Abstract
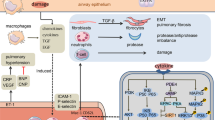
Chronic obstructive pulmonary disease (COPD) is a common, smoking-related, severe respiratory condition characterised by progressive, irreversible airflow limitation. Current treatment of COPD is symptomatic, with no drugs capable of halting the relentless progression of airflow obstruction. Better understanding of the airway inflammation, oxidative stress and alveolar destruction that characterise COPD has delineated new disease targets, with consequent identification of novel compounds with therapeutic potential.
These new drugs include aids to smoking cessation (e.g. bupropion) and improvements to existing therapies, for example long-acting rather than short-acting bronchodilators, as well as combination therapy. New antiproteases include acyl-enzyme and transition state inhibitors of neutrophil elastase (e.g. sivelestat and ONO-6818), matrix metalloprotease inhibitors (e.g. batimastat), cathepsin inhibitors and peptide protease inhibitors (e.g. DX-890 [EPI-HNE-4] and trappin-2). New antioxidants include Superoxide dismutase mimetics (e.g. AEOL-10113) and spin trap compounds (e.g. N-tert-butyl-α-phenylnitrone). New anti-inflammatory interventions include phosphodiesterase-4 inhibitors (e.g. cilomilast), inhibitors of tumour necrosis factor-α (e.g. humanised monoclonal antibodies), adenosine A2a receptor agonists (e.g. CGS-21680), adhesion molecule inhibitors (e.g. bimosiamose [TBC1269]), inhibitors of nuclear factor-KB (e.g. the naturally occurring compounds hypoestoxide and (−)-epigallocatechin-3-gallate) and activators of histone deacetylase (e.g. theophylline). There are also selective inhibitors of specific extracellular mediators such as chemokines (e.g. CXCR2 and CCR2 antagonists) and leukotriene B4 (e.g. SB201146), and of intracellular signal transduction molecules such as p38 mitogen activated protein kinase (e.g. RWJ67657) and phosphoinositide 3-kinase. Retinoids may be one of the few potential treatments capable of reversing alveolar destruction in COPD, and a number of compounds are in clinical trial (e.g. all-trans-retinoic acid). Talniflumate (MSI-1995), an inhibitor of human calcium-activated chloride channels, has been developed to treat mucous hypersecretion. In addition, the purinoceptor P2Y2 receptor agonist diquafosol (INS365) is undergoing clinical trials to increase mucus clearance.
The challenge to transferral of these new compounds from preclinical research to disease management is the design of effective clinical trials. The current scarcity of well characterised surrogate markers predicts that long-term studies in large numbers of patients will be needed to monitor changes in disease progression.











Similar content being viewed by others
References
Mannino DM. COPD: epidemiology, prevalence, morbidity and mortality, and disease heterogeneity. Chest 2002; 121 (5 Suppl.): 121S–6S
National Heart, Lung and Blood Institute, World Health Organization. Global initiative for chronic obstructive lung disease. Bethesda (MD): National Institutes of Health, 2001: 2701
Jeffery PK. Remodeling in asthma and chronic obstructive lung disease. Am J Respir Crit Care Med 2001; 164 (10 Pt 2): S28–38
Sullivan SD, Ramsey SD, Lee TA. The economic burden of COPD. Chest 2000; 117 (2 Suppl.): 5S–9S
Sandford AJ, Joos L, Pare PD. Genetic risk factors for chronic obstructive pulmonary disease. Curr Opin Pulm Med 2002; 8(2): 87–94
Anto JM, Vermeire P, Vestbo J, et al. Epidemiology of chronic obstructive pulmonary disease. Eur Respir J 2001; 17(5): 982–94
Seemungal T, Harper-Owen R, Bhowmik A, et al. Respiratory viruses, symptoms, and inflammatory markers in acute exacerbations and stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001; 164(9): 1618–23
Wilson R. Bacteria, antibiotics and COPD. Eur Respir J 2001; 17(5): 995–1007
Nathan RA, Geddes D, Woodhead M. Management of influenza in patients with asthma or chronic obstructive pulmonary disease. Ann Allergy Asthma Immunol 2001; 87(6): 447–54, 487
Barnes PJ. Chronic obstructive pulmonary disease. N Engl J Med 2000; 343(4): 269–80
Maestrelli P, Saetta M, Mapp CE, et al. Remodeling in response to infection and injury: airway inflammation and hypersecretion of mucus in smoking subjects with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001; 164 (10 Pt 2): S76–80
Tetley TD. Macrophages and the pathogenesis of COPD. Chest 2002; 121 (5 Suppl.): 156S–9S
Stockley RA. Neutrophils and the pathogenesis of COPD. Chest 2002; 121 (5 Suppl.): 151S–5S
