Abstract
Purpose
Plasmacytoid dendritic cells (PDCs) infiltration into breast cancer tissues is associated with poor prognosis. Also, CXCR4 shows compelling evidences to be exploited by cancer cells to migrate to distant sites. The present study investigated lymph node metastasis in the light of PDCs infiltration and the potential cross talk with CXCR4/SDF-1 chemokine axis.
Methods
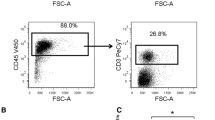
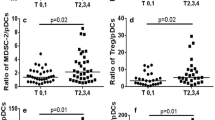
We assessed circulating PDCs proportions drained from the axillary tributaries, and the in situ expression of both CD303 and CXCR4 in breast cancer patients with positive lymph nodes (pLN) and negative lymph nodes (nLN) using immunohistochemistry and flow cytometry. We also analyzed the expression of SDF-1 in lymph nodes of pLN and nLN patients. We studied the effect of the secretome of PDCs of pLN and nLN patients on the expression of CXCR4 and activation of NF-κB in human breast cancer cell lines SKBR3 and MCF-7. TNF-α mRNA expression level in PDCs from both groups was determined by qPCR.
Results
Our findings indicate increased infiltration of PDCs in breast cancer tissues of pLN patients than nLN patients, which correlates with CXCR4+ cells percentage. Interestingly, SDF-1 is highly immunostained in lymph nodes of pLN patients compared to nLN patients. Our in vitro experiments demonstrate an upregulation of NF-κB expression and CXCR4 cells upon stimulation with PDCs secretome of pLN patients than those of nLN patients. Also, PDCs isolated from pLN patients exhibited a higher TNF-α mRNA expression than nLN patients. Treatment of MCF-7 cell lines with TNF-α significantly upregulates CXCR4 expression.
Conclusions
Our findings suggest a potential role for microenvironmental PDCs in breast cancer lymph node metastasis via CXCR4/SDF-1 axis.





Similar content being viewed by others
References
Scanlon EF, Murthy S (1991) The process of metastasis. CA Cancer J Clin 41(5):301–305
Bodenstine TM, Welch DR (2008) Metastasis suppressors and the tumor microenvironment. Cancer Microenviron 1(1):1–11
Nathanson SD (2003) Insights into the mechanisms of lymph node metastasis. Cancer 98(2):413–423
Romaniuk A, Lyndin M (2015) Immune microenvironment as a factor of breast cancer progression. Diagn Pathol 10:79
Man YG et al (2013) Tumor-infiltrating immune cells promoting tumor invasion and metastasis: existing theories. J Cancer 4(1):84–95
Steinman RM (1991) The dendritic cell system and its role in immunogenicity. Annu Rev Immunol 9:271–296
Zhou H, Wu L (2017) The development and function of dendritic cell populations and their regulation by miRNAs. Protein Cell 8(7):501–513
Ma Y et al (2013) Dendritic cells in the cancer microenvironment. J Cancer 4(1):36–44
Merad M et al (2013) The dendritic cell lineage: ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu Rev Immunol 31:563–604
Rovati B et al (2008) Flow cytometric detection of circulating dendritic cells in healthy subjects. Eur J Histochem 52(1):45–52
Rajagopal D et al (2010) Plasmacytoid dendritic cell-derived type I interferon is crucial for the adjuvant activity of Toll-like receptor 7 agonists. Blood 115(10):1949–1957
Treilleux I et al (2004) Dendritic cell infiltration and prognosis of early stage breast cancer. Clin Cancer Res 10(22):7466–7474
Sawant A et al (2012) Depletion of plasmacytoid dendritic cells inhibits tumor growth and prevents bone metastasis of breast cancer cells. J Immunol 189(9):4258–4265
Hiller D, Chu QD (2011) CXCR4 and axillary lymph nodes: review of a potential biomarker for breast cancer metastasis. Int J Breast Cancer 2011:420981
Bromley SK, Mempel TR, Luster AD (2008) Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat Immunol 9(9):970–980
