Abstract
Purpose
Burkitt’s lymphoma (BL) is an aggressive lymphoma subtype with high 18F-FDG avidity at 18F-FDG-PET/CT, but no validated criteria for PET/CT in treatment evaluation or prediction of outcome in BL are available. The aim of our study was to investigate whether the metabolic baseline PET/CT parameters can predict treatment response and prognosis in BL.
Materials and methods
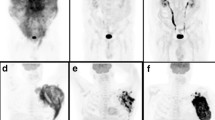
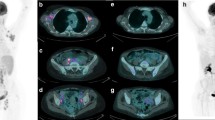
We retrospectively enrolled 65 patients who underwent baseline 18F-FDG-PET/CT, interim and end of treatment PET/CT. The PET images were analyzed visually and semi-quantitatively by measuring the maximum standardized uptake value body weight (SUVbw), the maximum standardized uptake value lean body mass (SUVlbm), the maximum standardized uptake value body surface area (SUVbsa), lesion to liver SUVmax ratio (L-L SUV R), lesion to blood-pool SUVmax ratio (L-BP SUV R), total metabolic tumor volume (tMTV) and total lesion glycolysis (TLG). Survival curves were plotted according to the Kaplan–Meier method.
Results
At a median follow-up of 40 months, the median PFS and OS were 34 and 39 months. MTV and TLG were significantly higher in patients with partial response compared to complete response group at end of treatment, while no significant differences were found at interim. Other metabolic PET/CT parameters were not related to treatment response. MTV and TLG were demonstrated to be independent prognostic factors for both PFS and OS; instead SUVbw, SUVlbm, SUVbsa, L-L SUV R and L-BP SUV R were not related to outcome survival.
Conclusions
Metabolic tumour features (MTV and TLG) were significantly correlated with response to treatment and long-term outcome.




Similar content being viewed by others
References
Swerdlow SH, Campo E, Harris NL, et al. World Health Organization classification of Tumours of Haematopoietic and lymphoid tissues. Lyon: IARC press; 2008.
Bishop PC, Rao VK, Wilson WH. Burkitt’s lymphoma: molecular pathogenesis and treatment. Cancer Investig. 2000;18:574–83.
Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood. 2004;104:3009–20.
Wästerlid T, Brown PN, Hagberg O, Hagberg H, Pedersen LM, D’Amore F, et al. Impact of chemotherapy regimen and rituximab in adult Burkitt lymphoma: a retrospective population-based study from the Nordic lymphoma group. Ann Oncol. 2013;24:1879–86.
Molineux EM, Rochford R, Griffin B, Newton R, Jackson G, Menon G, et al. Burkitt’s lymphoma. Lancet. 2012;379:1234–44.
Sandlund JT. Burkitt lymphoma: staging and response evaluation. Br J Hematol. 2012;156:761–5.
Just PA, Fieschi C, Baillet G, Galicier L, Oksenhendler E, Moretti JL. 18F-fluorodeoxyglucose positron emission tomography/computed tomography in AIDS-related Burkitt lymphoma. AIDS Patient Care STDs. 2008;22:695–700.
Zeng W, Lechowicz MJ, Winton E, Cho SM, Galt JR, Halkar R. Spectrum of FDG PET/CT findings in Burkitt lymphoma. Clin Nucl Med. 2009;4:355–8.
Karantanis D, Durski JM, Lowe VJ, Nathan MA, Mullan BP. Georgiou E, et al. 18F-FDG PET and PET/CT in Burkitt’s lymphoma. Eur J Radiol. 2012;75:e68–73.
Bailly C, Eugène T, Couec ML, Strullu M, Frampas E, Campion L, et al. Prognostic value and clinical impact of (18)FDG-PET in the management of children with Burkitt lymphoma after induction chemotherapy. Front Med (Lausanne). 2014;16(1):54.