Cosio MG, Majo J, Cosio MG. Inflammation of the airways and lung parenchyma in COPD: role of T cells. Chest 2002; 121 (5 Suppl.): 160S–5S
Jeffery PK. Lymphocytes, chronic bronchitis and chronic obstructive pulmonary disease. Novartis Found Symp 2001; 234: 149–61
Jeffery PK. Comparison of the structural and inflammatory features of COPD and asthma. Chest 2000; 117 (5 Suppl. 1): 251S–60S
Barnes PJ. Mechanisms in COPD: differences from asthma. Chest 2000; 117 (2 Suppl.): 10S–4S
Culpitt SV, Rogers DF. Evaluation of current pharmacotherapy of chronic obstructive pulmonary disease. Expert Opin Pharmacother 2000; 1(5): 1007–20
Calverley PM. Inhaled corticosteroids are beneficial in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2000; 161 (2 Pt 1): 341–2
Barnes PJ. Inhaled corticosteroids are not beneficial in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2000; 161 (2 Pt 1): 342–4
Shapiro SD. Evolving concepts in the pathogenesis of chronic obstructive pulmonary disease. Clin Chest Med 2000; 21(4): 621–32
Dawkins PA, Stockley RA. Animal models of chronic obstruc-tive pulmonary disease. Thorax 2001; 56(12): 972–7
Leckie MJ, Bryan SA, Hansel TT, et al. Novel therapy for COPD. Expert Opin Investig Drugs 2000; 9(1): 3–23
Agusti AGN. Surrogate markers in chronic obstructive pulmonary disease. Eur Respir Rev 2001; 10: 356–69
Kharitonov SA, Barnes PJ. Exhaled markers of pulmonary disease. Am J Respir Crit Care Med 2001; 163(7): 1693–722
Kostikas K, Papatheodorou G, Ganas K, et al. pH in expired breath condensate of patients with inflammatory airway diseases. Am J Respir Crit Care Med 2002; 165(10): 1364–70
Johnson M, Rennard S. Alternative mechanisms for long-acting beta(2)-adrenergic agonists in COPD. Chest 2001; 120(1): 258–70
Silagy C, Lancaster T, Stead L, et al. Nicotine replacement therapy for smoking cessation (Cochrane Review). Available in The Cochrane Library [database on CD ROM]. Updated quarterly. The Cochrane Collaboration; issue 2. Oxford: Update Software, 2002
Henningfield JE, Fant RV, Gitchell J, et al. Tobacco dependence: global public health potential for new medications development and indications. Ann N Y Acad Sci 2000; 909: 247–56
Hughes JR, Stead LF, Lancaster T. Antidepressants for smoking cessation. Cochrane Database Syst Rev 2002; (1): CD000031
Hughes JR, Stead LF, Lancaster T. Anxiolytics for smoking cessation. Cochrane Database Syst Rev 2000; (4): CD002849
Jamerson BD, Nides M, Jorenby DE, et al. Late-term smoking cessation despite initial failure: an evaluation of bupropion sustained release, nicotine patch, combination therapy, and placebo. Clin Ther 2001; 23(5): 744–52
Nardini S. Smoking cessation: the role of bupropion among new pharmacologic agents. Monaldi Arch Chest Dis 2001; 56(5): 467–72
Anderson JE, Jorenby DE, Scott WJ, et al. Treating tobacco use and dependence: an evidence-based clinical practice guideline for tobacco cessation. Chest 2002; 121(3): 932–41
Tashkin D, Kanner R, Bailey W, et al. Smoking cessation in patients with chronic obstructive pulmonary disease: a double-blind, placebo-controlled, randomised trial. Lancet 2001; 357(9268): 1571–5
Gourlay SG, Stead LF, Benowitz NL. Clonidine for smoking cessation. Cochrane Database Syst Rev 2000; (2): CD000058
Cerny EH, Levy R, Mauel J, et al. Preclinical development of a vaccine ‘against smoking’. Onkologie 2002; 25(5): 406–11
Pentel P, Malin D. A vaccine for nicotine dependence: targeting the drug rather than the brain. Respiration 2002; 69(3): 193–7
Hajek P, Stead LF. Aversive smoking for smoking cessation. Cochrane Database Syst Rev 2000; (2): CD000546
White AR, Rampes H, Ernst E. Acupuncture for smoking cessation. Cochrane Database Syst Rev 2002; (2): CD000009
Abbot NC, Stead LF, White AR, et al. Hypnotherapy for smoking cessation. Cochrane Database Syst Rev 2000; (2): CD001008
West R. Glucose for smoking cessation: does it have a role? CNS Drugs 2001; 15(4): 261–5
Disse B. Antimuscarinic treatment for lung diseases from research to clinical practice. Life Sci 2001; 68(22–23): 2557–64
Appleton S, Smith B, Veale A, et al. Long-acting beta2-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2000; (2): CD001104
Liesker JJ, Wijkstra PJ, Ten Hacken NH, et al. A systematic review of the effects of bronchodilators on exercise capacity in patients with COPD. Chest 2002; 121(2): 597–608
Jarvis B, Markham A. Inhaled salmeterol: a review of its efficacy in chronic obstructive pulmonary disease. Drugs Aging 2001; 18(6): 441–72
Lee AM, Jacoby DB, Fryer AD. Selective muscarinic receptor antagonists for airway diseases. Curr Opin Pharmacol 2001; 1(3): 223–9
