Abstract
Background
Nasopharyngeal carcinoma (NPC) is notable for its high incidence rates in select geographic and ethnic populations, especially among Chinese and Malay populations in Southeastern Asia. However, relevant biomarkers for the development and prognosis of NPC are not yet clear; therefore, discovering novel biomarkers will facilitate prediction of prognosis and development of targeted therapeutic tactics. This study aims to quest the potential prognostic value of NUAK1 (a downstream member of Akt) in NPC.
Methods
Immunohistochemistry was performed to measure the expression of NUAK1 in paraffin-embedded NPC samples. Statistical analysis, encompassing chi-square tests and Student’s t test, was also employed to evaluate the association between the expression of NUAK1 and clinicopathologic features. In addition, the survival analysis was used to detect the prognostic significance of NUAK1 in NPC.
Results
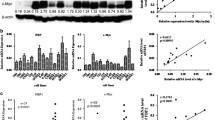
Excessive expression of NUAK1 was found in NPC tissues at mRNA levels. Statistical analysis revealed a correlation of NUAK1 expression with maximum neck lymph node diameter (p = 0.025) and WHO histological type (p = 0.021). Furthermore, according to survival analysis, there was clinical relevance between the upregulation of NUAK1 in NPC and DFS. Subgroup analysis indicated that the expression of NUAK1 was strongly associated with DFS (p = 0.027) and OS (p = 0.026) duration in the patients of N1–3 tumors, but not in patients with N0 tumors. The expression of NUAK1 was also strongly associated with OS (p = 0.044) and DFS (p = 0.007) in patients of late stage tumour (UICC3–5), but not in patients of early stage tumour (UICC1–2). In addition, COX regression illustrated that N classification and NUAK1 expression were independent prognostic factors for disease-free survival.
Conclusion
Our study postulated that NUAK1 is excessively expressed in NPC and may serve as a potential predictor of prognosis for NPC.





Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA 65(2):87–108
2 Wei KR, Zheng RS, Zhang SW, Liang ZH, Li ZM, Chen WQ (2017) Nasopharyngeal carcinoma incidence and mortality in China, 2013. Chin J Cancer 36(1):90. https://doi.org/10.1186/s40880-017-0257-9
You B, Shan Y, Shi S, Li X, You. Y (2015) Effects of ADAM10 upregulation on progression, migration, and prognosis of nasopharyngeal carcinoma. Cancer Sci 106(11):1506–1514
Zhou XJ, Wang Y, Zhang LJ, Chen JH (2015) Expression and clinical significance of LRRC4 in benign and malignant nasopharyngeal diseases. Genet Mol Res 14(4):16403–16409
Li J, Lu Z, Peng G, Tan N, Liu D, Huang Z, Zhang C, Duan X, Tang F (2014) N,N′-dinitrosopiperazine-mediated AGR2 is involved in metastasis of nasopharyngeal carcinoma. PLoS One 9(4):e92081
Huang L, Xu X, Hao Y, Chen J, Li L, Cheng J, Chen Z, Liang W, Yang J, Ao W, Hao X, Gao W (2015) Overexpression of CSF-1R in nasopharyngeal carcinoma. Rom J Morphol Embryol 56(4):1279–1283
Li M, Zheng C, Xu H, He W, Ruan Y, Ma J, Zheng J, Ye C, Li W (2017) Inhibition of AMPK-related kinase 5 (ARK5) enhances cisplatin cytotoxicity in non-small cell lung cancer cells through regulation of epithelial–mesenchymal transition. Am J Transl Res 9(4):1708–1719
Kusakai G, Suzuki A, Ogura T, Miyamoto S, Ochiai A, Kaminishi M, Esumi H (2004) ARK5 expression in colorectal cancer and its implications for tumor progression. Am J Pathol 164(3):987–995
Suzuki A et al (2004) ARK5 is a tumor invasion-associated factor downstream of Akt signaling. Mol Cell Biol 24(8):3526–3535
Chen D, Liu G, Xu N, You X, Zhou H, Zhao X, Liu Q (2017) Knockdown of ARK5 expression suppresses invasion and metastasis of gastric cancer. Cell Physiol Biochem 42(3):1025–1036
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, Barrette T, Pandey A, Chinnaiyan AM (2004) ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6(1):1–6
Obayashi M, Yoshida M, Tsunematsu T, Ogawa I, Sasahira T, Kuniyasu H, Imoto I, Abiko Y, Xu D, Fukunaga S, Tahara H, Kudo Y, Nagao T, Takata T (2016) microRNA-203 suppresses invasion and epithelial-mesenchymal transition induction via targeting NUAK1 in head and neck cancer. Oncotarget 7(7):8223–39. https://doi.org/10.18632/oncotarget.6972
Lasagna-Reeves CA, de Haro M, Hao S, Park J, Rousseaux MW, Al-Ramahi I, Jafar-Nejad P, Vilanova-Velez L, See L, De Maio A, Nitschke L, Wu Z, Troncoso JC, Westbrook TF, Tang J, Botas J, Zoghbi HY (2016) Reduction of Nuak1 decreases Tau and reverses phenotypes in a tauopathy mouse model. Neuron 92(2):407–418. https://doi.org/10.1016/j.neuron.2016.09.022 (Epub 6 Oct 2016)
