Abstract
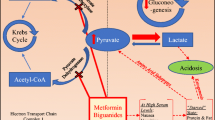
Lactic acidosis associated with metformin treatment is a rare but important adverse event, and unravelling the problem is critical. First, this potential event still influences treatment strategies in type 2 diabetes mellitus, particularly in the many patients at risk of kidney failure, in those presenting contraindications to metformin and in the elderly. Second, the relationship between metformin and lactic acidosis is complex, since use of the drug may be causal, co-responsible or coincidental. The present review is divided into three parts, dealing with the incidence, management and prevention of lactic acidosis occurring during metformin treatment. In terms of incidence, the objective of this article is to counter the conventional view of the link between metformin and lactic acidosis, according to which metformin-associated lactic acidosis is rare but is still associated with a high rate of mortality. In fact, the direct metformin-related mortality is close to zero and metformin may even be protective in cases of very severe lactic acidosis unrelated to the drug. Metformin has also inherited a negative class effect, since the early biguanide, phenformin, was associated with more frequent and sometimes fatal lactic acidosis. In the second part of this review, the objective is to identify the most efficient patient management methods based on our knowledge of how metformin acts on glucose/lactate metabolism and how lactic acidosis may occur (at the organ and cellular levels) during metformin treatment. The liver appears to be a key organ for both the antidiabetic effect of metformin and the development of lactic acidosis; the latter is attributed to mitochondrial impairment and subsequent adenosine triphosphate depletion, acceleration of the glycolytic flux, increased glucose uptake and the generation of lactate, which effluxes into the circulation rather than being oxidized further. Haemodialysis should systematically be performed in severe forms of lactic acidosis, since it provides both symptomatic and aetiological treatment (by eliminating lactate and metformin). In the third part of the review (prevention), the objective is to examine the list of contraindications to metformin (primarily related to renal and cardiovascular function). Diabetes is above all a vascular disease and metformin is a vascular drug with antidiabetic properties. Given the importance of the liver in lactate clearance, we suggest focusing on the severity of and prognosis for liver disease; renal dysfunction is only a prerequisite for metformin accumulation, which may only be dangerous per se when associated with liver failure. Lastly, in view of metformin’s impressive overall effectiveness profile, it would be paradoxical to deny the majority of patients with long-established diabetes access to metformin because of the high prevalence of contraindications. The implications of these contraindications are discussed.


Similar content being viewed by others
References
Chan N, Brain H, Feher M. Metformin-associated lactic acidosis: a rare or very rare clinical entity? Diabetic Med 1999 Apr; 16(4): 273–81
Misbin RI. The phantom of lactic acidosis due to metformin in patients with diabetes. Diabetes Care 2004 Jul; 27(7): 1791–3
Cohen R, Woods H. The clinical presentations and classifications of lactic acidosis. In: Cohen R, HF Woods, editors. Clinical and biochemical aspects of lactic acidosis. Boston (MA): Blackwell Scientific Publications, 1976: 40–52
Luft D, Deichsel G, Schmulling R, et al. Definition of clinically relevant lactic acidosis in patients with internal diseases. Am J Clin Pathol 1983 Oct; 80(4): 484–9
Arieff A. Pathogenesis of lactic acidosis. Diabetes Metab Rev 1989 Dec; 5(8): 637–49
Stacpoole P. Lactic acidosis. Endocrinol Metab Clin North Am 1993; 22: 221–45