van Noord JA, Smeets JJ, Custers FL, et al. Pharmacodynamic steady state of tiotropium in patients with chronic obstructive pulmonary disease. Eur Respir J 2002; 19(4): 639–44
Rees PJ. Tiotropium in the management of chronic obstructive pulmonary disease. Eur Respir J 2002; 19(2): 205–6
Littner MR, Ilowite JS, Tashkin DP, et al. Long-acting bronchodilation with once-daily dosing of tiotropium (Spiriva) in stable chronic obstructive pulmonary disease. Am J Crit Care Med 2000; 161: 1136–42
Vincken W, van Noord JA, Greefhorst AP, et al. Improved health outcomes in patients with COPD during 1 years treatment with tiotropium. Eur Respir J 2002; 19(2): 209–16
Casaburi R, Mahler DA, Jones PW, et al. A long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary disease. Eur Respir J 2002; 19(2): 217–24
Pineda R, Rennard SI. Anticholinergic bronchodilators in combination. Expert Opin Pharmacother 2000; 1(7): 1281–7
Manning HL. Bronchodilator therapy in chronic obstructive pulmonary disease. Curr Opin Pulm Med 2000; 6(2): 99–103
D’Urzo AD, De Salvo MC, Ramirez-Rivera A, et al. In patients with COPD, treatment with a combination of formoterol and ipratropium is more effective than a combination of salbutamol and ipratropium: a 3-week, randomized, double-blind, within-patient, multicenter study. Chest 2001; 119(5): 1347–56
ZuWallack RL, Mahler DA, Reilly D, et al. Salmeterol plus theophylline combination therapy in the treatment of COPD. Chest 2001; 119(6): 1661–70
Cazzola M, Di Lorenzo G, Di Perna F, et al. Additive effects of salmeterol and fluticasone or theophylline in COPD. Chest 2000; 118(6): 1576–81
Barnes PJ. Scientific rationale for inhaled combination therapy with long-acting beta2-agonists and corticosteroids. Eur Respir J 2002; 19(1): 182–91
Chapman KR. Seretide for obstructive lung disease. Expert Opin Pharmacother 2002; 3(3): 341–50
Mahler DA, Wire P, Hortsman D, et al. Effectiveness of fluticasone proprionate and salmeterol combination delivered via the Diskus device in the treatment of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2002; 166(8): 10–91
Szafranski W, Cukier A, Ramirez A, et al. Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease. Eur Respir J 2003; 21(1): 74–81
Newbold P, Jackson DM, Young A, et al. Dual D2 dopamine receptor and β2-adrenoceptor agonists for the modulation of sensory nerves in COPD. In: Hansel TT, Barnes PJ, editors. New Drugs for Asthma, Allergy and COPD. Basel: Karger, 2001: 68–71
Rogers DF. Tachykinin receptor antagonists for asthma and COPD. Expert Opin Ther Patents 2001; 11: 1097–121
Joos GF, Pauwels RA. Tachykinin receptor antagonists: potential in airways diseases. Curr Opin Pharmacol 2001; 1(3): 235–41
Rogers DF. Pharmacological regulation of the neuronal control of airway mucus secretion. Curr Opin Pharmacol 2002; 2(3): 249–55
Spina D. Airway nerves: neurotransmitter release. Curr Opin Pharmacol 2002; 2(3): 283–5
Janssens JP, de Muralt B, Titelion V. Management of dyspnea in severe chronic obstructive pulmonary disease. J Pain Symptom Manage 2000; 19(5): 378–92
Shapiro SD. Neutrophil elastase: path clearer, pathogen killer, or just pathologic? Am J Respir Cell Mol Biol 2002; 26(3): 266–8
Carrell RW, Lomas DA. Alphal-antitrypsin deficiency: a model for conformational diseases. N Engl J Med 2002; 346(1): 45–53
Stockley RA, Bayley DL, Unsal I, et al. The effect of augmentation therapy on bronchial inflammation in alphal-antitrypsin deficiency. Am J Respir Crit Care Med 2002; 165(11): 1494–8
Ohbayashi H. Neutrophil elastase inhibitors as treatment for COPD. Expert Opin Investig Drugs 2002; 11(7): 965–80
Kawabata K, Hagio T, Matsumoto S, et al. Delayed neutrophil elastase inhibition prevents subsequent progression of acute lung injury induced by endotoxin inhalation in hamsters. Am J Respir Crit Care Med 2000; 161(6): 2013–8
ZeiherBG, Matsuoka S, Kawabata K, et al. Neutrophil elastase and acute lung injury: prospects for sivelestat and other neutrophil elastase inhibitors as therapeutics. Crit Care Med 2002; 30 (5 Suppl.): S281–7
Reid PT, Sallenave JM. Neutrophil-derived elastases and their inhibitors: potential role in the pathogenesis of lung disease. Curr Opin Investig Drugs 2001; 2(1): 59–67
Sano C, Shimizu T, Sato K, et al. Effects of secretory leucocyte protease inhibitor on the production of the anti-inflammatory cytokines, IL-10 and transforming growth factor-beta (TGF-beta), by lipopolysaccharide-stimulated macrophages. Clin Exp Immunol 2000; 121(1): 77–85