Datta SR, Brunet A, Greenberg ME (1999) Cellular survival: a play in three Akts. Genes Dev 13(22):2905–27 (Review, PubMed PMID: 10579998)
Park BK, Zeng X, Glazer RI (2001) Akt1 induces extracellular matrix invasion and matrix metalloproteinase-2 activity in mouse mammary epithelial cells. Cancer Res 61(20):7647–53
Phippen NT, Bateman NW, Wang G, Conrads KA, Ao W, Teng P, Litzi TA, Oliver J, Maxwell GL, Hamilton CA, Darcy KM, Conrads TP (2016) NUAK1 (ARK5) Is associated with poor prognosis in ovarian cancer. Front Oncol 6:213
Tanno S, Tanno S, Mitsuuchi Y, Altomare DA, Xiao GH, Testa JR (2001) AKT activation up-regulates insulin-like growth factor I receptor expression and promotes invasiveness of human pancreatic cancer cells. Cancer Res 61(2):589–93
Grille SJ, Bellacosa A, Upson J, Klein-Szanto AJ, van Roy F, Lee-Kwon W, Donowitz M, Tsichlis PN, Larue L (2003) The protein kinase Akt induces epithelial mesenchymal transition and promotes enhanced motility and invasiveness of squamous cell carcinoma lines. Cancer Res 63(9):2172–2178
Mu QJ, Li HL, Yao Y, Liu SC, Yin CG, Ma XZ (2015) Chromodomain helicase/ATPase DNA-binding protein 1-like gene (CHD1L) expression and implications for invasion and metastasis of breast cancer. PLoS One 10(11):e0143030. https://doi.org/10.1371/journal.pone.0143030 (eCollection 2015)
Arboleda MJ, Lyons JF, Kabbinavar FF, Bray MR, Snow BE, Ayala R, Danino M, Karlan BY, Slamon DJ (2003) Overexpression of AKT2/protein kinase Bbeta leads to up-regulation of beta1 integrins, increased invasion, and metastasis of human breast and ovarian cancer cells. Cancer Res 63(1):196–206
Suzuki A, Kusakai G, Kishimoto A, Lu J, Ogura T, Esumi H (2003) ARK5 suppresses the cell death induced by nutrient starvation and death receptors via inhibition of caspase 8 activation, but not by chemotherapeutic agents or UV irradiation. Oncogene 22(40):6177–6182
Suzuki A, Kusakai G, Kishimoto A, Lu J, Ogura T, Lavin MF, Esumi H (2003) Identification of a novel protein kinase mediating Akt survival signaling to the ATM protein. J Biol Chem 278(1):48–53. (Epub 29 Oct 2002)
Shi L, Zhang B, Sun X, Lu S, Liu Z, Liu Y, Li H, Wang L, Wang X, Zhao C (2014) MiR-204 inhibits human NSCLC metastasis through suppression of NUAK1. Br J Cancer 9(12):2316–2327. https://doi.org/10.1038/bjc.2014.580 (Epub 20 Nov 2014)
Chen D, Liu G, Xu N, You X, Zhou H, Zhao X, Liu Q (2017) Knockdown of ARK5 expression suppresses invasion and metastasis of gastric cancer. Cell Physiol Biochem 42(3):1025–1036. https://doi.org/10.1159/000478685 (Epub 28 Jun 2017)
Riester M, Wei W, Waldron L, Culhane AC, Trippa L, Oliva E, Kim SH, Michor F, Huttenhower C, Parmigiani G, Birrer MJ (2014) Risk prediction for late-stage ovarian cancer by meta-analysis of 1525 patient samples. J Natl Cancer Inst. https://doi.org/10.1093/jnci/dju048
Suzuki G, Kusakai A, Kishimoto J, Lu T, Ogura MF, Lavin, Esumi H (2003) Identification of a novel protein kinase mediating Akt survival signaling to the ATM protein. J Biol Chem 278(1):48–53
Gil J, Bernard D, Humbert N, Carling D, Augert A, Martinez D, Martien S, Wang J, Abbadie C, Da Costa M, de Launoit Y, Navaratnam N (2010) Regulation of ploidy and senescence by the AMPK-related kinase NUAK1. EMBO J 29(2):376–386
Obayashi M et al (2016) microRNA-203 suppresses invasion and epithelial–mesenchymal transition induction via targeting NUAK1 in head and neck cancer. Oncotarget 7(7):8223–39. https://doi.org/10.18632/oncotarget.6972
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81672661), the Guangdong Province Natural Science Foundation (2015A030310126).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the ethics committee of the First Affiliated Hospital of Guangzhou Medical University. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study. And each patient had given a written informed consent about the use of the samples for medical research.
Rights and permissions
About this article
Cite this article
Liu, J., Tang, G., Huang, H. et al. Expression level of NUAK1 in human nasopharyngeal carcinoma and its prognostic significance. Eur Arch Otorhinolaryngol 275, 2563–2573 (2018). https://doi.org/10.1007/s00405-018-5095-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-018-5095-0