Carrillo-Cruz E, Mar ın-Oyaga VA, Rodrıguez MS, Borrego-Dorado I, de la Cruz Vicente F, Quiroga Cantero E, et al. Role of 18F-FDG-PET/CT in the management of Burkitt lymphoma. Eur J Haematol. 2015;94:23–30.
Wen WX, Huang JJ, Li WY, Zhang X, Xia Y, Jiang WQ, et al. Prognostic values of interim and post-therapy 18F-FDG PET/CT scanning in adult patients with Burkitt’s lymphoma. Chin J Cancer. 2015;2(34):608–13.
Albano D, Bosio G, Re A, Pagani C, Giubbini R, Bertagna F. Metabolic behaviour and prognostic value of early and end of treatment 18F-FDG PET/CT in adult burkitt lymphoma: role of Deauville and IHP criteria. Leuk Lymphoma. 2018. https://doi.org/10.1080/10428194.2018.1482541.
Meignan M, Gallamini A, Haioun C, Polliack A. Report on the second international workshop on interim positron emission tomography in lymphoma held in Menton, France, 8–9 April 2010. Leuk Lymphoma. 2010;51:2171–80.
Kostakoglu L, Chauvie S. Metabolic tumour volume metrics in lymphoma. Semin Nucl Med. 2018;48:50–66.
Xie M, Wu K, Liu Y, Jiang Q, Xie Y. Predictive value of F-18 FDG PET/CT quantization parameters in diffuse large B cell lymphoma: a meta-analysis with 702 participants. Med Oncol. 2015;32:446.
Procházka V, Klugar M, Bachanova V, Klugarová J, Tučková D, Papajík T. Comparing the accuracy of quantitative versus qualitative analyses of interim PET to prognosticate Hodgkin lymphoma: a systematic review protocol of diagnostic test accuracy. BMJ Open. 2016;5:6.
Hoelzer D, Walewski J, Döhner H, Viardot A, Hiddemann W, Spiekermann K, et al. Improved outcome of adult Burkitt lymphoma/leukemia with rituximab and chemotherapy: report of a large prospective multicenter trial. Blood. 2014;124:3870–9.
Ferreri AJ, Bruno Ventre M, Donadoni G, Cattaneo C, Fumagalli L, Foppoli M, et al. Safety and activity of a new intensive short-term chemoimmunotherapy in HIV-positive patients with Burkitt lymphoma. Br J Haematol. 2012;159:252–5.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–54.
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32:3059–68.
Barrington SF, Mikhaeel NG, Kostakoglu L, Meignan M, Hutchings M, Müeller SP, et al. Role of imaging in the staging and response assessment of lymphoma: consensus of the international conference on malignant lymphomas imaging working group. J Clin Oncol. 2014;32:3048–58.
Itti E, Lin C, Dupuis J, Paone G, Capacchione D, Rahmouni A, et al. Prognostic value of interim 18F-FDG PET in patients with diffuse large B-cell lymphoma: SUV-based assessment at 4 cycles of chemotherapy. J Nucl Med. 2009;50:527–33.
Casasnovas RO, Meignan M, Berriolo-Riedinger A, Bardet S, Julian A, Thieblemont C, et al. Groupe d’etude des lymphomes de l’adulte (GELA). SUVmax reduction improves early prognosis value of interim positron emission tomography scans in diffuse large B-cell lymphoma. Blood. 2011;118:37–43.
Itti E, Meignan M, Berriolo-Riedinger A, Biggi A, Cashen AF, Véra P, et al. An international confirmatory study of the prognostic value of early PET/CT in diffuse large B-cell lymphoma: comparison between Deauville criteria and ΔSUVmax. Eur J Nucl Med Mol Imaging. 2013;40:1312–20.
Dührsen U, Müller S, Hertenstein B, Thomssen H, Kotzerke J, Mesters R, et al. Positron emission tomography-guided therapy of aggressive non-Hodgkin lymphomas (PETAL): a multicenter, randomized phase III trial. J Clin Oncol. 2018;36:2024–34.