Tremblay GM, Vachon E, Larouche C, et al. Inhibition of human neutrophil elastase-induced acute lung injury in hamsters by recombinant human pre-elafin (trappin-2). Chest 2002; 121(2): 582–8
Delacourt C, Herigault S, Delclaux C, et al. Protection against acute lung injury by intravenous or intratracheal pretreatment with EPI-HNE-4, a new potent neutrophil elastase inhibitor. Am J Respir Cell Mol Biol 2002; 26(3): 290–7
Sallenave JM. Antimicrobial activity of antiproteinases. Biochem Soc Trans 2002; 30(2): 111–5
Imai K, Dalai SS, Chen ES, et al. Human collagenase (matrix metalloproteinase-1) expression in the lungs of patients with emphysema. Am J Respir Crit Care Med 2001; 163 (3 Pt 1): 786–91
Russell RE, Culpitt SV, DeMatos C, et al. Release and activity of matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 by alveolar macrophages from patients with chronic obstructive pulmonary disease. Am J Respir Cell Mol Biol 2002; 26(5): 602–9
Brown PD. Ongoing trials with matrix metalloproteinase inhibitors. Expert Opin Investig Drugs 2000; 9(9): 2167–77
Katori H, Baba Y, Imagawa Y, et al. Reduction of in vivo tumor growth by MMI-166, a selective matrix metalloproteinase inhibitor, through inhibition of tumor angiogenesis in squamous cell carcinoma cell lines of head and neck. Cancer Lett 2002; 178(2): 151–9
Muraishi Y, Mitani N, Fuse H, et al. Effect of a matrix metalloproteinase inhibitor (ONO-4817) on lung metastasis of murine renal cell carcinoma. Anticancer Res 2001; 21(6A): 3845–52
Corbel M, Caulet-Maugendre S, Germain N, et al. Inhibition of bleomycin-induced pulmonary fibrosis in mice by the matrix metalloproteinase inhibitor batimastat. J Pathol 2001; 193(4): 538–45
Foda HD, Rollo EE, Drews M, et al. Ventilator-induced lung injury upregulates and activates gelatinases and EMMPRIN: attenuation by the synthetic matrix metalloproteinase inhibitor, Prinomastat (AG3340). Am J Respir Cell Mol Biol 2001; 25(6): 717–24
Churg A, Dai J, Zay K, et al. Alpha-1-antitrypsin and a broad spectrum metalloprotease inhibitor, RS113456, have similar acute anti-inflammatory effects. Lab Invest 2001; 81(8): 1119–31
Hele D. First Siena International Conference on animal models of chronic obstructive pulmonary disease; Siena; 2001 Sep 30–Oct 2. Respir Res 2002; 3(1): 12
Conway JG, Andrews RC, Beaudet B, et al. Inhibition of tumor necrosis factor-alpha (TNF-alpha) production and arthritis in the rat by GW3333, a dual inhibitor of TNF-alpha-converting enzyme and matrix metalloproteinases. J Pharmacol Exp Ther 2001; 298(3): 900–8
Stockley RA. Proteases and antiproteases. Novartis Found Symp 2001; 234: 189–99
Taggart CC, Lowe GJ, Greene CM, et al. Cathepsin B, L, and S cleave and inactivate secretory leucoprotease inhibitor. J Biol Chem 2001; 276(36): 33345–52
Van Der Pouw Kraan TC, Kucukaycan M, Bakker AM, et al. Chronic obstructive pulmonary disease is associated with the -1055 IL-13 promoter polymorphism. Genes Immun 2002; 3(7): 436–9
Zheng T, Zhu Z, Wang Z, et al. Inducible targeting of IL-13 to the adult lung causes matrix metalloproteinase- and cathepsin-dependent emphysema. J Clin Invest 2000; 106: 1081–93
Russell REK, Thorley A, Culpitt SV, et al. Alveolar macrophage-mediated elastolysis: roles of matrix metalloproteinases, cysteine and serine proteases. Am J Physiol 2002; 283: L867–73
Culpitt SV, Rogers DF, Shah P, et al. Impaired inhibition by dexamethasone of cytokine release by alveolar macrophages from COPD patients. Am J Respir Crit Care Med 2003; 167(1): 24–31
Stankova J, Turcotte S, Harris J, et al. Modulation of leuko-triene B4 receptor-1 expression by dexamethasone: potential mechanism for enhanced neutrophil survival. J Immunol 2002; 168(7): 3570–6
Adcock IM. Glucocorticoid-regulated transcription factors. Pulm Pharmacol Ther 2001; 14(3): 211–9
Ito K, Lim S, Caramori G, et al. Cigarette smoking reduces histone deacetylase 2 expression, enhances cytokine expression, and inhibits glucocorticoid actions in alveolar macrophages. FASEB J 2001; 15(6): 1110–2
Ito K, Lim S, Caramori G, et al. A molecular mechanism of action of theophylline: induction of histone deacetylase activity to decrease inflammatory gene expression. Proc Natl Acad Sci U S A 2002; 99(13): 8921–6
Culpitt SV, De Matos C, Russell RE, et al. Effect of theophylline on induced sputum inflammatory indices and neutrophil chemotaxis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2002; 165(10): 1371–6
Torphy TJ, Page C. Phosphodiesterases: the journey towards therapeutics. Trends Pharmacol Sci 2000; 21(5): 157–9
Sturton G, Fitzgerald M. Phosphodiesterase 4 inhibitors for the treatment of COPD. Chest 2002; 121 (5 Suppl.): 192S–6S
