993; 29A Suppl. 6: S207
García-Bueno JM, Rifà J, Mesia R, et al. Piperacillin-tazobactam plus amikacin in the treatment of fever in neutropenic cancer patients [abstract]. Ann Oncol 1996; 7 Suppl. 5: 143
Marie JP, Vekhoff A, Cony-Makhoul P, et al. Piperacillin/ tazobactam + amikacin versus ceftazidime + amikacin in neutropenic patients with fever: an open multicentric trial [in French]. Presse Med 1995 Feb 25; 24: 397–401
Marie J-P, Marjanovic Z, Vekhoff A, et al. Piperacillin/tazobactam plus tobramycin versus ceftazidime as empiric therapy for fever in severely neutropenic patients. Support Care Cancer 1999; 7: 89–94
Rossini F, Isella M, Terruzzi E, et al. Empirical antibiotic therapy in severe neutropenia. Ceftriaxone + amikacin vs piperacillin-tazobactam + amikacin [abstract]. Br J Haematol 1998 Jul; 102(1): 93
Lee ES, Clark RE, Cawley P. Piperacillin/tazobactam/gentamicin (PTG) is a better initial empirical treatment than piperacillin/gentamicin (PG) in neutropenic pyrexia [abstract]. 8th International Congress on Infectious Diseases. 1998 May 15–18; Boston, 199
Bischoff M, Gengenbacher D, Blau W, et al. Arandomized clinical trial with piperacillin/tazobactam and piperacillin/tazobactam in combination with netilmicin in the treatment of fever of unknown origin in patients with severe neutropenia [abstract]. Onkologie 1997 Oct; 20 Suppl. 1: 132
Böhme A, Shah PM, Stille W, et al. Prospective randomized study to compare piperacillin/tazobactam versus cefepime as initial antimicrobial therapy in febrile neutropenic patients [abstract]. Br J Haematol 1998 Jul; 102(1): 94
Esteve J, Nomdedeu B, Mensa J, et al. Piperacillin/tazobactam vs. piperacillin/tazobactam plus amikacin as empiric therapy for fever in neutropenic patients [abstract]. Blood 1997; 90 (Suppl. 1, part 2): 229b
Hazel DL, Phelan L, Kinsey SE, et al. Piperacillin-tazobactam plus tobramycin is safe and effective therapy in children with febrile neutropenic episodes who are colonised with enterobacteriaceae expressing extended spectrum β-lactamases [abstract]. Blood 1998 Nov 15; 92 (Suppl. 1, part 1): 612a
Hess U, Böhme C, Rey K, et al. Monotherapy with piperacillin/tazobactam versus combination therapy with ceftazidime plus amikacin as an empiric therapy for fever in neutropenic cancer patients. Support Care Cancer 1998; 6: 402–9
Hughes WT, Pizzo PA, Wade JC, et al. Evaluation of new anti-infective drugs for the treatment of febrile episodes in neutropenic patients. Clin Infect Dis 1992; 15 Suppl. 1: S206–15
Sanders CV. Treatment of polymicrobial gynecologic and skin and skin-structure infections: worldwide clinical trials. Infect Dis Clin Pract 1995 Mar–Apr; 4 Suppl. 1: 26–32
Tassler H, Cullmann W, Elhardt D. Therapy of soft tissue infections with piperacillin/tazobactam. J Antimicrob Chemother 1993; 31 Suppl. A: 105–12
Tan JS, Wishnow RM, Talan DA, et al. Treatment of hospitalized patients with complicated skin and skin structure infections: double-blind, randomized, multicentre study of piperacillin-tazobactam versus ticarcillin/clavulanate. Antimicrob Agents Chemother 1993; 37: 1580–6
File TM, Tan JS. Efficacy and safety of piperacillin/tazobactam in skin and soft tissue infections. Eur J Surg 1994; Suppl. 573: 51–5
Sweet RL, Roy S, Faro S, et al. Piperacillin and tazobactam versus clindamycin and gentamicin in the treatment of hospitalized women with pelvic infection. Obstet Gynecol 1994 Feb; 83: 280–6
Nowé P. Piperacillin/tazobactam in complicated urinary tract infections. Intensive Care Med 1994 Jul; 20 Suppl. 3: S39–42
Sifuentes-Osornio J, Jakob E, Clara L, et al. Piperacillin/ tazobactam in the treatment of hospitalized patients with urinary tract infections: an open non-comparative and multicentered trial. J Chemother 1996 Apr; 8: 122–9
Ueda S, Yamashita T, Matsuoka K, et al. Tazobactam/piperacillin in chronic complicated urinary tract infection [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 782–6
Oshi M, Aso Y, Kishi H, et al. Clinical study on tazobactam/piperacillin for genitourinary tract infections [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 756–60
Koshida K, Hisazumi H. Clinical study of tazobactam/piperacillin in complicated urinary tract infections [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 770–3
Hayashi T, Watanabe T, Takenaka T, et al. Basic and clinical studies of tazobactam/piperacillin in the urological field [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 541–52
Yamashita M, Miyazaki S, Okada H, et al. Basic and clinical studies of tazobactam/piperacillin in the field of urology [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 529–40
