Abstract
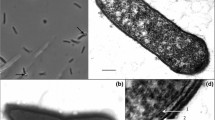
A new species of sporulating saccharolytic anaerobe, designated as Clostridium quinii sp. nov., is described. A gram-positive strain BS1, was isolated from the granular metanogenic sludge (UASB) from a waste-water treatment plant at a sugar refinery. The strain exhibits a series of morphological stages, developing from a spore to a small rod to a motile rod (peritrichous flagella) in the exponential growth phase, and then swelling to form cigar-shaped cells, exhibiting tumbling movements, in the late exponential growth phase before finally becoming large nonmotile ovoid cells in the stationary phase. Swelling occurs as a result of glucose being taken up and stored as a glycogen-like substance. The main fermentation products when growing on glucose is H2, CO2, formate, acetate and ethanol as well as small amounts of butyrate during exponential growth. Lactate is formed during the stationary phase, when glucose is abundant. Optimal conditions for growth is 40–45°C and pH of around 7.4. The type strain BS1 contains 28.0% mol G+C.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.
Similar content being viewed by others
References
Adamse AD (1980) New isolation of Clostridium aceticum (Wieringa)1. Antonie van Leeuwenhoek 46: 523–531
Adamse AD, Dienema MN, Zehnder AJB (1984) Studies on bacterial activities in aerobic and anacrobic waste water purification. Antonie van Leeuwenhoek 50: 665–682
Brough BE, Reid TC, Howard BH (1970) The biochemistry of the rumen bacterium “Quin's Oval” — Part 1. Fermentation of carbohydrates. N Z J Sci 13: 570–575
Buck JD (1982) Nonstaining (KOH) method for determination of gram reactions of marine bacteria. Appl Environ Microbiol 44: 992–993
Cato EP, George WL, Finegold SM (1986) Genus Clostridium. Prazmowski 1880, 23AL. In: Sneath PHA (ed) Bergey's manual of systematic bacteriology, vol. 2. William & Wilkins, Baltimore London, pp 1141–1200
Chung KT (1976) Inhibitory effect of H2 on growth of Clostridium cellobioparum. Appl Environ Microbiol 31: 342–348
Dolfing J, Bloemen WGBM (1985) Activity measurements as a tool to characterize the microbial composition of methanogenic environments. J Microbiol Methods 4: 1–12
Ferragut C, Leclerc H (1976) Etude comparative des méthodes de détermination du Tm de l'ADN bactérien. Ann Microbiol (Inst Pasteur) 127: 223–235
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW (eds) Methods in microbiology, vol 5B. Academic Press, London, pp 209–344
Hoffman A, Dimroth P (1990) The ATPase of Bacillus alcalophilus. Purification and properties of the enzyme. Eur J Biochem 194: 423–490
Holdeman LV, Cato EP, Moore WEC (1977) Anaerobic laboratory manual, 4th edn. Virginia Polytechnical Institute and State University, Blacksburg, Va., USA
Johnson JL (1981) Genetic characterization. In: Gerhardt P, Murry RGE, Costilow RN, Nester EW, Wood WA, Kodeg NR, Philips GB (eds) Manual of methods for general bacteriology. ASM, Washington DC, pp 450–472
Jones DT, Westhuizen A van der, Long S, Allcock ER, Ried SJ, Woods DR (1982) Solvent production and morphological changes in Clostridium acetobutylicum. Appl Environ Microbiol 43: 1434–1439
Kelly WJ, Asamudson RV, Hopcroft DH (1987) Isolation and characterization of a strictly anaerobic cellulolytic spore former: Clostridium chartatabidium sp. nov. Arch Microbiol 147: 169–173
Kim BH, Bellows P, Datta R, Zeikus JG (1984) Control of carbon and electron flow in Clostridium acetobutylicum fermentations. Utilization of carbon monoxide to inhibit hydrogen production and to enhance butanol yields. Appl Environ Microbiol 53: 644–650
LeRuyet P, Dubourgier H-C, Albagnac G, Prensier G (1985) Characterization of Clostridium thermolacticum sp. nov., a hydrolytic thermophilic anaerobe producing high amounts of lactate. Syst Appl Microbiol 6: 196–202
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Orpin CG (1972) The culture in vitro of the rumen bacterium Quin's Oval. J Gen Microbiol 73: 523–530
Scholten-Koerselman I, Houward F, Janssen P, Zehnder AJB (1986) Bacteriodes xylanolyticus sp. nov., a xylanolytic bacterium from methane producing cattle manure. Antoine van Leeuwenhoek 52: 543–554
Stouthamer AH (1973) A theoretical study on the amount of ATP required for synthesis of microbial cell material. Antonie van Leeuwenhoek 39: 545–565
Thiery JP (1967) Mise en évidence des polysaccharides sur coupe fine en microscopie électronique. J Microsc 67: 987–1018
Vicini JL, Brulla WJ, Davis CL, Bryant MP (1987) Quin's Oval and other microbiota in the rumens of molasses-fed sheep. Appl Environ Microbiol 53: 1273–1276
Wicken AJ, Howard BH (1967) On the taxonomic status of “Quin's Oval” organisms. J Gen Microbiol 47: 207–211
Wolin EA, Wolin MJ, Wolfe RS (1963) Formation of methane by bacterial extracts. J Biol Chem 238: 2882–2886
Zehnder AJB, Huser BA, Brock TD, Wuhrman K (1980) Characterization of an acetate-decarboxylating, non-hydrogen-oxidizing methane bacterium. Arch Microbiol 124: 1–11
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Svensson, B.H., Dubourguier, HC., Prensier, G. et al. Clostridium quinii sp. nov., a new saccharolytic anaerobic bacterium isolated from granular sludge. Arch. Microbiol. 157, 97–103 (1992). https://doi.org/10.1007/BF00245275
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00245275