Giembycz MA. Cilomilast: a second generation phosphodiesterase 4 inhibitor for asthma and chronic obstructive pulmonary disease. Expert Opin Investig Drugs 2001; 10(7): 1361–79
Compton CH, Gubb J, Nieman R, et al. Cilomilast, a selective phosphodiesterase-4 inhibitor for treatment of patients with chronic obstructive pulmonary disease: a randomised, dose-ranging study. Lancet 2001; 358(9278): 265–70
Huang Z, Ducharme Y, MacDonald D, et al. The next generation of PDE4 inhibitors. Curr Opin Chem Biol 2001; 5(4): 432–8
Nakata A, Ogawa K, Sasaki T, et al. Potential role of phosphodiesterase 7 in human T cell function: comparative effects of two phosphodiesterase inhibitors. Clin Exp Immunol 2002; 128(3): 460–6
Sanjar S. Glaxo Wellcome research pipeline in chronic obstructive pulmonary disease. Eur Respir Rev 2000; 10: 370–3
Keeling SE, Albinson FD, Ayres BE, et al. The discovery and synthesis of highly potent, A2a receptor agonists. Bioorg Med Chem Lett 2000; 10(4): 403–6
Fozard JR, Ellis KM, Villela Dantas MF, et al. Effects of CGS 21680, a selective adenosine A2A receptor agonist, on allergic airways inflammation in the rat. Eur J Pharmacol 2002; 438(3): 183–8
de Boer WI. Cytokines and therapy in COPD: a promising combination? Chest 2002; 121 (5 Suppl.): 209S–18S
Wouters EF, Creutzberg EC, Schols AM. Systemic effects in COPD. Chest 2002; 121 (5 Suppl.): 127S–30S
Sandborn WJ, Targan SR. Biologic therapy of inflammatory bowel disease. Gastroenterology 2002; 122(6): 1592–608
Fleischmann R, Iqbal I, Nandeshwar P, et al. Safety and efficacy of disease-modifying anti-rheumatic agents: focus on the benefits and risks of etanercept. Drug Saf 2002; 25(3): 173–97
Rabinowitz MH, Andrews RC, Becherer JD, et al. Design of selective and soluble inhibitors of tumor necrosis factor-alpha converting enzyme (TACE). J Med Chem 2001; 44(24): 4252–67
Murphy PM, Baggiolini M, Charo IF, et al. International union of pharmacology, XXII: nomenclature for chemokine receptors. Pharmacol Rev 2000; 52(1): 145–76
Traves SL, Culpitt SV, Russell RE, et al. Increased levels of the chemokines GROalpha and MCP-1 in sputum samples from patients with COPD. Thorax 2002; 57(7): 590–5
Hay DW, Sarau HM. Interleukin-8 receptor antagonists in pulmonary diseases. Curr Opin Pharmacol 2001; 1(3): 242–7
Auten RL, Richardson RM, White JR, et al. Nonpeptide CXCR2 antagonist prevents neutrophil accumulation in hyperoxia-exposed newborn rats. J Pharmacol Exp Ther 2001; 299(1): 90–5
Cutshall NS, Ursino R, Kucera KA, et al. Nicotinamide N-oxides as CXCR2 antagonists. Bioorg Med Chem Lett 2001; 11(14): 1951–4
Cutshall NS, Kucera KA, Ursino R, et al. Nicotinanilides as inhibitors of neutrophil chemotaxis. Bioorg Med Chem Lett 2002; 12(11): 1517–20
Carson K, Glynn R, Ye Q, et al. Pyrazines/quinoxalines as IL-8 receptor antagonists. Book of Abstracts 220th American Chemical Society National Meeting; 2000 Aug 20–24; Washington, DC. Washington, DC: American Chemical Society, 2000
Li JJ, Yue WS, Trivedi BK, et al. Structure-activity relationship of 2-amino-3-heteroaryl-quinoxalines as potent, non-peptide interleukin-8-receptor antagonists. Book of Abstracts 220th American Chemical Society National Meeting; 2000 Aug 20–24; Washington, DC; Washington, DC: American Chemical Society, 2000
Witherington J, Bordas V, Cooper DG, et al. Conformationally restricted indolopiperidine derivatives as potent CCR2B receptor antagonists. Bioorg Med Chem Lett 2001; 11(16): 2177–80
de Boer WI, Sont JK, van Schadewijk A, et al. Monocyte chemoattractant protein 1, interleukin 8, and chronic airways inflammation in COPD. J Pathol 2000; 190(5): 619–26
Mirzadegan T, Diehl F, Ebi B, et al. Identification of the binding site for a novel class of CCR2b chemokine receptor antagonists: binding to a common chemokine receptor motif within the helical bundle. J Biol Chem 2000; 275(33): 25562–71
Traynor TR, Herring AC, Dorf ME, et al. Differential roles of CC chemokine ligand 2/monocyte chemotactic protein-1 and CCR2 in the development of T1 immunity. J Immunol 2002; 168(9): 4659–66
Torphy TJ, Li L, Griswold DE. Monoclonal antibodies as a strategy for pulmonary diseases. Curr Opin Pharmacol 2001; 1(3): 265–71
Saetta M, Mariani M, Panina-Bordignon P, et al. Increased expression of the chemokine receptor CXCR3 and its ligand CXCL10 in peripheral airways of smokers with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2002; 165(10): 1404–9
Mohan K, Ding Z, Hanly J, et al. IFN-gamma-inducible T Cell alpha chemoattractant is a potent stimulator of normal human blood T lymphocyte transendothelial migration: differential regulation by IFN-gamma and TNF-alpha. J Immunol 2002; 168(12): 6420–8