Chatterjee S, Behnam B, Azad, Nimmagadda S (2014) The intricate role of CXCR4 in cancer. Adv Cancer Res 124:31–82
Teicher BA, Fricker SP (2010) CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res 16(11):2927–2931
Monti M et al (1990) Axillary versus peripheral blood levels of sialic acid, ferritin, and CEA in patients with breast cancer. Breast Cancer Res Treat 17(2):77–82
Okumura Y et al (2009) Circulating tumor cells in pulmonary venous blood of primary lung cancer patients. Ann Thorac Surg 87(6):1669–1675
Katsuno H et al (2008) Does the presence of circulating tumor cells in the venous drainage of curative colorectal cancer resections determine prognosis? A meta-analysis. Ann Surg Oncol 15(11):3083–3091
El-Shinawi M et al (2010) Capturing and characterizing immune cells from breast tumor microenvironment: an innovative surgical approach. Ann Surg Oncol 17(10):2677–2684
Boiocchi L et al (2013) BDCA-2 (CD303): a highly specific marker for normal and neoplastic plasmacytoid dendritic cells. Blood 122(2):296–297
Fung E et al (2010) Multiplexed immunophenotyping of human antigen-presenting cells in whole blood by polychromatic flow cytometry. Nat Protoc 5(2):357–370
Maecker HT, McCoy JP, Nussenblatt R (2012) Standardizing immunophenotyping for the Human Immunology Project. Nat Rev Immunol 12(3):191–200
Nouh MA et al (2011) Cathepsin B: a potential prognostic marker for inflammatory breast cancer. J Transl Med 9:1
Kaczmarek E, Gorna A, Majewski P (2004) Techniques of image analysis for quantitative immunohistochemistry. Rocz Akad Med Bialymst 49(Suppl 1):155–158
Rizzardi AE et al (2012) Quantitative comparison of immunohistochemical staining measured by digital image analysis versus pathologist visual scoring. Diagn Pathol 7:42
Gassmann M et al (2009) Quantifying western blots: pitfalls of densitometry. Electrophoresis 30(11):1845–1855
Tan HY, Ng TW (2008) Accurate step wedge calibration for densitometry of electrophoresis gels. Opt Commun 281(10):3013–3017
Jin P et al (2010) Molecular signatures of maturing dendritic cells: implications for testing the quality of dendritic cell therapies. J Transl Med 8(1):4
Xu C et al (2015) CXCR4 in breast cancer: oncogenic role and therapeutic targeting. Drug Des Dev Ther 9:4953–4964
Xiao G, Fu J (2011) NF-kappaB and cancer: a paradigm of Yin-Yang. Am J Cancer Res 1(2):192–221
Wu Y, Zhou BP (2010) TNF-α/NF-κB/Snail pathway in cancer cell migration and invasion. Br J Cancer 102(4):639–644
Ma Y et al (2011) The dendritic cell-tumor cross-talk in cancer. Curr Opin Immunol 23(1):146–152
Pulendran B, Tang H, Denning T (2008) Division of labor, plasticity, and crosstalk between dendritic cell subsets. Curr Opin Immunol 20(1):61–67
Aspord C et al (2007) Breast cancer instructs dendritic cells to prime interleukin 13-secreting CD4+T cells that facilitate tumor development. J Exp Med 204(5):1037–1047
Ostrand-Rosenberg S, Grusby MJ, Clements VK (2000) Cutting edge: STAT6-deficient mice have enhanced tumor immunity to primary and metastatic mammary carcinoma. J Immunol 165(11):6015–6019
Wei S et al (2005) Plasmacytoid dendritic cells induce CD8+ regulatory T cells in human ovarian carcinoma. Cancer Res 65(12):5020–5026
Mansfield AS et al (2011) Metastasis to sentinel lymph nodes in breast cancer is associated with maturation arrest of dendritic cells and poor co-localization of dendritic cells and CD8+ T cells. Virchows Arch 459(4):391–398
Kini Bailur J, Gueckel B, Pawelec G (2016) Prognostic impact of high levels of circulating plasmacytoid dendritic cells in breast cancer. J Transl Med 14(1):151
Randolph GJ, Ochando J, Partida-Sanchez S (2008) Migration of dendritic cell subsets and their precursors. Annu Rev Immunol 26:293–316
