Abstract
Background
C3 glomerulopathies (C3G) are characterized by uncontrolled activation of the alternative pathway of complement. In most patients these diseases progress towards end-stage renal disease, and the risk of recurrence after renal transplantation is high. In the majority of patients, only antibodies against the C3 convertase, termed C3Nef, can be found as a potential pathogenic factor. Although a large variety of therapeutic approaches have been used, no generally accepted therapy exists.
Methods
In four consecutive patients with C3G in whom all known complement factor mutations were excluded and only C3Nef could be identified as a potential cause of disease, a multimodal therapeutic regimen with plasma therapy, corticosteroids and mycophenolate mofetil was used.
Results
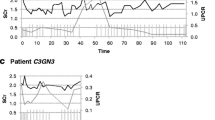
The multimodal regimen achieved normalization of renal function in all four patients, with complete remission in two patients and a distinct reduction of proteinuria in the other two patients. The single patient with C3 glomerulonephritis (C3GN) and marked terminal complement complex elevation only showed partial remission; further improvement was achieved following the addition of eculizumab to the therapeutic regimen. Repeatedly measured C3Nef levels did not correlate with disease course or therapeutic response in any of the patients.
Conclusions
As this multimodal therapeutic approach was effective in all four treated patients with suspected autoimmune etiology of C3G, it offers a treatment option for severely affected patients with this rare disease until more specific regimens are available.


Similar content being viewed by others
References
Leroy V, Fremeaux-Bacchi V, Peuchmaur M, Baudouin V, Deschenes G, Macher MA, Loirat C (2011) Membranoproliferative glomerulonephritis with C3NeF and genetic complement dysregulation. Pediatr Nephrol 26:419–424
Smith RJ, Alexander J, Barlow PN, Botto M, Cassavant TL, Cook HT, de Cordoba SR, Hageman GS, Jokiranta TS, Kimberling WJ, Lambris JD, Lanning LD, Levidiotis V, Licht C, Lutz HU, Meri S, Pickering MC, Quigg RJ, Rops AL, Salant DJ, Sethi S, Thurman JM, Tully HF, Tully SP, van der Vlag J, Walker PD, Wurzner R, Zipfel PF (2007) New approaches to the treatment of dense deposit disease. J Am Soc Nephrol 18:2447–2456
Lu DF, McCarthy AM, Lanning LD, Delaney C, Porter C (2007) A descriptive study of individuals with membranoproliferative glomerulonephritis. Nephrol Nurs J 34:295–302, quiz 303
Lu DF, Moon M, Lanning LD, McCarthy AM, Smith RJ (2012) Clinical features and outcomes of 98 children and adults with dense deposit disease. Pediatr Nephrol 27:773–781
Andresdottir MB, Assmann KJ, Hoitsma AJ, Koene RA, Wetzels JF (1999) Renal transplantation in patients with dense deposit disease: morphological characteristics of recurrent disease and clinical outcome. Nephrol Dial Transplant 14:1723–1731
Jozsi M, Richter H, Loschmann I, Skerka C, Buck F, Beisiegel U, Erdei A, Zipfel PF (2005) FHR-4A: a new factor H-related protein is encoded by the human FHR-4 gene. Eur J Hum Genet 13:321–329
Sethi S, Fervenza FC (2011) Membranoproliferative glomerulonephritis: pathogenetic heterogeneity and proposal for a new classification. Semin Nephrol 31:341–348
Hou J, Markowitz GS, Bomback AS, Appel GB, Herlitz LC, Barry Stokes M, D’Agati VD (2014) Toward a working definition of C3 glomerulopathy by immunofluorescence. Kidney Int 85:450–456
Bomback AS, Appel GB (2012) Pathogenesis of the C3 glomerulopathies and reclassification of MPGN. Nat Rev Nephrol 8:634–642
Sethi S, Fervenza FC (2012) Membranoproliferative glomerulonephritis—a new look at an old entity. N Engl J Med 366:1119–1131
