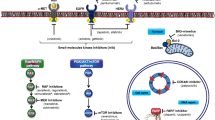
Abstract
Advances made in the treatment of childhood malignancies during the last four decades have resulted in overall cure rates of approximately 80%, but progress has slowed significantly during the last 10 years, underscoring the need for more effective and less toxic agents. Current research is focused on development of molecularly targeted agents, an era ushered in with the discovery of imatinib mesylate for the treatment of chronic myelogenous leukemia. Since imatinib’s introduction into the clinic, an increasing number of tyrosine kinase inhibitors have been developed and entered into clinical trials and practice. Parallel to the initial advances made in molecularly targeted agents has been the development of a spectrum of novel imaging modalities. Future goals for imaging in childhood cancer research thus include (1) patient identification based on target identification or other biologic characteristics of the tumor, (2) assessing pharmacokinetic-pharmacodynamic (PK-PD) effects, and (3) predictive value with an early indication of patient benefit. Development and application of novel imaging modalities for children with cancer can serve to streamline development of molecularly targeted agents.

Similar content being viewed by others
References
Jemal A, Siegel R, Ward E et al (2006) Cancer statistics, 2006. CA Cancer J Clin 56:106–130
Adamson PC, Weiner SL, Simone JV et al (2005) Making better drugs for children with cancer. Committee on Shortening the Time Line for New Cancer Treatments, National Cancer Policy Board. Institute of Medicine, Washington, DC
Ries L, Melbert D, Krapcho M et al (2008) SEER Cancer Statistics Review, 1975–2005. National Cancer Institute, Bethesda, MD, based on November 2007 SEER data submission. Available via http://seer.cancer.gov/csr/1975_2005/index.html
Bhatia S, Meadows AT (2006) Long-term follow-up of childhood cancer survivors: future directions for clinical care and research. Pediatr Blood Cancer 46:143–148
Oeffinger KC, Mertens AC, Sklar CA et al (2006) Chronic health conditions in adult survivors of childhood cancer. N Engl J Med 355:1572–1582
Skinner R, Wallace WH, Levitt GA (2006) Long-term follow-up of people who have survived cancer during childhood. Lancet Oncol 7:489–498
Druker BJ, Tamura S, Buchdunger E et al (1996) Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr–Abl positive cells. Nat Med 2:561–566
Druker BJ, Talpaz M, Resta DJ et al (2001) Efficacy and safety of a specific inhibitor of the BCR–ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 344:1031–1037
Madhusudan S, Ganesan TS (2004) Tyrosine kinase inhibitors in cancer therapy. Clin Biochem 37:618–635
Vlahovic G, Crawford J (2003) Activation of tyrosine kinases in cancer. Oncologist 8:531–538
Gridelli C, Bareschino MA, Schettino C et al (2007) Erlotinib in non-small cell lung cancer treatment: current status and future development. Oncologist 12:840–849
Nanda R (2007) Targeting the human epidermal growth factor receptor 2 (HER2) in the treatment of breast cancer: recent advances and future directions. Rev Recent Clin Trials 2:111–116
Buchdunger E, Cioffi CL, Law N et al (2000) Abl protein-tyrosine kinase inhibitor STI571 inhibits in vitro signal transduction mediated by c-kit and platelet-derived growth factor receptors. J Pharmacol Exp Ther 295:139–145
Linden HM, Stekhova SA, Link JM et al (2006) Quantitative fluoroestradiol positron emission tomography imaging predicts response to endocrine treatment in breast cancer. J Clin Oncol 24:2793–2799
Flaherty KT, Rosen MA, Heitjan DF et al (2008) Pilot study of DCE-MRI to predict progression-free survival with sorafenib therapy in renal cell carcinoma. Cancer Biol Ther 7:496–501
Druker BJ (2002) Perspectives on the development of a molecularly targeted agent. Cancer Cell 1:31–36
Lux ML, Rubin BP, Biase TL et al (2000) KIT extracellular and kinase domain mutations in gastrointestinal stromal tumors. Am J Pathol 156:791–795
Demetri GD, von Mehren M, Blanke CD et al (2002) Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med 347:472–480
Holdsworth CH, Badawi RD, Manola JB et al (2007) CT and PET: early prognostic indicators of response to imatinib mesylate in patients with gastrointestinal stromal tumor. Am J Roentgenol 189:W324–W330
Champagne MA, Capdeville R, Krailo M et al (2004) Imatinib mesylate (STI571) for treatment of children with Philadelphia chromosome-positive leukemia: results from a Children’s Oncology Group phase 1 study. Blood 104:2655–2660
Millot F, Guilhot J, Nelken B et al (2006) Imatinib mesylate is effective in children with chronic myelogenous leukemia in late chronic and advanced phase and in relapse after stem cell transplantation. Leukemia 20:187–192
Aplenc R, Strauss LC, Shusterman S et al (2008) Pediatric phase I trial and pharmacokinetic (PK) study of dasatinib: a report from the Children’s Oncology Group Phase I Consortium. ASCO Meeting Abstracts 26:3591
Gilbertson RJ (2004) Medulloblastoma: signalling a change in treatment. Lancet Oncol 5:209–218
Gilbertson RJ, Bentley L, Hernan R et al (2002) ERBB receptor signaling promotes ependymoma cell proliferation and represents a potential novel therapeutic target for this disease. Clin Cancer Res 8:3054–3064
Gilbertson RJ, Hill DA, Hernan R et al (2003) ERBB1 is amplified and overexpressed in high-grade diffusely infiltrative pediatric brain stem glioma. Clin Cancer Res 9:3620–3624
Daw NC, Furman WL, Stewart CF et al (2005) Phase I and pharmacokinetic study of gefitinib in children with refractory solid tumors: a Children’s Oncology Group Study. J Clin Oncol 23:6172–6180
Freeman BB 3rd, Daw NC, Geyer JR et al (2006) Evaluation of gefitinib for treatment of refractory solid tumors and central nervous system malignancies in pediatric patients. Cancer Invest 24:310–317
Jakacki RI, Tersak J, Blaney S et al (2006) A pediatric phase I trial and pharmacokinetic (PK) study of erlotinib (ERL) followed by the combination of ERL with temozolomide (TMZ): a Children’s Oncology Group study. Journal of Clinical Oncology 2006 ASCO Annual Meeting Proceedings Part I 24:A9015
Bender JL, Adamson PC, Reid JM et al (2008) Phase I trial and pharmacokinetic study of bevacizumab in pediatric patients with refractory solid tumors: a Children’s Oncology Group Study. J Clin Oncol 26:399–405
DuBois SG, Shusterman S, Ingle AM et al (2008) A pediatric phase I trial and pharmacokinetic (PK) study of sunitinib: a Children’s Oncology Group phase I consortium study. ASCO Meeting Abstracts 26:3561
Shankar LK (2008) Molecular imaging in evaluating treatment response. SNM Annual Meeting, New Orleans, LA
PhRMA 2008 report: Medicines in Development for Cancer. PhRMA, Washington
Author information
Authors and Affiliations
Corresponding author
Additional information
Dr. Adamson has no relevant financial relationships or potential conflicts of interest related to the material to be presented.
Rights and permissions
About this article
Cite this article
Adamson, P.C. Imaging in early phase childhood cancer trials. Pediatr Radiol 39 (Suppl 1), 38–41 (2009). https://doi.org/10.1007/s00247-008-1016-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-008-1016-5