Sterne J. Pharmacology and mode of action of hypoglycaemic guanidine derivatives. In: Campbell IW, editor. Oral hypoglycaemic agents. London: Academic Press, 1969: 193–245
Kreisberg R, Pennington L, Boshell B. Lactate turnover and gluconeogenesis in obesity: effect of phenformin. Diabetes 1970 Jan; 19(1): 64–9
Searle G, Siperstein M. Lactic acidosis associated with phenformin therapy: evidence that inhibited lactate oxidation is the causative factor. Diabetes 1975 Aug; 24(8): 741–5
Natrass M, Todd P, Hinks L, et al. Comparative effects of phenformin, metformin and glibenclamide in metabolic rhythms in maturity-onset diabetes. Diabetologia 1977 Apr; 13(2): 145–52
Oates N, Shah R, Idle J, et al. Genetic polymorphism of phenformin 4-hydroxylation. Clin Pharmacol Ther 1982 Jul; 32(1): 81–9
Williams R, Palmer J. Farewell to phenformin for treating diabetes mellitus. Ann Intern Med 1975 Oct; 83(4): 567–8
Cryer DR, Mills DJ, Nicholas SP, et al. Comparative outcomes study of metformin intervention versus conventional approach. Diabetes Care 2005 Mar; 28(3): 539–43
Salpeter SR, Geryber E, Pasternak GA, et al. Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev 2010 Jan; (1): CD002967
Bolen S, Feldman L, Vassy J, et al. Systematic review: comparative effectiveness and safety of oral medications for type 2 diabetes mellitus. Ann Intern Med 2007 Sept; 147(6): 386–99
Bodmer M, Jick SS, Meier C, et al. Metformin, sulfonylureas, or other antidiabetes drugs and the risk of lactic acidosis or hypoglycaemia. Diabetes Care 2008 Nov; 31(11): 2086–91
Brown JB, Pedula K, Barzilay J, et al. Lactic acidosis rates in type 2 diabetes. Diabetes Care 1998 Oct; 21(10): 1659–63
Fulop M, Hoberman H. Phenformin-associated metabolic acidosis. Diabetes 1976 Apr; 25(4): 292–6
Lalau J, Race J. Lactic acidosis in metformin therapy: searching for a link with metformin in reports of ‘metformin-associated lactic acidosis’. Diabetes Obes Metab 2001 Jun; 3(3): 195–201
Lalau J, Lacroix C, Compagnon P, et al. Role of metformin accumulation in metformin-associated lactic acidosis. Diabetes Care 1995 June; 18(6): 779–84
Lalau J, Race J, Brinquin L. Lactic acidosis in metformin therapy: relationship between plasma metformin concentration and renal function [letter]. Diabetes Care 1998 Aug; 21(8): 1366–7
Lalau J, Race J. Lactic acidosis in metformin-treated patients: prognostic value of arterial lactate levels and plasma metformin concentrations. Drug Saf 1999 Apr; 20(4): 377–84
Lalau J, Race J. Metformin and lactic acidosis in diabetic humans. Diabetes Obes Metab 2000 Jun; 2(3): 131–7
Lacroix C, Danger P, Wojciechowski F. Microassay of plasma and erythrocyte metformin by high performance liquid chromatography [in French]. Ann Biol Clin (Paris) 1991; 49(2): 98–101
Ahmad S, Beckett M. Recovery from pH 6.38: lactic acidosis complicated by hypothermia. Emerg Med 2002 Mar; 19(2): 169–71
Lalau J, Lacroix C. Measurement of metformin concentration in erythrocytes: clinical implications. Diabetes Obes Metab 2003 Mar; 5(2): 92–8
Robert F, Fendri S, Hary L, et al. Kinetics of plasma and erythrocyte metformin after acute administration in healthy subjects. Diabetes Metab 2003 Jun; 29(3): 279–83
Stades AME, Heikens JT, Erkelens DW, et al. Metformin and lactic acidosis: cause or coincidence? A review of case reports. J Intern Med 2004 Feb; 255(2): 179–87
Leverve X. Lactic acidosis: a new insight? Minerva Anestesiol 1999 May; 65(5): 205–9
Maran A, Cranston I, Lomas J, et al. Protection by lactate of cerebral function during hypoglycemia. Lancet 1994 Jan; I: 16–20
Vincent J. Lactate levels in critically ill patients. Acta Anaesthesiol Scand 1995; 39 Suppl. 107: 261–6
Veneman T, Mitrakou A, Mokan M, et al. Effect of hyperketonemia and hyperlacticacidemia on symptoms, cognitive dysfunction, and counterregulatory hormone responses during hypoglycemia in normal humans. Diabetes 1994 Nov; 43(11): 1311–7