Ogasawara K, Hida S, Weng Y, et al. Requirement of the IFN-alpha/beta-induced CXCR3 chemokine signalling for CD8+ T cell activation. Genes Cells 2002; 7(3): 309–20
Proudfoot AE. Chemokine receptors: multifaceted therapeutic targets. Nature Rev Immunol 2002; 2(2): 106–15
Chung KF. Anti-inflammatory cytokines in asthma and allergy: interleukin-10, interleukin-12, interferon-gamma. Mediators Inflamm 2001; 10(2): 51–9
Lim S, Roche N, Oliver BG, et al. Balance of matrix metal-loprotease-9 and tissue inhibitor of metalloprotease-1 from alveolar macrophages in cigarette smokers: regulation by interleukin-10. Am J Respir Crit Care Med 2000; 162 (4 Pt 1): 1355–60
Barnes PJ. New treatments for chronic obstructive pulmonary disease. Curr Opin Pharmacol 2001; 1(3): 217–22
Fedorak RN, Gangl A, Elson CO, et al. Recombinant human interleukin 10 in the treatment of patients with mild to moderately active Crohn’s disease: the Interleukin 10 Inflammatory Bowel Disease Cooperative Study Group. Gastroenterology 2000; 119(6): 1473–82
Kilfeather S. 5-lipoxygenase inhibitors for the treatment of COPD. Chest 2002; 121 (5 Suppl.): 197S–200S
Yokomizo T, Izumi T, Shimizu T. Leukotriene B4: metabolism and signal transduction. Arch Biochem Biophys 2001; 385(2): 231–41
Silbaugh SA, Stengel PW, Cockerham SL, et al. Pharmacologic actions of the second generation leukotriene B4 receptor antagonist LY29311: in vivo pulmonary studies. Naunyn Schmiedebergs Arch Pharmacol 2000; 361(4): 397–404
Crooks SW, Bayley DL, Hill SL, et al. Bronchial inflammation in acute bacterial exacerbations of chronic bronchitis: the role of leukotriene B4. Eur Respir J 2000; 15(2): 274–80
Beeh KM, Kornmann O, Buhl R, et al. Neutrophil chemotactic activity of sputum from patients with COPD: role of interleukin-8 and leukotriene B4. Chest 2003; 123(4): 1240–7
Janssen LJ. Isoprostanes: an overview and putative roles in pulmonary pathophysiology. Am J Physiol Lung Cell Mol Physiol 2001; 280(6): L1067–82
Christie PE, Henderson Jr WR. Lipid inflammatory mediators: leukotrienes, prostaglandins, platelet-activating factor. Clin Allergy Immunol 2002; 16: 233–54
Taha R, Olivenstein R, Utsumi T, et al. Prostaglandin H synthase 2 expression in airway cells from patients with asthma and chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2000; 161 (2 Pt 1): 636–40
Sinatra R. Role of COX-2 inhibitors in the evolution of acute pain management. J Pain Symptom Manage 2002; 24 (1 Suppl.): S18–27
Romano SJ, Slee DH. Targeting selectins for the treatment of respiratory diseases. Curr Opin Investig Drugs 2001; 2(7): 907–13
Davenpeck KL, Berens KL, Dixon RA, et al. Inhibition of adhesion of human neutrophils and eosinophils to P-selectin by the sialyl Lewis antagonist TBC1269: preferential activity against neutrophil adhesion in vitro. J Allergy Clin Immunol 2000; 105(4): 769–75
Noguera A, Batle S, Miralles C, et al. Enhanced neutrophil response in chronic obstructive pulmonary disease. Thorax 2001; 56(6): 432–7
Ghosh S, Karin M. Missing pieces in the NF-kappaB puzzle. Cell 2002; 109 Suppl.: S81–96
Teramoto S, Kume H. The role of nuclear factor-kappa B activation in airway inflammation following adenovirus infection and COPD. Chest 2001; 119(4): 1294–5
Gilmour PS, Rahman I, Hayashi S, et al. Adenoviral E1A primes alveolar epithelial cells to PM(10)-induced transcription of interleukin-8. Am J Physiol Lung Cell Mol Physiol 2001; 281(3): L598–606
Delhase M, Li N, Karin M. Kinase regulation in inflammatory response. Nature 2000; 406(6794): 367–8
Ojo-Amaize EA, Kapahi P, Kakkanaiah VN, et al. Hypoestoxide, a novel anti-inflammatory natural diterpene, inhibits the activity of Ikappa B kinase. Cell Immunol 2001; 209(2): 149–57
Yang F, Oz HS, Barve S, et al. The green tea polyphenol (−)-epigallocatechin-3-gallate blocks nuclear factor-kappa B activation by inhibiting I kappa B kinase activity in the intestinal epithelial cell line IEC-6. Mol Pharmacol 2001; 60(3): 528–33
Gill JS, Zhu X, Moore MJ, et al. Effects of NFkappaB decoy oligonucleotides released from biodegradable polymer microparticles on a glioblastoma cell line. Biomaterials 2002; 23(13): 2773–81
Montuschi P, Kharitonov SA, Barnes PJ. Exhaled carbon monoxide and nitric oxide in COPD. Chest 2001; 120(2): 496–501
Ichinose M, Sugiura H, Yamagata S, et al. Increase in reactive nitrogen species production in chronic obstructive pulmonary disease airways. Am J Respir Crit Care Med 2000; 162 (2 Pt 1): 701–6