Zhang Y, Meyer NC, Wang K, Nishimura C, Frees K, Jones M, Katz LM, Sethi S, Smith RJ (2012) Causes of alternative pathway dysregulation in dense deposit disease. Clin J Am Soc Nephrol 7:265–274
Servais A, Noel LH, Roumenina LT, Le Quintrec M, Ngo S, Dragon-Durey MA, Macher MA, Zuber J, Karras A, Provot F, Moulin B, Grunfeld JP, Niaudet P, Lesavre P, Fremeaux-Bacchi V (2012) Acquired and genetic complement abnormalities play a critical role in dense deposit disease and other C3 glomerulopathies. Kidney Int 82:454–464
Martinez-Barricarte R, Heurich M, Valdes-Canedo F, Vazquez-Martul E, Torreira E, Montes T, Tortajada A, Pinto S, Lopez-Trascasa M, Morgan BP, Llorca O, Harris CL, Rodriguez de Cordoba S (2010) Human C3 mutation reveals a mechanism of dense deposit disease pathogenesis and provides insights into complement activation and regulation. J Clin Invest 120:3702–3712
Abrera-Abeleda MA, Nishimura C, Smith JL, Sethi S, McRae JL, Murphy BF, Silvestri G, Skerka C, Jozsi M, Zipfel PF, Hageman GS, Smith RJ (2006) Variations in the complement regulatory genes factor H (CFH) and factor H related 5 (CFHR5) are associated with membranoproliferative glomerulonephritis type II (dense deposit disease). J Med Genet 43:582–589
Gewurz AT, Imherr SM, Strauss S, Gewurz H, Mold C (1983) C3 nephritic factor and hypocomplementaemia in a clinically healthy individual. Clin Exp Immunol 54:253–258
Fremeaux-Bacchi V, Weiss L, Demouchy C, May A, Palomera S, Kazatchkine MD (1994) Hypocomplementaemia of poststreptococcal acute glomerulonephritis is associated with C3 nephritic factor (C3NeF) IgG autoantibody activity. Nephrol Dial Transplant 9:1747–1750
Licht C, Heinen S, Jozsi M, Loschmann I, Saunders RE, Perkins SJ, Waldherr R, Skerka C, Kirschfink M, Hoppe B, Zipfel PF (2006) Deletion of Lys224 in regulatory domain 4 of Factor H reveals a novel pathomechanism for dense deposit disease (MPGN II). Kidney Int 70:42–50
Krmar RT, Holtback U, Linne T, Berg UB, Celsi G, Soderberg MP, Wernerson A, Szakos A, Larsson S, Skattum L, Barany P (2011) Acute renal failure in dense deposit disease: complete recovery after combination therapy with immunosuppressant and plasma exchange. Clin Nephrol 75[Suppl 1]:4–10
McGinley E, Watkins R, McLay A, Boulton-Jones JM (1985) Plasma exchange in the treatment of mesangiocapillary glomerulonephritis. Nephron 40:385–390
Kurtz KA, Schlueter AJ (2002) Management of membranoproliferative glomerulonephritis type II with plasmapheresis. J Clin Apher 17:135–137
Berthe-Aucejo A, Sacquepee M, Fila M, Peuchmaur M, Perrier-Cornet E, Fremeaux-Bacchi V, Deschenes G (2014) Blockade of alternative complement pathway in dense deposit disease. Case Rep Nephrol 2014:201568
Tarshish P, Bernstein J, Tobin JN, Edelmann CM Jr (1992) Treatment of mesangiocapillary glomerulonephritis with alternate-day prednisone–a report of the International Study of Kidney Disease in Children. Pediatr Nephrol 6:123–130
Appel GB, Cook HT, Hageman G, Jennette JC, Kashgarian M, Kirschfink M, Lambris JD, Lanning L, Lutz HU, Meri S, Rose NR, Salant DJ, Sethi S, Smith RJ, Smoyer W, Tully HF, Tully SP, Walker P, Welsh M, Wurzner R, Zipfel PF (2005) Membranoproliferative glomerulonephritis type II (dense deposit disease): an update. J Am Soc Nephrol 16:1392–1403
Daina E, Noris M, Remuzzi G (2012) Eculizumab in a patient with dense-deposit disease. N Engl J Med 366:1161–1163
Jones G, Juszczak M, Kingdon E, Harber M, Sweny P, Burns A (2004) Treatment of idiopathic membranoproliferative glomerulonephritis with mycophenolate mofetil and steroids. Nephrol Dial Transplant 19:3160–3164
Nicolas C, Vuiblet V, Baudouin V, Macher MA, Vrillon I, Biebuyck-Gouge N, Dehennault M, Gie S, Morin D, Nivet H, Nobili F, Ulinski T, Ranchin B, Marinozzi MC, Ngo S, Fremeaux-Bacchi V, Pietrement C (2014) C3 nephritic factor associated with C3 glomerulopathy in children. Pediatr Nephrol 29:85–94