King P, Parkin H, McDonald IAB, et al. The effect of intravenous lactate on cerebral function during hypoglycemia. Diabet Med 1997 Jan; 14(1): 19–28
King P, Kong M, Parkin H, et al. Intravenous lactate prevents cerebral dysfunction during hypoglycemia in insulin-dependent diabetes mellitus. Clin Sci 1998 Feb; 94(2): 157–63
Cusi K, Consoli A, DeFronzo R. Metabolic effects of metformin on glucose and lactate in non insulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1996 Nov; 96(11): 4059–67
Wiernsperger N, Bayley C. The antihyperglycaemic effect of metformin: therapeutic and cellular mechanisms. Drugs 1999; 58 Suppl. 1: 31–9
Wilcock C, Bayley C. Sites of metformin-stimulated metabolism. Biochem Pharmacol 1990 Jun; 39(11): 1831–4
Bailey C, Wilcock C, Day C. Effect of metformin on glucose metabolism in the splanchnic bed. Br J Pharmacol 1992 Apr; 105(4): 1009–15
Radziuk J, Zhang Z, Wiernsperger N, et al. Effects of metformin on lactate uptake and gluconeogenesis in the perfused rat liver. Diabetes 1997 Sep; 46(4): 1406–13
Leverve X, Guigas B, Detaille D, et al. Mitochondrial metabolism and type-2 diabetes: a specific target of metformin. Diabetes Metab 2003 Sep; 29 (4 Pt 2): 6S88–94
Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J 2000 Jun; 348 (Pt 3): 607–14
Zhou G, Myers R, Li Y, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 2001 Oct; 108(8): 1167–74
Foretz M, Leclerc J, Hebrard S, Viollet B. Metformin inhibits hepatic gluconeogenesis through an AMPK-independent mechanism [abstract no. 1507]. 68th Scientific Sessions of the American Diabetic Association; 2008 Jun 6–10; San Francisco (CA), A423
Wang DS, Jonker JW, Kato Y, et al. Involvement of organic cation transporter 1 in the hepatic and intestinal distribution of metformin. J Pharmacol Exp Ther 2002; 63(4): 844–8
Shu Y, Sheardown SA, Brown C, et al. Effect of genetic variation in the organic cation transporter 1 (OCT1) on metformin action. J Clin Invest 2007 Feb; 117(2): 1422–31
Wang DS, Kusuhara H, Kato Y, et al. Involvement of organic cation transporter 1 in the lactic acidosis caused by metformin. Mol Pharmacol 2003 Apr; 63(4): 844–8
Dykens JA, Jamieson J, Marroquin L, et al. Biguanide-induced mitochondrial dysfunction yields increased lactate production and cytotoxicity of aerobically-poised HepG2 cells and human hepatocytes in vitro. Toxicol Appl Pharmacol 2008; 233: 203–10
Wilcock C, Wyre N, Bailey C. Subcellular distribution of metformin in rat liver. J Pharm Pharmacol 1991 Jun; 43(6): 442–4
Wilcock C, Bayley C. Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica 1994 Jan; 24(1): 49–57
Lalau J, Andrejak M, Morinière P, et al. Hemodialysis in the treatment of lactic acidosis in diabetics treated by metformin: a study of metformin elimination. Int J Clin Pharmacol Ther Toxicol 1989 Jun; 24(6): 683–93
Lalau J, Masmoudi K. Unexpected recovery from prolonged hypoglycaemic coma: a protective role of metformin [letter]? Intens Care Med 2005 Mar; 31(3): 493
Assan R, Heuclin C, Ganeval D, et al. Metformin-induced lactic acidosis in the presence of acute renal failure. Diabetologia 1977 May; 13(3): 211–7
Lalau J, Mourlhon C, Bergeret A, et al. Consequences of metformin intoxication [letter]. Diabetes Care 1998 Nov; 21(11): 2036–7
European prescribing information for Glucophage®. Lyon: Merck Serono, 2005
Holstein A, Stumvoll M. Contraindications can damage your health: is metformin a case in point? Diabetologia 2005 Dec; 48(12): 2454–9
Fontana L. Modulating human aging and age-associated diseases. Biochim Biophys Acta 2009 Oct; 1790(10): 1133–8
Tahrani AA, Varughese GI, Scarpello JH, et al. Metformin, heart failure, and lactic acidosis: is metformin absolutely contraindicated? BMJ 2007 Sept; 335: 508–12
Gjedde S, Christiansen A, Pedersen S, et al. Survival following a metformin overdose of 63 g: a case report. Pharmacol Toxicol 2003 Aug; 93(2): 98–9
Knaus WA, Wagner DP, Draper EA, et al. The APACHE III prognostic system: risk prediction of hospital mortality for critically ill hospitalized adults. Chest 1991 Dec; 100(6): 1619–36