Corradi M, Montuschi P, Donnelly LE, et al. Increased nitrosothiols in exhaled breath condensate in inflammatory airway diseases. Am J Respir Crit Care Med 2001; 163(4): 854–8
Evans SM, Whittle BJ. Interactive roles of Superoxide and inducible nitric oxide synthase in rat intestinal injury provoked by non-steroidal anti-inflammatory drugs. Eur J Pharmacol 2001; 429(1–3): 287–96
Beaton H, Boughton-Smith N, Hamley P, et al. Thienopyridines: nitric oxide synthase inhibitors with potent in vivo activity. Bioorg Med Chem Lett 2001; 11(8): 1027–30
Hallinan EA, Tsymbalov S, Dorn CR, et al. Synthesis and biological characterization of L-N(6)-(l-iminoethyl)lysine 5-tetrazole-amide, a prodrug of a selective iNOS inhibitor. J Med Chem 2002; 45(8): 1686–9
Erin EM, Hansel TT, Kharitonov SA, et al. A selective inhibitor of inducible nitric oxide synthase inhibits exhaled breath nitric oxide in asthma. Am J Respir Crit Care Med 2002; 165 Suppl.: A187
Kyriakis JM, Avruch J. Mammalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev 2001; 81(2): 807–69
Liu R, O’Connell M, Johnson K, et al. Extracellular signal-regulated kinase 1/extracellular signal-regulated kinase 2 mitogen-activated protein kinase signaling and activation of activator protein 1 and nuclear factor kappaB transcription factors play central roles in interleukin-8 expression stimulated by monosodium urate monohydrate and calcium pyrophosphate crystals in monocytic cells. Arthritis Rheum 2000; 43(5): 1145–55
DiCamillo SJ, Carreras I, Panchenko MV, et al. Elastase-released epidermal growth factor recruits epidermal growth factor receptor and extracellular signal-regulated kinases to down-regulate tropoelastin mRNA in lung fibroblasts. J Biol Chem 2002; 277(21): 18938–46
Han Z, Chang L, Yamanishi Y, et al. Joint damage and inflammation in c-Jun N-terminal kinase 2 knockout mice with passive murine collagen-induced arthritis. Arthritis Rheum 2002; 46(3): 818–23
Weston CR, Davis RJ. The JNK signal transduction pathway. Curr Opin Genet Dev 2002; 12(1): 14–21
Bennett BL, Sasaki DT, Murray BW, et al. SP600125, an anthrapyrazolone inhibitor of Jun N-terminal kinase. Proc Natl Acad Sci U S A 2001; 98(24): 13681–6
Han Z, Boyle DL, Chang L, et al. c-Jun N-terminal kinase is required for metalloproteinase expression and joint destruction in inflammatory arthritis. J Clin Invest 2001; 108(1): 73–81
Lee JC, Kumar S, Griswold DE, et al. Inhibition of p38 MAP kinase as a therapeutic strategy. Immunopharmacology 2000; 47(2–3): 185–201
Chan ED, Riches DW. IFN-gamma + LPS induction of iNOS is modulated by ERK, JNK/SAPK, and p38(mapk) in a mouse macrophage cell line. Am J Physiol Cell Physiol 2001; 280(3): C441–50
Underwood DC, Osborn RR, Bochnowicz S, et al. SB 239063, a p38 MAPK inhibitor, reduces neutrophilia, inflammatory cytokines, MMP-9, and fibrosis in lung. Am J Physiol Lung Cell Mol Physiol 2000; 279(5): L895–902
Fijen JW, Zijlstra JG, De Boer P, et al. Suppression of the clinical and cytokine response to endotoxin by RWJ-67657, a p38 mitogen-activated protein-kinase inhibitor, in healthy human volunteers. Clin Exp Immunol 2001; 124(1): 16–20
Branger J, van den BB, Weijer S, et al. Anti-inflammatory effects of a p38 mitogen-activated protein kinase inhibitor during human endotoxemia. J Immunol 2002; 168(8): 4070–7
Cantley LC. The phosphoinositide 3-kinase pathway. Science 2002; 296(5573): 1655–7
Yoon SO, Park SJ, Yoon SY, et al. Sustained production of H2O2 activates proMMP-2 through receptor tyrosine kinases/ PI3-K/NF-kB pathway. J Biol Chem 2002; 277(33): 30271–82
Sasaki T, Irie-Sasaki J, Jones RG, et al. Function of PI3Kgamma in thymocyte development, T cell activation, and neutrophil migration. Science 2000; 287(5455): 1040–6
McGowan SE. Contributions of retinoids to the generation and repair of the pulmonary alveolus. Chest 2002; 121 (5 Suppl.): 206S–8S
Massaro D, Massaro GD. Pulmonary alveolus formation: critical period, retinoid regulation and plasticity. Novartis Found Symp 2001; 234: 229–36
Frankenberger M, Hauck RW, Frankenberger B,et al. All transretinoic acid selectively down-regulates matrix metal-loproteinase-9 (MMP-9) and up-regulates tissue inhibitor of metalloproteinase-1 (TIMP-1) in human bronchoalveolar lavage cells. Mol Med 2001; 7(4): 263–70
Mao JT, Goldin JG, Demand J, et al. A pilot study of all-trans-retinoic acid for the treatment of human emphysema. Am J Respir Crit Care Med 2002; 165(5): 718–23
MacNee W. Oxidative stress and lung inflammation in airways disease. Eur J Pharmacol 2001; 429(1–3): 195–207
