Summary
1. The plantHaplophyllum acutifolium has yielded eudesmine, acetamide, skimmianine, and a new base with the composition C16H19NO, mp 122–123°C, which has been called acutine.
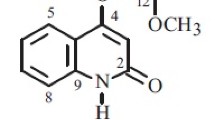
2. It has been shown that acutine has the structure of 2-(hept-4-enyl)-1,4-dihydroquinolin-4-one.
Similar content being viewed by others
Literature cited
D. M. Gulyamova, I. A. Bessonova, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 850 (1971).
D. M. Razzakova, I. A. Bessonova, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 665 (1972).
E. E. Hays, I. C. Wells, et al., J. Biol. Chem.,159, 725 (1945); I. C. Wells, J. Biol. Chem.,196, 331 (1952); J. W. Cornforth and A. F. James, Biochem. J.,63, 124 (1956).
N. J. Leonard, H. F. Herbrandson, and E. M. Van Heyningen, J. Amer. Chem. Soc.,68, 1279 (1946); Ch. R. Hauser and G. A. Reynolds, J. Amer. Chem. Soc.,70, 2402 (1948).
J. Reisch, R. Pagnucco, and N. Jantos, Phytochem.,7, 997 (1968).
R. Tschsche and W. Werner, Tetrahedron,23, 1878 (1967).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 206–210, March–April, 1973.
Rights and permissions
About this article
Cite this article
Razzakova, D.M., Bessonova, I.A. & Yunusov, S.Y. Alkaloids of Haplophyllum acutifolium. Chem Nat Compd 9, 199–202 (1973). https://doi.org/10.1007/BF00563344
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00563344