Abstract
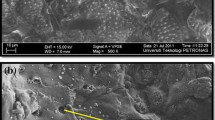
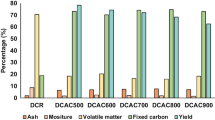
We studied the adsorption of nitrogen oxides according to various porous materials room temperature, which can effectively to adsorb the nitrogen oxides. Based on results of measuring NOx concentration, the activated carbon materials have been shown to adsorb NOx well, and they have been shown to more efficiently removing NO2, which is very harmful to the environment. In particular, in the case of the coconut activated carbon, the removal efficiency of NO and NO2 was very excellent, and the removal rate of was 30% for 100 minutes and 19.5% for 840 minutes. Our measurement results confirmed that the activated carbon materials have a very large specific surface area and pore volume. In addition, the coconut activated carbon appears to have a large specific surface area and pore volume to remove NOx over a long period of time. Based on the experimental results, it is expected to be used as a material that efficiently removes nitrogen oxides at room temperature.
Similar content being viewed by others
References
J. F. Currie, A. Essalik and J-C. Marusic, Sens. Actuators B 59, 235 (1999).
H. Ritchie and M. Roser, Our World in Data (2017), https://ourworldindata.org/co2-and-other-greenhouse-gas-emissions.
K. Gillingham and J. H. Stock, J. Econ. Perspect. 32, 53 (2018).
S. Sillman, Atmos. Environ. 33, 1821 (1999).
S-B. Lee et al., Aerosol Air Qual. Res. 10, 540 (2010).
H. J. Seo et al., Appl.Sci.Converg.Technol. 26, 2180 (2017).
O. I. Polivaev et al., IOP Conf. Ser.: Mater. Sci. Eng. 632, 012027 (2019).
C. Wang, Y. Liu, S. Zheng and A. Jiang, Energy 153, 149 (2018).
J. V. Caneghem, J. D. Greef, C. Block and C. Vandecasteele, J. Clean. Prod. 112, 4452 (2016).
H. Yu et al., Nanomaterials 10, 897 (2020).
M. Xu et al., Constr. Build. Mater. 221, 375 (2019).
O. I. Polivaevm et al., IOP Conf. Ser.: Mater. Sci. Eng. 632, 012027 (2019).
V. Valtchev and L. Tosheva, Chem. Rev. 113, 6734 (2013).
Y-C. Chiang, P-C. Chiang and C-P. Huang, Carbon 39, 523 (2001).
B. Kasprzyk-Hordern, Adv. Colloid Interface Sci. 110, 19 (2004).
J. Li et al., Catal. Today 175, 147 (2011).
S. Brunauer, P. H. Emmett and E. Teller, J. Am. Chem. Soc. 60, 309 (1938).
E. P. Barrett, L. G. Joyner and P. P. Halenda, J. Am. Chem. Soc. 73, 373 (1951).
K. Silas, W. A. W. A. K. Ghani, T. S. Y. Choong and U. Rashid, Catal. Rev. 61, 134 (2019).
A. M. Rubel and J. M. Stencel, Energy Fuels 10, 704 (1996).
Acknowledgments
This work was supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) and the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (No. 20181110200070).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lim, T., La, Y., Jeon, O.S. et al. Pore Structure Analysis to Adsorb NOx Gas based on Porous Materials. J. Korean Phys. Soc. 77, 790–796 (2020). https://doi.org/10.3938/jkps.77.790
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3938/jkps.77.790