Kiefer P, Vogt J, Radermacher P. From mucolytic to antioxidant and liver protection: new aspects in the intensive care unit career of N-acetylcysteine. Crit Care Med 2000; 28(12): 3935–6
Poole PJ, Black PN. Oral mucolytic drugs for exacerbations of chronic obstructive pulmonary disease: systematic review. BMJ 2001; 322(7297): 1271–4
Kasielski M, Nowak D. Long-term administration of N-acetylcysteine decreases hydrogen peroxide exhalation in subjects with chronic obstructive pulmonary disease. Respir Med 2001; 95(6): 448–56
Decramer M, Dekhuijzen PN, Troosters T, et al. The Bronchitis Randomized On NAC Cost-Utility Study (BRONCUS): hypothesis and design. BRONCUS-trial Committee. Eur Respir J 2001; 17(3): 329–36
Epperly MW, Defilippi S, Sikora C, et al. Radioprotection of lung and esophagus by overexpression of the human manganese Superoxide dismutase transgene. Mil Med 2002; 167 (2 Suppl.): 71–3
Salvemini D, Riley DP. Nonpeptidyl mimetics of Superoxide dismutase in clinical therapies for diseases. Cell Mol Life Sci 2000; 57(11): 1489–92
Salvemini D, Mazzon E, Dugo L, et al. Pharmacological manipulation of the inflammatory cascade by the Superoxide dismutase mimetic, M40403. Br J Pharmacol 2001; 132(4): 815–27
Oury TD, Thakker K, Menache M, et al. Attenuation of bleomycin-induced pulmonary fibrosis by a catalytic antioxi-dant metalloporphyrin. Am J Respir Cell Mol Biol 2001; 25(2): 164–9
Piganelli JD, Flores SC, Cruz C, et al. A metalloporphyrin-based Superoxide dismutase mimic inhibits adoptive transfer of autoimmune diabetes by a diabetogenic T-cell clone. Diabetes 2002; 51(2): 347–55
Saito K, Yoshioka H. Protective effect of spin trap agent, N-tert-butyl-alpha-phenylnitrone on hyperoxia-induced oxidative stress and its potential as a nitric oxide donor. Free Radic Res 2002; 36(2): 143–9
Lauderback CM, Breier AM, Hackett J, et al. The pyrrolopyrimidine U101033E is a potent free radical scavenger and prevents Fe(II)-induced lipid peroxidation in synaptosomal membranes. Biochim Biophys Acta 2000; 1501(2–3): 149–61
Rogers DF. Mucus pathophysiology in COPD: differences to asthma, and pharmacotherapy. Monaldi Arch Chest Dis 2000; 55(4): 324–32
Rogers DF. Mucoactive drugs for asthma and COPD: any place in therapy? Expert Opin Investig Drugs 2002; 11(1): 15–35
Grandjean EM, Berthet P, Ruffmann R, et al. Efficacy of oral long-term N-acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther 2000; 22(2): 209–21
Grandjean EM, Berthet PH, Ruffmann R, et al. Cost-effectiveness analysis of oral N-acetylcysteine as a preventive treatment in chronic bronchitis. Pharmacol Res 2000; 42(1): 39–50
Kellerman DJ. P2Y(2) receptor agonists: a new class of medication targeted at improved mucociliary clearance. Chest 2002; 121 (5 Suppl.): 201S–5S
Pendergast W, Yerxa BR, Douglass III JG, et al. Synthesis and P2Y receptor activity of a series of uridine dinucleoside 5′-polyphosphates. Bioorg Med Chem Lett 2001; 11(2): 157–60
Jackson AD. Airway goblet-cell mucus secretion. Trends Pharmacol Sci 2001; 22(1): 39–45
Rogers DF. The airway goblet cell. Int J Biochem Cell Biol 2003; 35(1): 1–6
Aarbiou J, Rabe KF, Hiemstra PS. Role of defensins in inflammatory lung disease. Ann Med 2002; 34(2): 96–101
Matsushita I, Hasegawa K, Nakata K, et al. Genetic variants of human beta-defensin-1 and chronic obstructive pulmonary disease. Biochem Biophys Res Commun 2002; 291(1): 17–22
Aarbiou J, Ertmann M, van Wetering S, et al. Human neutrophil defensins induce lung epithelial cell proliferation in vitro. J Leukoc Biol 2002; 72(1): 167–74
Hiemstra PS, van Wetering S, Stolk J. Neutrophil serine proteinases and defensins in chronic obstructive pulmonary disease: effects on pulmonary epithelium. Eur Respir J 1998; 12(5): 1200–8
Ram FS, Brocklebank DM, Muers M, et al. Pressurised metered-dose inhalers versus all other hand-held inhalers devices to deliver bronchodilators for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2002; (1): CD002170
Brocklebank D, Ram F, Wright J, et al. Comparison of the effectiveness of inhaler devices in asthma and chronic obstructive airways disease: a systematic review of the literature. Health Technol Assess 2001; 5(26): 1–149
Acknowledgements
The authors have provided no information on sources of funding or on conflicts of interest directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Donnelly, L.E., Rogers, D.F. Therapy for Chronic Obstructive Pulmonary Disease in the 21st Century. Drugs 63, 1973–1998 (2003). https://doi.org/10.2165/00003495-200363190-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363190-00002