Nyirenda MJ, Sandeep T, Grant I, et al. Severe acidosis in patients taking metformin: rapid reversal and survival despite high APACHE score. Diabet Med 2006 Apr; 23(4): 432–5
Dell’Aglio D, Perino LJ, Kazzi Z, et al. Acute metformin overdose: examining serum pH, lactate level, and metformin concentrations in survivors versus nonsurvivors: a systematic review of the literature. Ann Emerg Med 2009 Dec; 54(6): 818–23
Gras V, Bouffandeau B, Montravers P, et al. Effect of metformin on survival rate in experimental sepsis. Diabetes Metab 2006 Apr; 32(2): 147–50
Bouskela E, Wiensperger N. Effects of metformin on hemorrhagic shock, blood volume and ischemia/reperfusion on nondiabetic hamsters. J Vasc Med Biol 1993; 4: 41–6
Wiernsperger N. 50 years later: is metformin a vascular drug with antidiabetic properties? Br J Vasc Dis 2007 Sept/Oct; 7(5): 204–10
Chan JCN, Davidons JA. Survival benefits of metformin in high-risk populations. In: Bailey CJ, Campbell IW, Chan JCN et al., editors. Metformin: the gold standard. Chichester: Wiley, 2007: 125–34
Scarpello JHB, Howlett HCS. Metformin therapy and clinical uses. Diabetes Vasc Dis Res 2008 Sept; 5(3): 157–67
Sirtori C, Franceschini G, Gianfranceschi G et al. Metformin improves peripheral vascular flow in non hyperlipidemic patients with arterial disease. J Cardiovasc Pharmacol 1984 Sep/Oct; 6(5): 914–23
Group UKPDS. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998 Sept; II: 854–65
Kao J, Tobis J, McClelland RL, et al. Relation of metformin treatment to clinical events in diabetic patients undergoing percutaneous intervention. Am J Cardiol 2004 Jun; 93(11): 1347–50
Sgambato S, Varrichio M, Tesauro P, et al. The use of metformin in ischemic cardiopathy. Clin Ther 1980 Jul; 94(1): 77–85
Eurich DT, Tsuyuki RT, Majundar SR, et al. Improved clinical outcome associated with metformin in patients with diabetes and heart failure. Diabetes Care 2005 Oct; 28(10): 2345–51
Masoudi FA, Inzucchi SE, Wang Y, et al. Thiazolidinediones, metformin, and outcomes in older patients with diabetes and heart failure: an observational study. Circulation 2005 Feb; 111(5): 583–90
Evans JMM, Ogston SA, Emslie-Smith A, et al. Risk of mortality and adverse cardiovascular outcomes in type 2 diabetes: a comparison of patients treated with sulfonylureas and metformin. Diabetologia 2006 May; 49(5): 930–6
Montanari G, Bondioli A, Rizzato G, et al. Treatment with low dose metformin in patients with peripheral vascular disease. Pharmacol Res 1992 Jan; 25(1): 63–73
Kakkar AK, Besterman WH, Lefer DJ. Preconditioning of the diabetic myocardium with acute metformin treatment. J Am Coll Cardiol 2004; 3: 1116–21
Weil M, Afifi A. Experimental and clinical studies on lactate and pyruvate as indicators of the severity of acute circulatory failure (shock). Circulation 1970 Jun; 41(6): 989–1001
Cady Jr L, Weil M, Afifi A, et al. Quantisation of severity of critical illness with special reference to blood lactate. Crit Care Med 1973 Mar/Apr; 1(2): 75–80
Batandier C, Guigas B, Detaille D, et al. The ROS production induced by a reverse-electron flux at respiratory complex 1 is hampered by metformin. J Bioenerg Biomembr 2006 Feb; 38(1): 33–42
Detaille D, Guigas B, Chauvin C, et al. Metformin prevents high-glucose-induced endothelial cell death through a mitochondrial permeability transition-dependent process. Diabetes 2005 Jul; 54(7): 2179–87
Arieff A, Gertz E, Park R, et al. Lactic acidosis and the cardiovascular system in the dog. Clin Sci 1983 Jun; 64(6): 573–80
Scheen A. Clinical pharmacokinetics of metformin. Clin Pharmacokinet 1996 May; 30(5): 359–71
Lalau J, Race J, Andreeli F, et al. Metformin retention independent of renal failure in intestinal occlusion. Diabetes Metab 2001 Feb; 27(1): 24–8
Levey AS, Bosch JP, Lewis JB, et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999 Mar; 130(6): 461–70
