Abstract
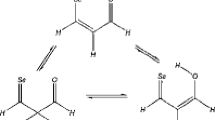
The anti- and syn-conformers of thiophene-2-carbonyl fluoride (A) and selenophene-2-carbonyl fluoride (B) have been studied in the gas phase. The transition states have also been obtained for the interconversion of the anti- and syn-conformers. The methods used are MP2 and DFT/B3LYP and the basis sets used for all atoms are 6-311++G(d,p). The optimized geometries, dipole moments, moment of inertia, energies, energy differences and rotational barriers are reported. This study has been extended to include solvent effect. Some of the vibrational frequencies of the conformers are reported with appropriate assignments. The results indicate that in the gas phase the syn conformer is more stable and the CCSD(T)//MP2 energy differences are 2.97 kJ/mol (A) and 3.02 kJ/mol (B) and barriers of rotation are 38.50 kJ/mol (A) and 36.89 kJ/mol (B). The structures and vibrational frequencies of (A) and (B) are not much affected by the solvents but the more polar conformer gets more stabilized. The major effect of the solvents is that energy difference decreases but rotational barrier increases. The peculiar characteristic of fluorine affecting conformational preference is not observed.
Chapter PDF
Similar content being viewed by others
Keywords
References
Han, I.-S., Kim, C.K., Jung, H.J., Lee, I.: Ab Initio Studies on the Rotational Equilibria of 2-Substituted Furan and Thiophene Carbonyl Derivatives. Theor. Chim. Acta. 93, 199–210 (1996)
Banks, J.W., Batsanov, A.S., Howard, J.A.K., O’Hagan, D., Rzepa, H.S., Martin-Santamaria, S.: The Preferred Conformation of α-Fluoroamides. J. Chem. Soc. Perkin Trans. 2, 2409–2411 (1999)
Abraham, R.J., Bretschneider, E., Orville-Thomas, W.J.: Internal Rotation in Molecules, ch. 13. Wiley, London
Ashish, H., Ramasami, P.: Rotational Barrier and Thermodynamical Parameters of Furfural, Thiofurfural, and Selenofurfural in the Gas and Solution Phases: Theoretical Study Based on Density Functional Theory Method. Mol. Phys. 106, 175–185 (2008)
Braathen, G.O., Kveseth, K., Nielsen, C.J.: Molecular Structure and Conformational Equilibrium of Gaseous Thiophene-2-aldehyde as Studied by Electron Diffraction and Microwave, Infrared, Raman and Matrix Isolation Spectroscopy. J. Mol. Struc. 145, 45–68 (1986)
Concistrè, M., Luca, G.D., Longeri, M., Pileio, G., Emsley, J.W.: The Structure and Conformations of 2-Thiophenecarboaldehyde Obtained from Partially Average Dipolar Couplings. Chem. Phys. Chem. 6, 1483–1491 (2005)
Fleming, G.D., Koch, R., Vallette, M.M.C.: Theoretical Study of the Syn and Anti Thiophene-2-aldehyde Conformers using Density Functional Theory and Normal Coordinate Analysis. Spectrochim. Acta A 65, 935–945 (2006)
Gronowitz, S.: Selenophene, a Twin-Brother of Thiophene? Phosphorus Sulfur 136, 59–90 (1998)
Salatelli, E., Zanirato, P.: The Conversion of Furan-, Thiophene- and Selenophene-2-carbonylazides into Isocyanates: A DSC Analysis. Arkivoc xi, 6–16 (2002)
Kim, H., Yoon, Y.-J., Kim, H., Cha, E.-Y., Lee, H.S., Kim, J.-H., Yi, K.Y., Lee, S., Cheon, H.G., Yoo, S.-E., Lee, S.-S., Shin, J.-G., Li, K.-H.: Vitro Metabolism of a New Cardioprotective Agent. KR-33028 in the Human Liver Microsomes and Cryopreserved Human Hepatocytes 28, 1287–1292 (2005)
Frisch, M.J., et al.: Gaussian 03, Revision B04. Gaussian Inc., Wallingford (2004)
Dennington II, R., Keith, T., Millam, J., Eppinnett, K., Hovell, W.L., Gilliland, R.: GaussView, Version 3.09. Semichem, Inc., Shawnee Mission (2003)
Mennucci, B., Tomasi, J.: Continuum Solvation Models: A New Approach to the Problem of Solute’s Charge Distribution and Cavity Boundaries. J. Chem. Phys. 106, 5151–5158 (1997)
Cancès, E., Mennucci, B., Tomasi, J.: A New Integral Equation Formalism for the Polarizable Continuum Model: Theoretical Background and Applications to Isotropic and Anisotropic Dielectrics. J. Chem. Phys. 107, 3032–3041 (1997)
Mennucci, B., Cancès, E., Tomasi, J.: Evaluation of Solvent Effects in Isotropic and Anisotropic Dielectrics and in Ionic Solutions with a Unified Integral Equation Method: Theoretical Bases, Computational Implementation, and Numerical Applications. J. Phys. Chem. B 101, 10506–10517 (1997)
Tomasi, J., Mennucci, B., Cancès, E.: The IEF Version of the PCM Solvation Method: An Overview of a New Method Addressed to Study Molecular Solutes at the QM Ab Initio Level. J. Mol. Struct. (Theochem.) 464, 211–226 (1999)
Ramasami, P.: Theoretical Study of 2-Selenophenecarboaldehyde in the Gas and Solution Phases: Rotational Barrier, Energy difference and Thermodynamics Parameters. Communicated
Abdallah, H.H., Ramasami, P.: Rotational Barrier, Energy difference and Thermodynamical Parameters of 2-Furoylfluoride, its Sulfur and Selenium Analogues: Theoretical Study in the Gas and Solution Phases. Communicated
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Abdallah, H.H., Ramasami, P. (2009). First Principle Study of the Anti– and Syn–Conformers of Thiophene–2–Carbonyl Fluoride and Selenophene–2–Carbonyl Fluoride in the Gas and Solution Phases. In: Allen, G., Nabrzyski, J., Seidel, E., van Albada, G.D., Dongarra, J., Sloot, P.M.A. (eds) Computational Science – ICCS 2009. ICCS 2009. Lecture Notes in Computer Science, vol 5545. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-01973-9_13
Download citation
DOI: https://doi.org/10.1007/978-3-642-01973-9_13
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-01972-2
Online ISBN: 978-3-642-01973-9
eBook Packages: Computer ScienceComputer Science (R0)