Rousset-Rouviere C, Cailliez M, Garaix F, Bruno D, Laurent D, Tsimaratos M (2014) Rituximab fails where eculizumab restores renal function in C3nef-related DDD. Pediatr Nephrol 29:1107–1111
McCaughan JA, O’Rourke DM, Courtney AE (2012) Recurrent dense deposit disease after renal transplantation: an emerging role for complementary therapies. Am J Transplant 12:1046–1051
Bomback AS, Smith RJ, Barile GR, Zhang Y, Heher EC, Herlitz L, Stokes MB, Markowitz GS, D’Agati VD, Canetta PA, Radhakrishnan J, Appel GB (2012) Eculizumab for dense deposit disease and C3 glomerulonephritis. Clin J Am Soc Nephrol 7:748–756
Vivarelli M, Pasini A, Emma F (2012) Eculizumab for the treatment of dense-deposit disease. N Engl J Med 366:1163–1165
Pickering MC, Warren J, Rose KL, Carlucci F, Wang Y, Walport MJ, Cook HT, Botto M (2006) Prevention of C5 activation ameliorates spontaneous and experimental glomerulonephritis in factor H-deficient mice. Proc Natl Acad Sci USA 103:9649–9654
Bomback AS (2014) Anti-complement therapy for glomerular diseases. Adv Chronic Kidney Dis 21:152–158
Pottel H, Hoste L, Martens F (2012) A simple height-independent equation for estimating glomerular filtration rate in children. Pediatr Nephrol 27:973–979
Rother U (1982) A new screening test for C3 nephritis factor based on a stable cell bound convertase on sheep erythrocytes. J Immunol Methods 51:101–107
Moore I, Strain L, Pappworth I, Kavanagh D, Barlow PN, Herbert AP, Schmidt CQ, Staniforth SJ, Holmes LV, Ward R, Morgan L, Goodship TH, Marchbank KJ (2009) Association of factor H autoantibodies with deletions of CFHR1, CFHR3, CFHR4, and with mutations in CFH, CFI, CD46, and C3 in patients with atypical hemolytic uremic syndrome. Blood 115:379–387
Holmes LV, Strain L, Staniforth SJ, Moore I, Marchbank K, Kavanagh D, Goodship JA, Cordell HJ, Goodship TH (2013) Determining the population frequency of the CFHR3/CFHR1 deletion at 1q32. PLoS One 8:e60352
Nester CM, Smith RJ (2013) Treatment options for C3 glomerulopathy. Curr Opin Nephrol Hypertens 22:231–237
Paixao-Cavalcante D, Lopez-Trascasa M, Skattum L, Giclas PC, Goodship TH, de Cordoba SR, Truedsson L, Morgan BP, Harris CL (2012) Sensitive and specific assays for C3 nephritic factors clarify mechanisms underlying complement dysregulation. Kidney Int 82:1084–1092
Hofer J, Janecke AR, Zimmerhackl LB, Riedl M, Rosales A, Giner T, Cortina G, Haindl CJ, Petzelberger B, Pawlik M, Jeller V, Vester U, Gadner B, van Husen M, Moritz ML, Wurzner R, Jungraithmayr T, German-Austrian HUSSG (2013) Complement factor H-related protein 1 deficiency and factor H antibodies in pediatric patients with atypical hemolytic uremic syndrome. Clin J Am Soc Nephrol 8:407–415
Yokoyama H, Wada T, Zhang W, Yamaya H, Asaka M (2007) Advances in apheresis therapy for glomerular diseases. Clin Exper Nephrol 11:122–127
Choi MJ, Eustace JA, Gimenez LF, Atta MG, Scheel PJ, Sothinathan R, Briggs WA (2002) Mycophenolate mofetil treatment for primary glomerular diseases. Kidney Int 61:1098–1114
Sepe V, Libetta C, Giuliano MG, Adamo G, Dal Canton A (2008) Mycophenolate mofetil in primary glomerulopathies. Kidney Int 73:154–162
Acknowledgments
This work was supported by contract research “glycomic/glycobiologie” of the Baden-Württemberg Stiftung. We thank Maren Lingnau for technical assistance.
Disclosure
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Häffner, K., Michelfelder, S. & Pohl, M. Successful therapy of C3Nef-positive C3 glomerulopathy with plasma therapy and immunosuppression. Pediatr Nephrol 30, 1951–1959 (2015). https://doi.org/10.1007/s00467-015-3111-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-015-3111-9