Suzuki K, Horiba M, Kato S, et al. Clinical study of tazobactam/piperacillin in complicated urinary tract infections [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 513–20
Masuda A, Watanabe S, Usui Y, et al. Clinical investigation of tazobactam/piperacillin in urology [in Japanese]. Chemotherapy Tokyo 1994 Oct; 42 Suppl. 2: 765–9
Smyth ETM, Barr JG, Hogg GM. Hidden costs of antibiotics [letter]. Pharmacoeconomics 1996 Jul; 10: 99–100
Huse DM, Russell MW, Drowns S, et al. Economic impact of piperacillin/tazobactam in the treatment of suspected polymicrobial infections. J Clin Outcomes Manage 1998 Jan–Feb; 5: 20–30
Dietrich ES, Eisinger U, Hess U, et al. Cost-effectiveness of piperacillin/tazobactam versus ceftazidime/amikacin for the treatment of febrile episodes in neutropenic cancer patients [abstract]. 38th Interscience Conference on Antimicrobial Agents and Chemotherapy. 1998 Sep 24–27; San Diego, 605
Jhee SS, Gill MA, Yellin AE, et al. Pharmacoeconomics of piperacillin/tazobactam and imipenem/cilastatin in the treatment of patients with intra-abdominal infections. Clin Ther 1995 Jan–Feb; 17: 126–35
Marra F, Reynolds R, Stiver G, et al. Piperacillin/tazobactam versus imipenem: a double-blind, randomized formulary feasibility study at a major teaching hospital. Diagn Microbiol Infect Dis 1998; 30: 355–68
Smyth ETM, Hogg GM, Barr JG. Prescribing costs of piperacillin/tazobactam in comparison with cefotaxime/metronidazole and aminoglycoside combination therapy [abstract]. 2nd European Congress of Chemotherapy and 7th Biennial Conference on Antiinfective Agents and Chemotherapy. 1998 May 10–13; Hamburg.
Dominguez-Gil A, Pérez M, Santos L, et al. Cost-effectiveness of piperacillin-tazobactam versus ceftazidime in patients with febrile neutropenia [abstract]. Value in Health 1998; 1(1): 68
Kilburg A, Casser U, Rychlik R. Medical and economic relevance of treatment of febrile neutropenic cancer patients with piperacillin/tazobactam compared to ceftazidime [abstract]. Am J Manage Care 1997 Mar; 3 Suppl.: S58–9
Kuye O, Teal J, DeVries VG, et al. Safety profile of piperacillin/ tazobactam in phase I and III clinical studies. J Antimicrob Chemother 1993; 31 Suppl. A: 113–24
Pfyffer GE, Weder W, Strässle A, et al. Immune thrombocytopenia caused by piperacillin/tazobactam. Clin Infect Dis 1998; 27: 650–1
Pitout JDD, Sanders CC, Sanders Jr JWE. Antimicrobial resistance with focus on beta-lactam resistance in Gram-negative bacilli. Am J Med 1997 Jul; 103: 51–9
Cars O. Colonisation and infection with resistant Gram-positive cocci: epidemiology and risk factors. Drugs 1997; 54 Suppl. 6: 4–10
Cormican MG, Jones RN. Emerging resistance to antimicrobial agents in Gram-positive bacteria: enterococci, staphylococci and nonpneumococcal streptococci. Drugs 1996; 51 Suppl. 1: 6–12
Pitout JDD, Thomson KS, Hanson ND, et al. β-lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumonia, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob Agents Chemother 1998; 42(6): 1350–4
Sutherland R. beta-Lactam/beta-lactamase inhibitor combinations: development, antibacterial activity and clinical applications. Infection 1995 Jul–Aug; 23: 191–200
Klastersky J. Science and pragmatism in the treatment and prevention of neutropenic infection. J Antimicrob Chemother 1998; 41 Suppl. D: 13–24
Rice LB, Eckstein EC, DeVente J, et al. Ceftazidime-resistant Klebsiella pneumoniae isolates recovered at the Cleveland Department of Veterans Affairs Medical Center. Clin Infect Dis 1996 Jul; 23: 118–24
Postelnick MJ, Peterson LR, Noskin GA. Effect of changes in antimicrobial agent use on bacterial pathogens recovered from hospitalized patients: a 6-year assessment. J Clin Outcomes Manage 1998 Jul/Aug; 5(4): 21–32
Rice LB, Willey SH, Papanicolaou A, et al. Outbreak of ceftazidime resistance caused by extended-spectrum β-lactamases at a Massachusetts chronic-care facility. Antimicrob Agents Chemother 1990; 34(11): 2193–9
Quale J, Landman D, Saurina G, et al. Manipulation of a hospital antimicrobial formulary to control an outbreak of vancomycin-resistant enterococci. Clin Infect Dis 1996; 23: 1020–5
Noskin G, Postelnick M, Peterson L. Control of vancomycin resistant Enterococcus faecium by restriction of third generation cephalosporins. 94th General Meeting of the American Society for Microbiology. 1994 May 23–27; Las Vegas, L37
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perry, C.M., Markham, A. Piperacillin/Tazobactam. Drugs 57, 805–843 (1999). https://doi.org/10.2165/00003495-199957050-00017
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199957050-00017