Warren RE, Strachan MWJ, Wild S, et al. Introducing estimated glomerular filtration rate (eGFR) into clinical practice in the UK: implications for the use of metformin. Diabet Med 2007 May; 24(5): 494–7
Shaw JS, Wilmot RL, Kilpatrick ES. Establishing pragmatic estimated GFR thresholds to guide metformin prescribing. Diabet Med 2007 Oct; 24(10): 1160–3
Lalau J, Vermersch A, Hary L, et al. Type 2 diabetes in the elderly: an assessment of metformin. Int J Clin Pharmacol Ther Toxicol 1990 Aug; 28(8): 329–32
Rachmani R, Slavachevski I, Levi Z, et al. Metformin in patients with type 2 diabetes mellitus: reconsideration of traditional contraindications. Eur J Intern Med 2002 Oct; 13(7): 428–33
Johnson JA, Majumdar SR, Simpson SH, et al. Decreased mortality associated with the use of metformin compared with sulfonylurea monotherapy in type 2 diabetes. Diabetes Care 2002 Dec; 25(12): 2244–8
Cohen R. Role of the liver and the kidney in acid-base regulation and its disorders. Br J Anesthesiol 1991 Aug; 67(2): 154–64
Seidowsky A, Nseir S, Houdret N, et al. Metformin-associated lactic acidosis: a prognosis and therapeutic study. Crit Care Med 2009 Jul; 37(7): 2191–6
Pugh RN, Murray-Lyon IM, Dawson JL, et al. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 1973 Aug; 60(8): 646–9
Emslie-Smith AM, Boyle DI, Evans JM, et al. Contraindications to metformin therapy in patients with type 2 diabetes: a population-based study of adherence to prescribing guidelines. Diabetic Med 2001 Jun; 18(6): 483–8
Holstein A, Nahrwold D, Hinze S, et al. Contraindications to metformin are largely discarded. Diabetic Med 1999 Aug; 16(8): 692–6
Jones GC, Macklin JP, Alexander WD. Contraindications to the use of metformin. Evidence suggests that it is time to amend the list. BMJ 2003 Apr; 326: 4–5
McCormack J, Johns K, Tildesley H. Metformin’s contraindications should be contraindicated. CAMJ 2005 Aug; 173(5): 502–4
Prikis M, Mesler EL, Hood VL, et al. When a friend can become an enemy! Recognition and management of metformin-associated lactic acidosis. Kidney Int 2007 Nov; 72(9): 1157–60
Golay A. Metformin and body weight. Int J Obes 2008 Jan; 32(1): 61–72
El-Mir MY, Detaille D, R-Villanueva G, et al. Neuroprotective role of antidiabetic drug metformin against apoptotic cell death in primary cortical neurons. J Mol Neurosci 2008; 34(1): 77–87
Libby G, Alessi DR, Donnelly LA, et al. New users of metformin are at low risk of incident cancer. Diabetes Care 2009 Sept; 32(9): 1620–5
Zhen D, Chen Y, Tang X. Metformin reverses the deleterious effects of high glucose on osteoblast function. J Diabetes Complications. Epub 2009 Jul 21
Amiel SA, Dixon T, Mann R, et al. Hypoglycaemia in type 2 diabetes. Diabet Med 2008 Mar; 25(3): 245–54
Mitri J, Hamdy O. Diabetes medications and body weight. Expert Opin Drug Saf 2009 Sep; 8(5): 573–84
Patel RR. Thiazolidinediones and congestive heart failure: a judicious balance of risks and benefits. Cardiol Rev 2009 May–Jun; 17(3): 132–5
Habib ZA, Havstad SL, Wells K, et al. Thiazolidinedione use and the longitudinal risk of fractures in patients with type 2 diabetes mellitus. J Clin Endocrinol Metab 2010 Feb; 95(2): 592–600
Monami M, Balzi D, Lamanna C, et al. Are sulphonylureas all the same? A cohort study on cardiovascular and cancer-related mortality. Diabetes Metab Res Rev 2007 Sep; 23(6): 479–84
Draznin B. Mitogenic action of insulin: friend, foe or ‘frenemy’? Diabetologia 2010 Feb; 53(2): 229–33
Acknowledgements
No funding was received for the preparation of this review. Jean-Daniel Lalau has been a compensated speaker for Boehringer-Ingelheim, Eli Lilly, Novartis, Novo Nordisk, Sanofi-aventis and Takeda.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lalau, JD. Lactic Acidosis Induced by Metformin. Drug-Safety 33, 727–740 (2010). https://doi.org/10.2165/11536790-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11536790-000000000-00000