Abstract
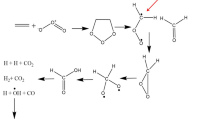
Calculations of a number of model structures within the scheme of the Fishcer indole synthesis were made on the basis of a bonding variant of perturbation theory in the self-consistent-field (SCF) MO LCAO method. A quantum-chemical interpretation of the effect of substituents on the course of the thermal process is given. The kinetics of the thermal and acid-catalyzed indolization of substituted cyclohexanone arylhydrazones to tetrahydrocarbazoles were studied by spectrophotometry. It was shown that the experimental data are in satisfactory agreement with the calculated values. It was concluded that a concerted mechanism (a [3,3]-sigmatropic shift) for the step involving the formation of a carbon-carbon bond in the Fischer reaction is preferred.
Similar content being viewed by others
Literature Cited
H. Robinson, Usp. Khim., 40, 1434 (1971).
Yu. P. Kitaev and T. V. Troepol'skaya, Khim. Geterotsikl. Soedin., No. 8, 1011 (1978).
I. I. Grandberg and V. I. Sorokin, Usp. Khim., 43, 266 (1974).
I. I. Grandberg, Zh. Org. Khim., 19, 2439 (1983).
B. Robinson, The Fischer Indole Synthesis, Wiley Interscience, New York (1982).
N. M. Przheval'skii, M. E. Kletskii, I. I. Grandberg, and L. Yu. Kostromina, Khim. Geterotsikl. Soedin., No. 6, 779 (1985).
H. Heimgartner, H. J. Hansen, and H. Schmid, in: Iminium Salts in Organic Chemistry, Wiley, New York (1979), p. 655.
A. W. Douglas, J. Am. Chem. Soc., 101, 5676 (1979).
A. W. Douglas, J. Am. Chem. Soc., 100, 6463 (1978).
G. L. Glish and R. G. Cooks, J. Am. Chem. Soc., 100, 6720 (1978).
N. M. Przheval'skii, L. Yu. Kostromina, and I. I. Grandberg, Khim. Geterotsikl. Soedin., No. 9, 1207 (1985).
M. M. Mestechkin, The Density-Matrix Method in Molecular Theory [in Russian], Naukova Dumka, Kiev (1977), p. 352.
Yu. B. Vysotskii and B. P. Zemskii, Khim. Geterotsikl. Soedin., No. 7, 984 (1980).
Yu. B. Vysotskii, B. P. Zemskii, T. V. Stupnikova, R. S. Sagitullin, A. N. Kost, and O. P. Shvaika, Khim. Geterotsikl. Soedin., No. 11, 1496 (1979).
Yu. B. Vysotskii, B. P. Zemskii, T. V. Stupnikova, V. N. Kalafat, R. S. Sagitullin, and V. P. Marshtupa, Khim. Geterotsikl. Soedin., No. 9, 1277 (1982).
M. V. Bazilevskii, The Molecular Orbital Method and the Reactivities of Organic Molecules [in Russian], Khimiya, Moscow (1969), p. 304.
S. Jolidon and H. J. Hansen, Helv. Chim. Acta, 60, 978 (1977).
P. Schiess and E. Sendi, Helv. Chim. Acta, 61, 1364 (1978).
R. N. Elgersma, “Einige aspekten van de indolsynthes volgens Fischer,” Dissertation, Rotterdam (1969).
K. H. Pausacker and C. I. Schubert, J. Chem. Soc., No. 7, 1814 (1950).
S. McLean and R. I. Reed, J. Chem. Soc., Part 3, 2519 (1955).
H. Ishii, Acc. Chem. Res., 14, 275 (1981).
Khim. Geterotsikl. Soedin., No. 1, 58 (1971).
H. Albrecht, Tetrahedron Lett., 545 (1971).
B. A. Shainyan and A. N. Mirskova, Usp. Khim., 48, 201 (1979).
Houben-Weyl, Methoden der organischen Chemie, Vol. 4, Book 1, Georg Thieme Verlag, Stuttgart.
D. Biggs and W. D. H. Lyn, J. Chem. Soc., Perkin 2, No. 6, 691 (1976).
H. Decker and P. Becker, Ann. Chem., No. 395, 362 (1913).
F. D. Chattaway and K. J. P. Orton, J. Chem. Soc., Part 1, No. 79, 461 (1901).
R. O. Matevosyan, N. Ya. Postovskii, and A. K. Chirkov, Zh. Obshch. Khim., 29, 858 (1959).
I. I. Grandberg and S. N. Dashkevich, Khim. Geterotsikl. Soedin., No. 3, 342 (1971).
F. Koch, Ber., No. 20, 2460 (1887).
A. N. Frolov, O. V. Kul'bitskaya, and A. V. El'tsov, Zh. Org. Khim., 15, 2118 (1979).
J. D. Entwistle, R. A. W. Johnstone, and A. H. Wilby, Tetrahedron, 38, 419 (1982).
A. Charrier, Gazz. Chim. Ital., No. 461, 367 (1916).
K. H. Bloss and C. E. Timberlake, J. Org. Chem., 28, 267 (1963).
E. Campaigne and R. D. Lake, J. Org. Chem., 24, 479 (1959).
V. F. Martynov, Zh. Obshch. Khim., 27, 1191 (1957).
N. M. Przheval'skii, I. I. Grandberg, N. A. Klyuev, and A. B. Belikov, Khim. Geterotsikl. Soedin., No. 10, 1349 (1978).
N. Campbell and E. B. McCall, J. Chem. Soc., No. 10, 2870 (1950).
N. A. Jones and M. L. Tomlinson, J. Chem. Soc., Part 4, 4114 (1953).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 898–907, July, 1986.
Rights and permissions
About this article
Cite this article
Vysotskii, Y.B., Przheval'skii, N.M., Zemskii, B.P. et al. Mechanism of the Fischer indole synthesis. Quantum-chemical interpretation of the rearrangement of substituted cyclohexanone arylhydrazones to tetrahydrocarbazoles. Chem Heterocycl Compd 22, 713–722 (1986). https://doi.org/10.1007/BF00522729
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00522729