Kanoun S, Rossi C, Berriolo-Riedinger A, Dygai-Cochet I, Cochet A, Humbert O, et al. Baseline metabolic tumour volume is an independent prognostic factor in Hodgkin lymphoma. Eur J Nucl Med Mol Imaging. 2014;41:1735–43.
Moskowitz AJ, Schoder H, Gavane S, Thoren KL, Fleisher M, Yahalom J, et al. Prognostic significance of baseline metabolic tumor volume in relapsed and refractory Hodgkin lymphoma. Blood. 2017;130:2196–203.
Sharma P, Gupta A, Patel C, Bakhshi S, Malhotra A, Kumar R. Pediatric lymphoma: metabolic tumor burden as quantitative index for treatment response evaluation. Ann Nucl Med. 2012;26:58–66.
Sasanelli M, Meignan M, Haioun C, Berriolo-Riedinger A, Casasnovas RO, Biggi A, et al. Pretherapy metabolic tumour volume is an independent predictor of outcome in patients with diffuse large B-cell lymphoma. Eur J Nucl Med Mol Imaging. 2014;41:2017–22.
Mikhaeel NG, Smith D, Dunn JT, Phillips M, Møller H, Fields PA, et al. Combination of baseline metabolic tumour volume and early response on PET/CT improves progression-free survival prediction in DLBCL. Eur J Nucl Med Mol Imaging. 2016;43:1209–19.
Meignan M, Cottereau AS, Versari A, Chartier L, Dupuis J, Boussetta S, et al. Baseline metabolic tumor volume predicts outcome in high–tumor-burden follicular lymphoma: a pooled analysis of three multicenter studies. J Clin Oncol. 2016;34:3618–26.
Weiler-Sagie M, Bushelev O, Epelbaum R, Dann EJ, Haim N, Avivi I, et al. (18)F-FDG avidity in lymphoma readdressed: a study of 766 patients. J Nucl Med. 2010;51:25–30.
Cottereau AS, Becker S, Broussais F, Casasnovas O, Kanoun S, Roques M, et al. Prognostic value of baseline total metabolic tumor volume (TMTV0) measured on FDG-PET/CT in patients with peripheral T-cell lymphoma (PTCL). Ann Oncol. 2016;27:719–24.
Cottereau AS, Buvat I, Kanoun S, Versari A, Casasnovas O, Chauvie S, et al. Is there an optimal method for measuring baseline metabolic tumor volume in diffuse large B cell lymphoma? Eur J Nucl Med Mol Imaging. 2018;450:1463–4.
Cottereau AS, Lanic H, Mareschal S, Meignan M, Vera P, Tilly H, et al. Molecular profile and FDG-PET/CT Total metabolic tumor volume improve risk classification at diagnosis for patients with diffuse large B-cell lymphoma. Clin Cancer Res. 2016;22:3801–9.
Toledano MN, Desbordes P, Banjar A, Gardin I, Vera P, Ruminy P, et al. Combination of baseline FDG PET/CT total metabolic tumour volume and gene expression profile have a robust predictive value in patients with diffuse large B-cell lymphoma. Eur J Nucl Med Mol Imaging. 2018;45:680–8.
Okuyucu K, Ozaydin S, Alagoz E, Ozgur G, Ince S, Oysul FG, et al. Prognosis estimation under the light of metabolic tumor parameteres on initial FDG-PET/CT in patients with primary extranodal lymphoma. Radiol Oncol. 2016;50:360–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
Informed consent was obtained from all individual participant included in the study.
Rights and permissions
About this article
Cite this article
Albano, D., Bosio, G., Pagani, C. et al. Prognostic role of baseline 18F-FDG PET/CT metabolic parameters in Burkitt lymphoma. Eur J Nucl Med Mol Imaging 46, 87–96 (2019). https://doi.org/10.1007/s00259-018-4173-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-4173-2