Blanco P et al (2008) Dendritic cells and cytokines in human inflammatory and autoimmune diseases. Cytokine Growth Factor Rev 19(1):41–52
Mackay CR (2008) Moving targets: cell migration inhibitors as new anti-inflammatory therapies. Nat Immunol 9(9):988–998
Zou YR et al (1998) Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature 393(6685):595–599
Zhao BC et al (2011) CXCR4/SDF-1 axis is involved in lymph node metastasis of gastric carcinoma. World J Gastroenterol 17(19):2389–2396
Hu TH et al (2014) SDF-1/CXCR4 promotes epithelial-mesenchymal transition and progression of colorectal cancer by activation of the Wnt/beta-catenin signaling pathway. Cancer Lett 354(2):417–426
Liang Z et al (2005) Silencing of CXCR4 blocks breast cancer metastasis. Cancer Res 65(3):967–971
Kang H et al (2005) Stromal cell derived factor-1: its influence on invasiveness and migration of breast cancer cells in vitro, and its association with prognosis and survival in human breast cancer. Breast Cancer Res 7(4):R402–R410
Shim H et al (2006) Lower expression of CXCR4 in lymph node metastases than in primary breast cancers: potential regulation by ligand-dependent degradation and HIF-1alpha. Biochem Biophys Res Commun 346(1):252–258
Walenkamp AME et al (2017) CXCR4 ligands: the next big hit? J Nucl Med 58(Suppl 2):77S–82S
El-Ghonaimy EA et al (2015) Positive lymph-node breast cancer patients—activation of NF-kappaB in tumor-associated leukocytes stimulates cytokine secretion that promotes metastasis via C–C chemokine receptor CCR7. FEBS J 282(2):271–282
Reizis B et al (2011) Plasmacytoid dendritic cells: recent progress and open questions. Annu Rev Immunol 29:163–183
Li YM et al (2004) Upregulation of CXCR4 is essential for HER2-mediated tumor metastasis. Cancer Cell 6(5):459–469
Helbig G et al (2003) NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem 278(24):21631–21638
Chua AW et al (2010) Butein downregulates chemokine receptor CXCR4 expression and function through suppression of NF-kappaB activation in breast and pancreatic tumor cells. Biochem Pharmacol 80(10):1553–1562
Pandey MK et al (2007) Butein, a tetrahydroxychalcone, inhibits nuclear factor (NF)-kappaB and NF-kappaB-regulated gene expression through direct inhibition of IkappaBalpha kinase beta on cysteine 179 residue. J Biol Chem 282(24):17340–17350
Balkwill F (2006) TNF-alpha in promotion and progression of cancer. Cancer Metastasis Rev 25(3):409–416
Yang F, Zhao Z, Zhao N (2017) Clinical implications of tumor necrosis factor receptor 2 in breast cancer. Oncol Lett 14(2):2393–2398
Acknowledgements
This work was conducted in Cancer Biology Research Laboratory (CBRL), Department of Zoology, Faculty of Science, Cairo University, Egypt. We thank Dr. Eslam El-Ghonaimy and Dr. Hossam Taha Mohamed (Researchers at CBRL, Faculty of Science, Cairo University) for their help in Western blotting and qPCR experiments, respectively.
Funding
This study was funded by Cairo University, Scientific Research Sector (SAI, HH, SE, MMM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest regarding the publication of this article.
Ethical approval
All procedures performed in the study were in accordance with the ethical standards of Ain shams University research ethics committee, the Egyptian national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gadalla, R., Hassan, H., Ibrahim, S.A. et al. Tumor microenvironmental plasmacytoid dendritic cells contribute to breast cancer lymph node metastasis via CXCR4/SDF-1 axis. Breast Cancer Res Treat 174, 679–691 (2019). https://doi.org/10.1007/s10549-019-05129-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05129-8